Advanced Total Synthesis and Commercial Scale-Up of Complex Benzofuranone Intermediates
Introduction to Patented Synthetic Technology
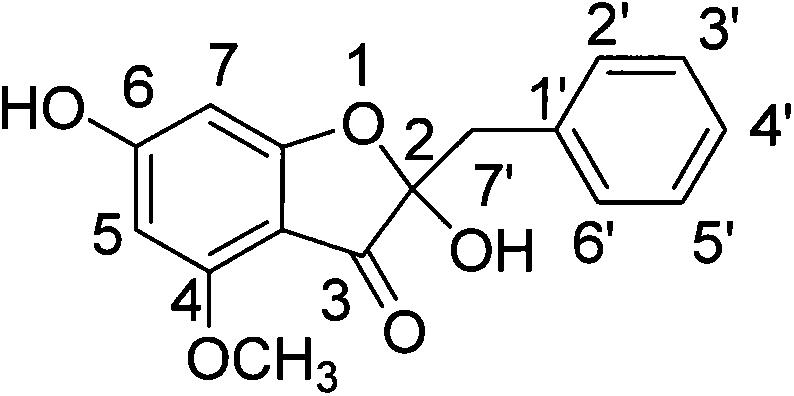
The pharmaceutical and fine chemical industries constantly demand high-purity reference standards to ensure the quality and safety of natural product extracts, particularly in the authentication of propolis. Patent CN103360352A discloses a groundbreaking total synthesis method for 4-methoxy-2,6-dihydroxy-2-benzyl-3(2H)-benzofuranone, a novel aurone derivative originally isolated from poplar gum. This compound serves as a critical reference substance for quantifying poplar gum adulteration in propolis, addressing a significant quality control gap in the nutraceutical sector. The disclosed methodology represents a significant leap forward in synthetic organic chemistry, moving away from extraction-dependent supply chains which are plagued by low natural abundance and complex isolation procedures. By establishing a fully synthetic route, manufacturers can secure a stable supply of this high-value intermediate independent of seasonal botanical variations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aurone and benzofuranone derivatives has relied heavily on the oxidative cyclization of 2-hydroxychalcones. Conventional literature methods frequently employ stoichiometric amounts of toxic heavy metal oxidants such as thallium(III) nitrate or mercury(II) acetate to effect the ring closure. From a commercial manufacturing perspective, these reagents present severe liabilities, including high disposal costs for hazardous waste, stringent environmental regulatory hurdles, and significant risks to operator safety. Furthermore, traditional aldol condensation approaches often suffer from poor regioselectivity when attempting to introduce specific substitution patterns like the 4-methoxy-2,6-dihydroxy motif found in the target molecule. Direct functionalization of the benzofuranone core is notoriously difficult due to the similar reactivity of the phenolic hydroxyl groups, often leading to complex mixtures of regioisomers that are economically unviable to separate on an industrial scale.
The Novel Approach
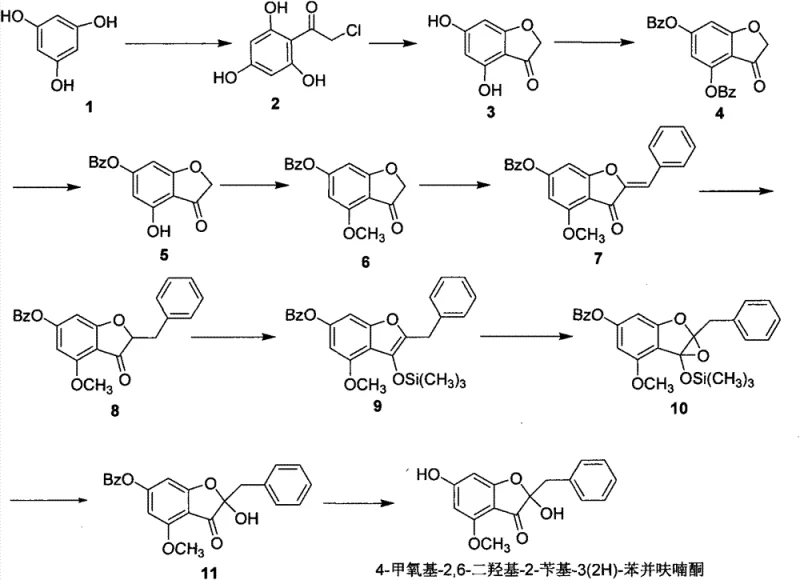
The patented technology introduces a sophisticated, stepwise strategy that circumvents these historical bottlenecks by utilizing a protective group manifold to achieve precise regiocontrol. Instead of relying on toxic oxidative cyclization, the synthesis constructs the 3(2H)-benzofuranone mother nucleus via a benign base-catalyzed intramolecular cyclization of an alpha-halo ketone precursor. The core innovation lies in the differential protection of the phenolic hydroxyls; by initially protecting both the 4 and 6 positions with benzyl groups, followed by selective catalytic hydrogenation to remove only the 4-position benzyl group, the process creates a unique chemical environment. This allows for the specific methylation of the 4-hydroxyl group while leaving the 6-position available for final deprotection. This logical disconnection transforms a chemically ambiguous transformation into a highly predictable and controllable sequence, ensuring high purity and yield.
