Optimizing Dasatinib Intermediate Production: A Technical Analysis of Novel Synthetic Routes
Optimizing Dasatinib Intermediate Production: A Technical Analysis of Novel Synthetic Routes
The pharmaceutical landscape is continuously evolving, driven by the demand for more efficient and sustainable manufacturing processes for critical active pharmaceutical ingredients (APIs). A prime example of this technological shift is found in the synthesis of 2-chloro-6-methylaniline, a pivotal building block for the tyrosine kinase inhibitor Dasatinib. As detailed in the recent patent CN112679364B, filed in late 2022, a novel synthetic methodology has been disclosed that addresses long-standing inefficiencies in producing this key pharmaceutical intermediate. This report provides a deep technical dissection of this innovation, offering valuable insights for R&D directors seeking robust pathways, procurement managers aiming for cost optimization, and supply chain leaders focused on scalability. By shifting away from hazardous sulfonation chemistries toward a controlled nitro-reduction and diazotization strategy, this patent outlines a route that promises enhanced safety profiles and improved operational economics for global manufacturers.
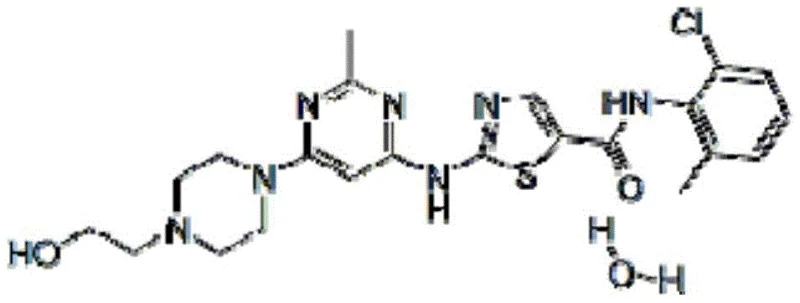
Dasatinib, marketed under the brand name Sprycel, is a second-generation BCR-ABL inhibitor used extensively in the treatment of chronic myelogenous leukemia (CML) and Philadelphia chromosome-positive acute lymphoblastic leukemia (Ph+ ALL). The efficacy of this life-saving medication relies heavily on the quality and availability of its precursors. The structure of Dasatinib, as illustrated above, incorporates the 2-chloro-6-methylaniline motif, making the efficient synthesis of this aniline derivative a matter of strategic importance for the entire oncology drug supply chain. Traditional methods have often been plagued by complex purification steps and environmental concerns, creating bottlenecks that this new patent aims to resolve through rational molecular design and process intensification.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2-chloro-6-methylaniline has relied on routes that involve the blocking of reactive positions using sulfonic acid groups to direct subsequent substitutions. As depicted in the prior art reaction schemes, these conventional methods typically commence with the condensation of o-toluidine and urea to form N,N'-bis(o-tolyl)urea. This intermediate is then subjected to sulfonation using chlorosulfonic acid to introduce a blocking group at the para-position relative to the amino group. While chemically feasible, this approach presents severe drawbacks for modern manufacturing environments. The use of chlorosulfonic acid introduces significant safety hazards due to its extreme corrosivity and the release of toxic hydrogen chloride gas during the reaction. Furthermore, the subsequent removal of the sulfonic acid blocking group requires high-temperature hydrolysis with concentrated sulfuric acid, a process that demands specialized, corrosion-resistant equipment and generates substantial amounts of acidic wastewater that is difficult and costly to treat.
