Advanced Condensed Ring Liquid Crystal Compounds for High-Speed Display Manufacturing
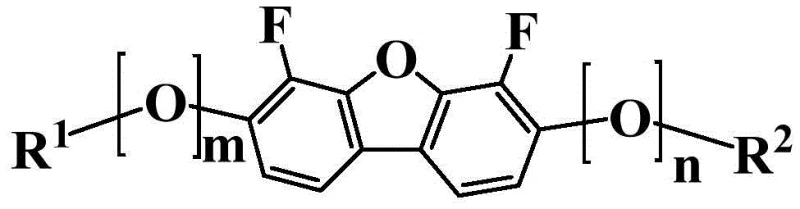
The rapid evolution of the flat-panel display industry demands continuous innovation in the underlying material science, particularly for liquid crystal display (LCD) elements that require high-speed response and low power consumption. Patent CN110650948A introduces a groundbreaking class of liquid crystal compounds characterized by a unique condensed ring structure, specifically designed to address the limitations of prior art materials. This technology represents a significant leap forward in the development of organic electronic materials, offering a robust solution for manufacturers seeking to enhance the performance metrics of their display panels. The core innovation lies in the molecular architecture which simultaneously achieves a high nematic-isotropic phase transition temperature (Tni) and a large absolute value of dielectric constant anisotropy (|Δε|). For R&D directors and procurement specialists in the display sector, understanding the implications of this patent is crucial for securing a competitive edge in the market. By leveraging these advanced intermediates, companies can formulate liquid crystal compositions that exhibit superior stability against external factors such as moisture, heat, and light, thereby extending the operational lifespan of the final electronic devices.
Furthermore, the versatility of the general formula (i) described in the patent allows for extensive structural modulation through various substituents, including alkyl, alkenyl, and fluorinated groups. This flexibility enables precise tuning of physical properties such as rotational viscosity (γ1) and refractive index anisotropy (Δn), which are paramount for optimizing display performance in different driving modes like VA, IPS, and FFS. As a reliable liquid crystal material supplier, recognizing the potential of these condensed ring compounds is essential for staying ahead in the supply chain of high-tech electronic components. The patent not only covers the compounds themselves but also details efficient manufacturing methods and intermediates, providing a comprehensive roadmap for industrial production. This holistic approach ensures that the transition from laboratory synthesis to commercial-scale manufacturing is seamless, mitigating risks associated with process scalability and yield consistency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of liquid crystal materials has been constrained by the trade-off between thermal stability and dielectric properties. Conventional compounds possessing a dibenzofuran structure, as cited in prior art documents like German Patent Application Publication No. 102015002298, often fail to achieve sufficiently high clearing points (Tni). This limitation restricts the operating temperature range of the liquid crystal composition, potentially leading to display failures under extreme environmental conditions. Moreover, achieving a large negative dielectric anisotropy (Δε) without compromising other physical parameters has been a persistent challenge. Traditional synthesis routes for such complex aromatic systems frequently involve harsh reaction conditions, multiple protection-deprotection steps, and the use of expensive or toxic reagents that complicate purification and waste management. These inefficiencies translate into higher production costs and longer lead times, creating bottlenecks for procurement managers aiming to optimize the bill of materials for display panels. Additionally, the impurity profiles of conventionally synthesized liquid crystals can negatively impact the voltage holding ratio and resistance of the final display element, necessitating rigorous and costly quality control measures.
The Novel Approach
The methodology outlined in CN110650948A overcomes these hurdles through a sophisticated design of the condensed ring core. By strategically incorporating specific linking groups and terminal substituents, the new compounds demonstrate a marked improvement in Tni while maintaining a large |Δε|. This dual enhancement allows formulators to increase the proportion of non-polar compounds with low rotational viscosity in the mixture, ultimately resulting in faster response times for the display. The novel approach also streamlines the synthetic pathway, utilizing transition metal-catalyzed cross-coupling reactions followed by an elegant intramolecular cyclization step. This strategy reduces the number of synthetic steps compared to traditional methods, thereby minimizing material loss and solvent consumption. From a supply chain perspective, this efficiency is transformative, as it simplifies the manufacturing process and enhances the overall reliability of the supply of high-purity liquid crystal intermediates. The ability to fine-tune the molecular structure through variables such as the nature of the ring systems (cyclohexylene, phenylene, naphthalene) and the type of linking groups (ether, thioether, ester) provides unprecedented control over the material's performance characteristics.
Mechanistic Insights into Transition Metal-Catalyzed Cyclization
The synthesis of these high-performance liquid crystal compounds relies heavily on precise organometallic chemistry, specifically leveraging transition metal catalysis to construct the complex condensed ring framework. The core mechanistic pathway involves a sequential process starting with the boronation of a precursor aromatic compound. This step typically employs strong organometallic bases such as sec-butyllithium or lithium diisopropylamide (LDA) at cryogenic temperatures ranging from -100°C to -20°C to generate a reactive anionic species. This anion is then trapped with a trialkyl borate, such as trimethyl borate or triisopropyl borate, to form the corresponding boronic acid derivative. The subsequent step is a palladium-catalyzed cross-coupling reaction, likely a Suzuki-Miyaura type coupling, where the boronated intermediate reacts with a halogenated aromatic partner. Preferred catalysts include tetrakis(triphenylphosphine)palladium(0) or [1,1'-bis(diphenylphosphino)ferrocene]dichloropalladium(II), which facilitate the formation of the carbon-carbon bond under mild conditions in solvents like tetrahydrofuran or toluene.