Mechanistic Insights into Regioselective Functionalization
The success of this synthesis hinges on the meticulous control of electronic and steric factors during the functionalization of the benzofuranone core. The initial Friedel-Crafts acylation of phloroglucinol with chloroacetyl chloride is directed by the strong activating nature of the three hydroxyl groups, favoring acylation at the position flanked by two hydroxyls to form the 2,4,6-trihydroxyacetophenone derivative. Subsequent cyclization under basic conditions proceeds via an intramolecular SN2 mechanism where the phenoxide anion attacks the alpha-carbon of the chloroacetyl side chain, closing the five-membered furanone ring. The critical mechanistic step occurs during the selective hydrogenation of the dibenzylated intermediate. Under controlled conditions using 10% Pd/C, the benzyl ether at the 4-position is cleaved preferentially over the 6-position ether, likely due to subtle steric differences or coordination effects with the adjacent carbonyl group, although the exact mechanistic rationale involves complex surface catalysis phenomena. This selectivity is the linchpin of the entire route, enabling the subsequent installation of the methoxy group exclusively at the 4-position using methyl iodide and base.
Following the establishment of the oxygenation pattern, the installation of the 2-benzyl-2-hydroxy motif requires a departure from simple alkylation. The process employs an aldol condensation with benzaldehyde to generate an exocyclic double bond, which is subsequently reduced to the saturated benzyl group. To introduce the crucial 2-hydroxyl group, the ketone is converted into a silyl enol ether using LDA and TMSCl at cryogenic temperatures (-70°C) to ensure kinetic control. This enol ether is then subjected to epoxidation using m-CPBA. The resulting epoxy intermediate undergoes a regioselective ring-opening reaction upon treatment with TBAF. The fluoride ion not only cleaves the trimethylsilyl protecting group but also triggers the rearrangement and hydrolysis of the epoxide, effectively installing the tertiary hydroxyl group at the C2 position. This cascade of enolization, epoxidation, and ring-opening demonstrates a high level of synthetic sophistication, allowing for the construction of a quaternary carbon center with high fidelity.
How to Synthesize 4-Methoxy-2,6-Dihydroxy-2-Benzyl-3(2H)-Benzofuranone Efficiently
The synthesis of this complex benzofuranone derivative is achieved through an 11-step linear sequence that prioritizes reagent availability and operational simplicity. The process begins with the acylation of commodity-grade phloroglucinol, followed by cyclization to establish the heterocyclic core. The middle stages focus on the precise manipulation of oxidation states and protecting groups to differentiate the phenolic positions. The final stages involve carbon-carbon bond formation via aldol condensation and the stereoselective installation of the C2 hydroxyl group via an epoxide intermediate. For process chemists looking to implement this route, attention to detail in the selective hydrogenation and cryogenic enolization steps is paramount to maintaining high overall yield. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and workup procedures, are outlined in the guide below.
- Perform Friedel-Crafts acylation of phloroglucinol with chloroacetyl chloride using AlCl3 to form the acetophenone intermediate.
- Cyclize the intermediate under basic conditions to form the 3(2H)-benzofuranone core, followed by benzyl protection of phenolic hydroxyls.