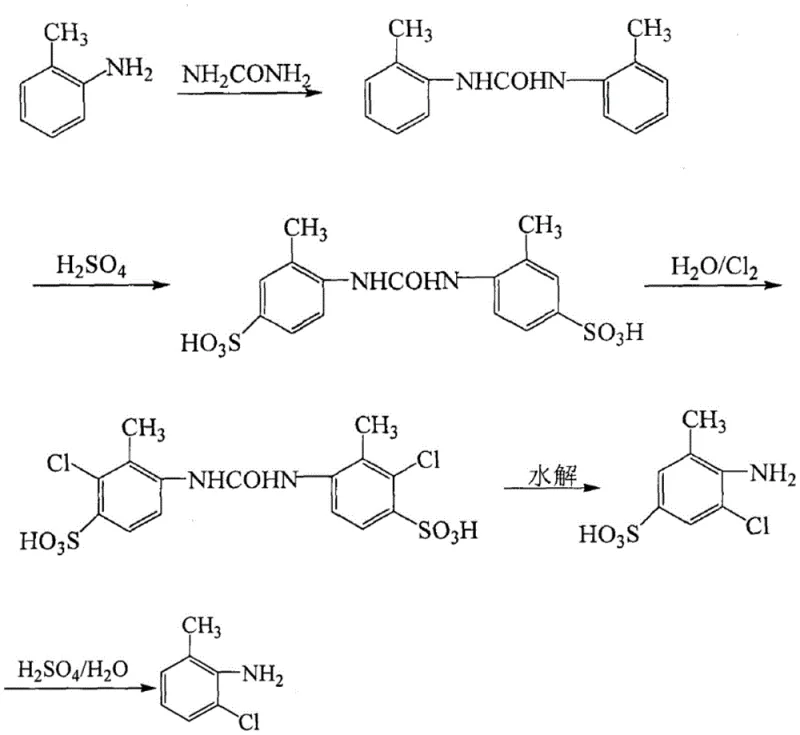
Beyond the safety and environmental liabilities, the conventional sulfonation-based route suffers from operational inefficiencies that directly impact the bottom line. The multi-step nature of the process, involving condensation, sulfonation, chlorination, and multiple hydrolysis steps, inherently leads to material losses at each stage. Patent literature indicates that the overall yield of these traditional methods can be as low as 36%, which is economically unsustainable for high-volume API production. The cumbersome post-treatment procedures, necessitated by the removal of inorganic salts and sulfonated byproducts, further extend production cycles and increase utility consumption. For a reliable pharmaceutical intermediate supplier, relying on such a fragile and polluting process poses a risk to supply continuity and regulatory compliance, especially as global environmental standards become increasingly stringent regarding the discharge of sulfur-containing effluents.
The Novel Approach
In stark contrast to the legacy sulfonation pathways, the method disclosed in patent CN112679364B introduces a streamlined and chemically elegant solution that bypasses the need for sulfonic acid blocking groups entirely. The core innovation lies in the strategic manipulation of the oxidation states of the nitrogen substituents on the aromatic ring. The new route begins with 2-nitro-6-methylaniline, a readily available starting material. The first critical step involves the protection of the existing amino group via acetylation, forming an acetamide derivative. This protection is crucial as it prevents unwanted side reactions during the subsequent reduction phase. Following protection, the nitro group at the ortho-position is selectively reduced to an amino group using catalytic hydrogenation. This sets the stage for a classic Sandmeyer reaction, where the newly formed amino group is converted into a chloro substituent via diazotization and treatment with cuprous chloride. Finally, the acetyl protecting group is removed through mild acidic hydrolysis to reveal the target 2-chloro-6-methylaniline.
