Scalable Synthesis of 2-Nitro-4-Bromobenzaldehyde via Diazonium Chemistry for Commercial API Production
The pharmaceutical industry continuously seeks robust synthetic routes for critical intermediates that balance safety, efficiency, and scalability. Patent CN115417771A introduces a transformative preparation method for 2-nitro-4-bromobenzaldehyde, a pivotal building block in the synthesis of various bioactive molecules and agrochemical agents. This innovation addresses long-standing safety concerns associated with traditional oxidation protocols by leveraging a diazonium coupling strategy. By shifting the synthetic paradigm from hazardous oxidative cleavage to a controlled diazotization-hydrolysis sequence, the technology offers a pathway that is not only chemically elegant but also industrially viable. For R&D teams and supply chain managers, this represents a significant opportunity to secure a more reliable pharmaceutical intermediate supplier relationship, ensuring consistent quality while mitigating the risks inherent in handling explosive reagents during large-scale production campaigns.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-nitro-4-bromobenzaldehyde has relied heavily on the oxidation of corresponding toluene derivatives, a process fraught with significant operational hazards and inefficiencies. As illustrated in the reaction scheme below, conventional routes often employ sodium periodate as the terminal oxidant to convert the methyl group into an aldehyde functionality. This reagent is classified as easily explosive, imposing severe restrictions on storage, handling, and transportation, which drastically inflates logistical costs and safety compliance burdens. Moreover, the reaction conditions required for effective oxidation are often harsh, leading to difficult post-processing and purification challenges that result in substantial product loss. The amplification effect in these oxidative processes is notoriously poor, meaning that yields observed in the laboratory frequently collapse when attempted in multi-kilogram reactors, rendering the method economically unfeasible for commercial scale-up of complex pharmaceutical intermediates.
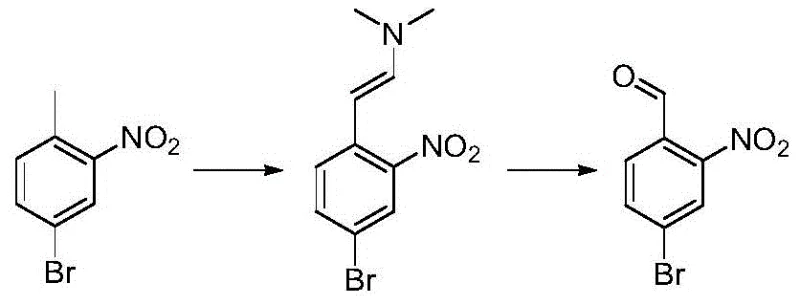
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes 2-nitro-4-bromoaniline as the starting material, bypassing the dangerous oxidation step entirely. This innovative route proceeds through the formation of a diazonium salt, which subsequently couples with formaldehyde oxime in the presence of a specific catalyst system to generate the target aldehyde after hydrolysis. This strategic shift eliminates the need for hazardous oxidants like sodium periodate, thereby fundamentally altering the safety profile of the manufacturing process. The reaction conditions are significantly milder, typically operating between 0°C and 10°C for the coupling phase, which allows for precise thermal control and minimizes the formation of unwanted by-products. Consequently, this approach not only enhances operator safety but also streamlines the downstream purification process, leading to higher overall recovery rates and a more sustainable manufacturing footprint for high-purity pharmaceutical intermediates.
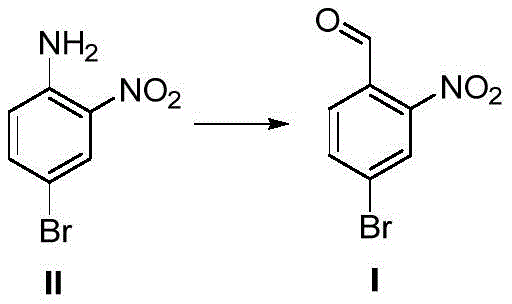
Mechanistic Insights into Diazonium Coupling and Hydrolysis
The core of this technological advancement lies in the precise orchestration of the diazotization and subsequent coupling reactions. Initially, 2-nitro-4-bromoaniline undergoes diazotization with sodium nitrite under acidic conditions (typically hydrochloric acid) at low temperatures (0-10°C) to form the reactive diazonium salt. This intermediate is inherently unstable and must be generated in situ to prevent decomposition. The addition of formaldehyde oxime in the presence of a copper-based catalyst system (comprising copper sulfate, sodium sulfite, and ammonium chloride) facilitates a nucleophilic attack or radical coupling mechanism that installs the one-carbon aldehyde precursor. The pH of the reaction system is critically maintained between 5 and 7 using sodium acetate to ensure optimal reactivity of the oxime while stabilizing the diazonium species. This delicate balance prevents premature hydrolysis or side reactions, ensuring that the carbon-nitrogen bond formation proceeds with high selectivity, which is paramount for achieving the reported high yields in fine chemical manufacturing.