- Execute selective hydrogenation to remove the 4-position benzyl group, methylate the 4-hydroxyl, and proceed through aldol condensation and epoxidation to finalize the structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from extraction-based sourcing to this total synthesis route offers transformative advantages in terms of cost stability and supply security. The reliance on natural extraction from poplar gum inherently limits supply volume and subjects pricing to agricultural volatility and geopolitical factors affecting raw material harvesting. In contrast, this synthetic route utilizes widely available commodity chemicals such as phloroglucinol, chloroacetyl chloride, and benzaldehyde as starting materials. These feedstocks are produced on a massive global scale, ensuring that the supply chain is robust and resistant to shortages. Furthermore, the elimination of complex chromatographic purification steps typically required for natural product isolation significantly reduces processing time and solvent consumption, leading to substantial cost savings in manufacturing overhead.
- Cost Reduction in Manufacturing: The synthetic pathway is designed to minimize the use of exotic or prohibitively expensive reagents. By replacing toxic heavy metal oxidants like thallium or mercury with catalytic hydrogenation and mild peracid oxidation, the process eliminates the need for specialized hazardous waste treatment facilities and expensive metal scavenging resins. The use of heterogeneous catalysts like Pd/C allows for easy recovery and reuse, further driving down the cost of goods sold. Additionally, the high regioselectivity of the protection-deprotection strategy minimizes the formation of difficult-to-separate isomers, reducing yield losses associated with purification and thereby enhancing the overall economic efficiency of the production line.
- Enhanced Supply Chain Reliability: Adopting this synthetic route decouples the supply of this critical reference standard from the seasonal and biological constraints of plant cultivation. Natural extraction yields are often low and variable, creating bottlenecks for quality control laboratories that require consistent batches of reference material. A synthetic manufacturing model guarantees batch-to-batch consistency in terms of purity and impurity profile, which is essential for regulatory compliance in pharmaceutical testing. The ability to produce the compound on demand ensures that lead times are drastically shortened, allowing downstream users to maintain lean inventory levels without risking stockouts of essential analytical standards.
- Scalability and Environmental Compliance: The unit operations selected for this synthesis are inherently scalable and align with modern green chemistry principles. Reactions such as Friedel-Crafts acylation and base-catalyzed cyclization are well-understood processes that can be safely transferred from gram-scale laboratory flasks to multi-ton industrial reactors. The avoidance of persistent organic pollutants and heavy metals simplifies the environmental permitting process and reduces the facility's ecological footprint. This compliance advantage is increasingly valuable as global regulations on chemical manufacturing become more stringent, future-proofing the supply chain against regulatory shocks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 4-methoxy-2,6-dihydroxy-2-benzyl-3(2H)-benzofuranone. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this intermediate into their quality control workflows or product development pipelines.
Q: What are the critical challenges in synthesizing 4-methoxy-2,6-dihydroxy-2-benzyl-3(2H)-benzofuranone?
A: The primary challenge lies in achieving regioselective substitution on the benzofuranone ring. Specifically, differentiating between the 4 and 6 hydroxyl positions to install a methoxy group at position 4 while retaining a hydroxyl at position 6 requires a strategic protection-deprotection sequence using benzyl groups, as direct methylation often lacks selectivity.
Q: Why is this synthetic route preferred over traditional aurone synthesis methods?
A: Traditional aurone synthesis often relies on oxidative cyclization of chalcones using toxic heavy metals like thallium(III) nitrate or mercury(II) acetate. This patented route avoids hazardous oxidants by utilizing catalytic hydrogenation and mild epoxidation with m-CPBA, significantly improving environmental compliance and operator safety.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes robust and scalable unit operations such as Friedel-Crafts acylation, standard base-catalyzed cyclization, and heterogeneous catalytic hydrogenation (Pd/C). These are well-established industrial processes that facilitate easy scale-up from laboratory to multi-ton production without requiring exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Methoxy-2,6-Dihydroxy-2-Benzyl-3(2H)-Benzofuranone Supplier
At NINGBO INNO PHARMCHEM, we understand that the integrity of your quality control processes depends on the purity and reliability of your reference standards. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether for R&D screening or routine QC testing. Our state-of-the-art rigorous QC labs and adherence to stringent purity specifications guarantee that every batch of 4-methoxy-2,6-dihydroxy-2-benzyl-3(2H)-benzofuranone meets the highest international standards, providing you with the confidence needed for accurate propolis authentication.
We invite you to collaborate with us to optimize your supply chain for this critical intermediate. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how our synthetic expertise can reduce your overall procurement costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us support your commitment to quality and excellence in the pharmaceutical and nutraceutical industries.
