Advanced Synthesis of Venlafaxine Amine Impurity I for Pharmaceutical Quality Control
Advanced Synthesis of Venlafaxine Amine Impurity I for Pharmaceutical Quality Control
The pharmaceutical industry faces increasing regulatory pressure to identify and quantify trace impurities in active pharmaceutical ingredients (APIs), particularly for widely used antidepressants like Venlafaxine Hydrochloride. Patent CN112778143A introduces a groundbreaking preparation method for Venlafaxine Amine Impurity I, a critical related substance that must be strictly monitored to ensure drug safety and efficacy. This technology addresses a significant gap in the market where high-purity reference standards were previously difficult to source or prohibitively expensive. By establishing a robust synthetic route starting from common industrial chemicals, this innovation enables pharmaceutical manufacturers to implement rigorous quality control protocols without relying on scarce external references.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
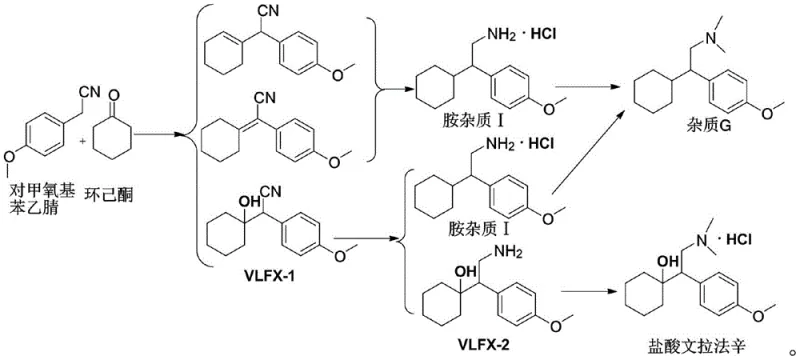
Historically, the acquisition of Venlafaxine Amine Impurity I for analytical purposes has been fraught with logistical and economic challenges. Traditional approaches often relied on the methylation of Amine Impurity G, a reference standard listed in the European Pharmacopoeia. However, Impurity G is characterized by extremely high market prices and limited availability, creating a bottleneck for quality assurance laboratories. Furthermore, the methylation process itself introduces additional complexity and potential side reactions, which can compromise the purity of the final reference material. This dependency on scarce precursors not only inflates the cost of goods sold for analytical standards but also introduces significant supply chain volatility, making consistent batch-to-batch comparison difficult for generic drug manufacturers.
The Novel Approach
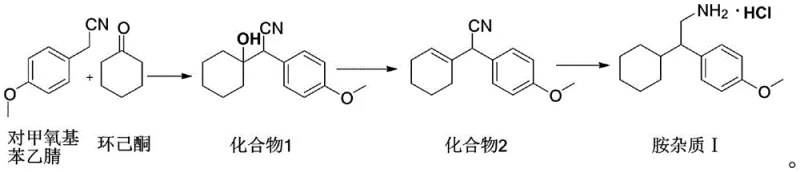
The methodology disclosed in CN112778143A revolutionizes this landscape by proposing a direct, three-step synthesis starting from p-methoxybenzyl acetonitrile and cyclohexanone. This route completely bypasses the need for expensive pharmacopoeial standards, utilizing instead abundant and cost-effective raw materials that are staples in the fine chemical industry. The process is designed for high efficiency, achieving an overall yield that makes commercial production of the impurity standard economically viable. By controlling the reaction parameters precisely, the inventors have demonstrated the ability to produce the target amine impurity with a purity exceeding 99 percent, which is essential for its role as a quantitative reference in HPLC analysis. This shift from sourcing to synthesizing represents a strategic advantage for any organization aiming to secure their supply of critical quality control materials.
Mechanistic Insights into the Three-Step Synthetic Route
The core of this technology lies in a carefully orchestrated sequence of condensation, dehydration, and reduction reactions. The process initiates with a phase-transfer catalyzed condensation between p-methoxybenzyl acetonitrile and cyclohexanone. In this step, a strong base such as sodium hydroxide deprotonates the alpha-carbon of the nitrile, facilitating a nucleophilic attack on the ketone carbonyl. The use of a phase transfer catalyst, preferably tetrabutylammonium bromide, is critical here as it enhances the interaction between the organic and aqueous phases, driving the reaction to completion and yielding the hydroxynitrile intermediate (Compound 1) with exceptional purity. This mechanistic precision ensures that the subsequent steps begin with a high-quality substrate, minimizing the propagation of impurities.