Following the coupling event, the resulting intermediate undergoes acid-catalyzed hydrolysis to reveal the final aldehyde group. This step involves heating the reaction mixture to approximately 100°C in the presence of concentrated hydrochloric acid. The hydrolysis mechanism effectively cleaves the nitrogen-containing moiety, releasing nitrogen gas and yielding the desired 2-nitro-4-bromobenzaldehyde. From an impurity control perspective, this two-step sequence is superior because it avoids the over-oxidation issues common in direct methyl oxidation, where carboxylic acid by-products are difficult to separate. The use of steam distillation and recrystallization in the workup further refines the product, consistently delivering purity levels greater than 98%. This rigorous control over the reaction pathway ensures that the impurity profile remains clean, a critical requirement for customers seeking a reliable pharmaceutical intermediate supplier for GMP-grade applications.
How to Synthesize 2-Nitro-4-Bromobenzaldehyde Efficiently
The implementation of this synthesis requires careful attention to temperature control and reagent stoichiometry to maximize the benefits of the patented process. The procedure begins with the preparation of the diazonium salt, followed immediately by the coupling with formaldehyde oxime under catalytic conditions. Operators must monitor the reaction progress via TLC to ensure complete consumption of the starting aniline before proceeding to the hydrolysis stage. The detailed standardized synthesis steps, including specific mass ratios and timing for reagent addition, are outlined in the structured guide below to assist technical teams in replicating this high-yielding protocol.
- Diazotization: React 2-nitro-4-bromoaniline with sodium nitrite in aqueous hydrochloric acid at 0-10°C to form the stable diazonium salt intermediate.
- Coupling Reaction: Introduce formaldehyde oxime and a copper-based catalyst system to the diazonium solution, maintaining pH 5-7 and temperature at 0-10°C.
- Hydrolysis and Purification: Acidify the mixture and reflux at 100°C to hydrolyze the intermediate, followed by distillation and recrystallization to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route translates into tangible strategic advantages beyond mere chemical curiosity. By eliminating the dependency on sodium periodate, manufacturers can significantly reduce the regulatory overhead and insurance costs associated with storing and transporting explosive materials. This simplification of the raw material portfolio enhances supply chain resilience, as 2-nitro-4-bromoaniline and formaldehyde oxime are commodity chemicals with stable global availability. Furthermore, the improved yield stability and simplified purification workflow directly contribute to cost reduction in pharmaceutical intermediate manufacturing by minimizing waste generation and reducing the consumption of solvents and energy required for extensive chromatographic separations. These factors collectively lower the total cost of ownership for the intermediate, allowing downstream API producers to optimize their margins without compromising on quality standards.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous oxidants like sodium periodate removes a major cost driver from the bill of materials. Additionally, the higher reaction yield (reported up to 80%) means that less starting material is required to produce the same amount of final product, effectively lowering the unit cost. The simplified workup, which relies on distillation and recrystallization rather than complex column chromatography, reduces solvent consumption and labor hours, leading to substantial operational savings that can be passed down the supply chain.
- Enhanced Supply Chain Reliability: Sourcing explosive reagents often involves long lead times and strict regulatory approvals, creating bottlenecks in production schedules. By switching to a route that utilizes stable, non-hazardous reagents, manufacturers can achieve more predictable production cycles and reducing lead time for high-purity pharmaceutical intermediates. The robustness of the process against scale-up effects ensures that supply commitments can be met consistently, even when demand surges, providing a stable foundation for long-term procurement planning.
- Scalability and Environmental Compliance: The mild reaction conditions (0-10°C and 100°C) are compatible with standard glass-lined or stainless steel reactors found in most multipurpose chemical plants, facilitating easy technology transfer from pilot to commercial scale. The avoidance of heavy metal oxidants and the generation of fewer hazardous by-products align with modern green chemistry principles, simplifying wastewater treatment and environmental compliance. This ease of commercial scale-up ensures that the technology can meet global demand volumes without requiring specialized or capital-intensive infrastructure upgrades.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2-nitro-4-bromobenzaldehyde using this advanced methodology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits for potential partners and technical evaluators.
Q: Why is the diazonium route preferred over sodium periodate oxidation for 2-nitro-4-bromobenzaldehyde?
A: The diazonium route eliminates the use of sodium periodate, an easily explosive reagent that poses significant safety risks and complicates industrial scale-up. Furthermore, the new method offers superior yield stability and simplified post-processing compared to the harsh oxidation conditions of conventional methods.
Q: What is the expected purity and yield of this synthetic method?
A: According to patent data, this optimized process achieves a molar yield of approximately 80% with a final product purity exceeding 98% as confirmed by gas chromatography, making it highly suitable for stringent pharmaceutical applications.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes standard reactor equipment and avoids extreme pressure or cryogenic conditions. The mild temperature range (0-10°C for coupling, 100°C for hydrolysis) and aqueous workup facilitate safe and efficient commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Nitro-4-Bromobenzaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 2-nitro-4-bromobenzaldehyde meets the exacting standards required by the global pharmaceutical industry. Our facility is equipped to handle the specific thermal and safety requirements of diazonium chemistry, providing a secure and efficient manufacturing environment for your most sensitive projects.
We invite you to collaborate with us to leverage this innovative synthetic route for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your project's economic viability and accelerate your time to market.