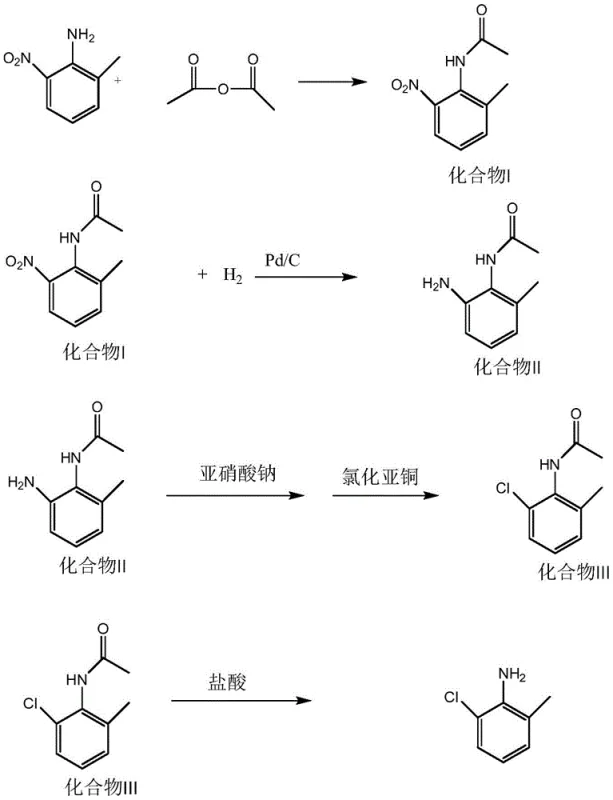
This novel approach offers transformative advantages for cost reduction in API manufacturing. By eliminating the use of chlorosulfonic acid and concentrated sulfuric acid, the process drastically reduces the requirement for exotic alloy reactors and simplifies the engineering controls needed for operator safety. The reaction conditions are milder and easier to control, with the patent specifying temperatures ranging from reflux for acetylation to near-ambient conditions for the diazotization step. Moreover, the solvents employed, such as chloroform and methanol, are common industrial solvents that can be efficiently recovered and recycled, minimizing raw material costs and waste disposal fees. The reported overall yield of this new method exceeds 60%, representing a significant improvement over the 36% yield of the prior art. This increase in efficiency translates directly to higher throughput and lower cost per kilogram, making it an attractive option for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into the Protection-Reduction-Chlorination Strategy
The success of this synthetic route hinges on the precise orchestration of functional group transformations, specifically the interplay between the amino and nitro groups on the toluene scaffold. The initial acetylation step serves a dual purpose: it protects the primary amine from oxidation during the subsequent reduction of the nitro group, and it modulates the electronic properties of the ring to facilitate selective reactivity. By converting the strongly activating amino group into a moderately activating acetamide, the process prevents over-reaction or polymerization that could occur if the free amine were present during the vigorous conditions of the Sandmeyer reaction. The reduction of the nitro group to the amine is typically achieved using a palladium on carbon (Pd/C) catalyst under hydrogen pressure. This heterogeneous catalysis is highly selective, leaving the acetamide and the aromatic ring intact while efficiently converting the nitro functionality. The mechanistic pathway involves the sequential addition of hydrogen atoms to the nitrogen-oxygen bonds, eventually cleaving them to form water and the primary amine.
Following the formation of the diamine intermediate (protected as a mono-acetamide), the introduction of the chlorine atom is achieved through a diazotization-substitution sequence. In the presence of strong acid and sodium nitrite, the primary amine is converted into a highly reactive diazonium salt. This species is unstable and must be generated in situ at low temperatures (typically -5°C to 5°C) to prevent decomposition. The addition of cuprous chloride (CuCl) facilitates the replacement of the diazonium group with a chlorine atom via a radical mechanism characteristic of the Sandmeyer reaction. The copper catalyst mediates the single-electron transfer processes required to break the carbon-nitrogen bond and form the carbon-chlorine bond. Finally, the removal of the acetyl protecting group is accomplished through acid-catalyzed hydrolysis. The protonation of the carbonyl oxygen makes the carbonyl carbon more susceptible to nucleophilic attack by water, leading to the cleavage of the amide bond and the regeneration of the free amine, yielding the final 2-chloro-6-methylaniline product with high purity.
How to Synthesize 2-Chloro-6-Methylaniline Efficiently
The implementation of this patented methodology requires careful attention to reaction parameters to maximize yield and safety. The process is divided into four distinct operational stages, each with specific requirements for reagents, solvents, and temperature control. The initial protection step utilizes acetic anhydride in a non-polar solvent like chloroform, requiring reflux conditions to drive the reaction to completion. The subsequent hydrogenation step demands a pressurized reactor capable of handling hydrogen gas safely, with precise temperature monitoring to prevent runaway exotherms. The diazotization phase is the most critical regarding safety, necessitating strict temperature control to maintain the stability of the diazonium intermediate. Detailed standard operating procedures for each of these stages are essential for technology transfer and successful scale-up.