Following the initial condensation, the hydroxynitrile undergoes an acid-catalyzed dehydration to form an unsaturated nitrile (Compound 2). This transformation is typically mediated by concentrated sulfuric acid, which promotes the elimination of the hydroxyl group to generate the double bond within the cyclohexyl ring. The final and most crucial step involves the catalytic hydrogenation of this unsaturated nitrile. Using a palladium on carbon (Pd/C) catalyst under hydrogen pressure, the nitrile group is reduced to a primary amine, and the double bond is simultaneously saturated. This dual reduction capability is vital, as it converts the intermediate directly into the target Amine Impurity I structure. The careful selection of reducing agents and conditions prevents over-reduction or the formation of secondary amines, thereby securing the structural integrity required for a reliable reference standard.
How to Synthesize Venlafaxine Amine Impurity I Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. It emphasizes the importance of stoichiometric control and purification techniques such as recrystallization to achieve the necessary >99% purity. The detailed standardized synthesis steps below outline the specific reagents, temperatures, and workup procedures required to replicate this high-yield process effectively.
- Condense p-methoxybenzyl acetonitrile with cyclohexanone using a phase transfer catalyst and alkali to form the hydroxynitrile intermediate.
- Perform acid-catalyzed dehydration of the hydroxynitrile intermediate to generate the unsaturated nitrile compound.
- Execute catalytic hydrogenation reduction of the unsaturated nitrile to yield the final amine impurity with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers profound strategic benefits beyond mere technical feasibility. The primary advantage is the drastic simplification of the supply chain for critical reference materials. By eliminating the dependency on imported, high-cost pharmacopoeial standards, organizations can insulate themselves from global market fluctuations and geopolitical supply disruptions. The raw materials identified in this process are commodity chemicals with stable pricing and widespread availability, ensuring that production can be sustained continuously without the risk of stockouts that plague niche specialty chemical markets.
- Cost Reduction in Manufacturing: The economic impact of switching to this direct synthesis route is substantial. By removing the need to purchase expensive Impurity G precursors, the variable cost of producing the reference standard is significantly lowered. Additionally, the high yields reported in the patent examples mean that less raw material is wasted, further improving the cost-efficiency profile. This reduction in input costs allows for more competitive pricing of the final API or provides internal savings that can be reinvested into R&D initiatives, enhancing the overall financial health of the manufacturing operation.
- Enhanced Supply Chain Reliability: Reliance on single-source suppliers for rare impurities creates a single point of failure in the quality control supply chain. This novel method diversifies the supply base by enabling in-house production or sourcing from multiple fine chemical manufacturers capable of executing the described chemistry. The robustness of the reaction conditions, which do not require exotic catalysts or extreme pressures, means that multiple contract manufacturing organizations (CMOs) can potentially qualify to produce this material, thereby creating a resilient and redundant supply network that guarantees business continuity.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard unit operations such as stirring, filtration, and distillation that are easily transferred from the laboratory to the plant floor. Furthermore, the choice of reagents and solvents aligns well with modern environmental, health, and safety (EHS) standards. The ability to recycle solvents and the absence of heavy metal contaminants in the final product simplify waste treatment protocols. This ease of scale-up ensures that as demand for Venlafaxine grows, the supply of its critical impurity standards can expand in parallel without requiring massive capital investment in specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this route for adoption.
Q: Why is synthesizing Venlafaxine Amine Impurity I directly superior to methylation?
A: Direct synthesis avoids the reliance on expensive and scarce European Pharmacopoeia reference standards (Impurity G), significantly reducing material costs and supply chain risks while achieving over 99% purity.
Q: What are the key reaction conditions for the dehydration step?
A: The dehydration step utilizes concentrated sulfuric acid in dichloromethane at room temperature, ensuring high conversion to the unsaturated nitrile intermediate with minimal byproduct formation.
Q: Can this process be scaled for commercial reference standard production?
A: Yes, the process uses readily available industrial raw materials like cyclohexanone and p-methoxybenzyl acetonitrile, and employs standard unit operations like filtration and recrystallization suitable for scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Venlafaxine Amine Impurity I Supplier
At NINGBO INNO PHARMCHEM, we understand that the integrity of your final drug product depends on the quality of your reference standards. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch of Venlafaxine Amine Impurity I meets the >99% purity threshold demanded by global regulatory bodies.
We invite you to collaborate with us to optimize your supply chain for Venlafaxine impurities. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production volumes. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your quality control objectives efficiently and reliably.