Following the coupling reaction, the critical intramolecular cyclization takes place to close the condensed ring system. This transformation is driven by the deprotonation of a specific hydroxyl or thiol group (-Yi3-H) on the coupled intermediate using a strong base such as sodium hydride, potassium carbonate, or tripotassium phosphate. The resulting anion undergoes a nucleophilic attack on an adjacent electrophilic center, effectively closing the ring and establishing the rigid condensed structure that defines the compound's liquid crystalline properties. This cyclization step is pivotal as it locks the molecular conformation, enhancing the thermal stability and mesomorphic behavior of the final product. The reaction conditions for this step are carefully optimized, typically occurring at temperatures between 30°C and 120°C in polar aprotic solvents like DMF or ether-based solvents. Understanding this mechanism is vital for R&D teams aiming to replicate or scale this chemistry, as precise control over stoichiometry, temperature, and addition rates is required to minimize side reactions and ensure high purity of the target liquid crystal intermediate.
How to Synthesize Condensed Ring Liquid Crystal Compounds Efficiently
Implementing the synthesis of these advanced materials requires a disciplined approach to process chemistry, focusing on reproducibility and safety. The patented method offers a clear pathway that integrates standard fine chemical operations with specialized organometallic techniques. To achieve the high purity levels demanded by the display industry, each reaction step must be monitored closely, and workup procedures should be designed to efficiently remove metal catalysts and inorganic salts. The following guide outlines the strategic phases of the synthesis, emphasizing the critical control points that determine the success of the manufacturing campaign. By adhering to these principles, production teams can ensure consistent quality and yield, which are essential for meeting the stringent specifications of global display panel manufacturers.
- Perform boronation of the precursor compound using organometallic reagents like sec-butyllithium or LDA at low temperatures (-78°C to -40°C) followed by reaction with trialkyl borate.
- Execute a transition metal-catalyzed coupling reaction between the boronated intermediate and a halogenated aromatic compound using palladium catalysts such as Pd(PPh3)4 or Pd(dppf)Cl2.
- Conduct an intramolecular cyclization reaction by deprotonating the coupled intermediate with a strong base like sodium hydride or potassium carbonate to form the final condensed ring structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN110650948A presents substantial opportunities for optimizing the sourcing strategy of liquid crystal materials. The streamlined synthetic route directly contributes to cost reduction in display material manufacturing by eliminating unnecessary synthetic steps and reducing the consumption of expensive reagents. The use of commercially available catalysts and solvents further lowers the barrier to entry for production, ensuring that the supply chain remains resilient against fluctuations in raw material availability. Moreover, the improved efficiency of the process means that less waste is generated per unit of product, aligning with increasingly strict environmental regulations and sustainability goals. This operational efficiency translates into a more stable supply of high-quality intermediates, reducing the risk of production delays for downstream display assembly lines.
- Cost Reduction in Manufacturing: The novel synthesis pathway significantly lowers the overall cost of goods sold by improving atom economy and reducing the number of purification stages required. By avoiding complex protection group chemistry and utilizing robust catalytic systems, the process minimizes material loss and energy consumption. This economic advantage allows buyers to negotiate better pricing structures while maintaining high margins on the final display products. Additionally, the reduced need for exotic reagents simplifies inventory management and lowers storage costs associated with hazardous chemicals.
- Enhanced Supply Chain Reliability: The reliance on standard chemical building blocks and widely used catalytic technologies ensures that the supply chain is not dependent on single-source or geographically constrained raw materials. This diversification of supply sources mitigates the risk of disruptions caused by geopolitical events or logistical bottlenecks. Furthermore, the scalability of the process means that suppliers can rapidly ramp up production volumes to meet surging demand from the consumer electronics sector without compromising on quality or delivery timelines.
- Scalability and Environmental Compliance: The manufacturing method is inherently designed for scale-up, with reaction conditions that are easily transferable from pilot plants to large-scale commercial reactors. The use of less toxic solvents and the generation of manageable waste streams facilitate compliance with environmental health and safety standards. This proactive approach to environmental stewardship not only reduces regulatory risks but also enhances the brand reputation of companies adopting these greener manufacturing practices in the competitive electronic materials market.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical specifications and commercial viability of the liquid crystal compounds disclosed in the patent. These insights are derived directly from the experimental data and technical descriptions provided in the documentation, offering clarity for stakeholders evaluating the integration of these materials into their product portfolios. Understanding these details is key to making informed decisions about material selection and process adoption.
Q: What are the key performance advantages of the compounds described in CN110650948A?
A: The compounds exhibit a high nematic-isotropic phase transition temperature (Tni) and a large negative dielectric constant anisotropy (Δε), which are critical for reducing rotational viscosity and enabling fast response times in VA and IPS mode liquid crystal displays.
Q: How does the synthesis route ensure scalability for industrial production?
A: The manufacturing method utilizes widely available transition metal catalysts and standard organic solvents like THF and toluene. The reaction conditions, ranging from cryogenic deprotonation to moderate heating for cyclization, are compatible with existing fine chemical infrastructure, facilitating commercial scale-up.
Q: Why is the condensed ring structure significant for modern display technology?
A: Unlike conventional dibenzofuran structures which may suffer from insufficient clearing points, the novel condensed ring architecture provides enhanced thermal stability and optimal dielectric properties, allowing for the formulation of liquid crystal mixtures that maintain stable nematic phases over wide temperature ranges.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Liquid Crystal Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced materials play in the next generation of display technologies. Our expertise in process development and scale-up positions us as an ideal partner for bringing the innovations of CN110650948A to the global market. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for high-performance electronic applications. We are committed to delivering value through technical excellence and operational reliability, helping our clients navigate the complexities of the specialty chemical supply chain.
We invite you to collaborate with us to explore the full potential of these condensed ring liquid crystal compounds for your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your production volume and quality requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our capabilities can drive efficiency and innovation in your display material sourcing strategy. Together, we can accelerate the development of superior display solutions that meet the evolving demands of the global market.