- Protect the amino group of 2-nitro-6-methylaniline using acetic anhydride in a non-polar solvent like chloroform.
- Reduce the nitro group to an amino group using a hydrogenation catalyst system such as H2/Pd-C in methanol.
- Perform diazotization with sodium nitrite in acid, followed by substitution with cuprous chloride to introduce the chlorine atom.
- Hydrolyze the protecting group under acidic conditions to yield the final 2-chloro-6-methylaniline product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route represents a strategic opportunity to optimize the sourcing of critical oncology intermediates. The shift away from hazardous reagents and complex waste streams directly correlates with reduced operational expenditures and mitigated supply risks. By partnering with manufacturers who utilize this advanced technology, pharmaceutical companies can secure a more stable supply of high-purity intermediates while aligning their supply chains with sustainability goals. The simplicity of the workup procedures also means faster batch turnover times, enhancing the responsiveness of the supply chain to market demands.
- Cost Reduction in Manufacturing: The elimination of chlorosulfonic acid and the associated corrosion-resistant infrastructure significantly lowers capital expenditure (CAPEX) and maintenance costs. Furthermore, the ability to recycle solvents like chloroform and methanol reduces the recurring cost of raw materials. The higher overall yield means less starting material is required to produce the same amount of final product, driving down the variable cost per unit. Qualitative analysis suggests that the simplified purification steps reduce the consumption of energy and auxiliary chemicals, contributing to substantial cost savings in the overall manufacturing budget.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as acetic anhydride, sodium nitrite, and cuprous chloride ensures that the supply chain is not vulnerable to the shortages of specialized reagents often seen with sulfonating agents. The robustness of the catalytic hydrogenation step, using recoverable Pd/C, adds another layer of reliability. Since the process avoids the generation of difficult-to-treat sulfonated waste, there is less risk of production stoppages due to environmental compliance issues or wastewater treatment capacity limits, ensuring consistent delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory benchtop to multi-ton production without the engineering challenges posed by high-temperature sulfuric acid hydrolysis. The reduction in hazardous waste generation aligns with green chemistry principles, making it easier for manufacturing sites to maintain their environmental permits. The use of standard unit operations such as filtration, distillation, and extraction facilitates technology transfer to contract manufacturing organizations (CMOs) globally, expanding the potential supplier base and reducing geopolitical supply risks.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its adoption. The following questions address common inquiries regarding the practical application and benefits of this patented method. These answers are derived directly from the technical disclosures within the patent documentation, providing a factual basis for decision-making.
Q: Why is the new synthetic route for 2-chloro-6-methylaniline considered superior to traditional sulfonation methods?
A: The novel route avoids the use of highly corrosive chlorosulfonic acid and eliminates the need for high-temperature sulfuric acid hydrolysis, significantly reducing safety hazards and equipment corrosion risks while improving overall yield.
Q: What are the key purity specifications achievable with this patented method?
A: Experimental data from the patent indicates that the final product can achieve a purity of approximately 98.6% after standard workup procedures, meeting stringent requirements for pharmaceutical intermediate applications.
Q: How does this process impact environmental compliance in large-scale manufacturing?
A: By utilizing recyclable solvents like chloroform and methanol and avoiding the generation of heavy sulfonated waste streams, the process offers a greener alternative that simplifies wastewater treatment and aligns with modern environmental regulations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-6-Methylaniline Supplier
As the global demand for targeted cancer therapies continues to rise, the need for efficient and sustainable production of key intermediates like 2-chloro-6-methylaniline has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this industry shift, leveraging advanced synthetic methodologies to deliver superior value to our partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of major pharmaceutical companies without compromising on quality. We operate with stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of intermediate meets the exacting standards required for GMP API synthesis.
We invite procurement leaders and R&D teams to engage with us to explore how this optimized synthetic route can benefit your specific projects. By collaborating with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your volume needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of technical excellence and commercial reliability.
