Advanced Side-Fluorine Biphenyl Diluents: Enabling Fast-Response High Birefringence Liquid Crystals
Introduction to Next-Generation Liquid Crystal Diluents
The rapid evolution of Augmented Reality (AR) and high-performance display technologies demands liquid crystal materials that can switch states in less than one millisecond to prevent color distortion and ensure seamless user experiences. Patent CN107721811B addresses this critical industry bottleneck by disclosing a novel class of side-fluorine-containing biphenyl diluents specifically engineered for high-birefringence liquid crystal systems. Unlike traditional diluents that often compromise optical performance for viscosity, this invention achieves a rare balance: it significantly lowers rotational viscosity while preserving a high birefringence index greater than 0.25. The core innovation lies in the molecular architecture, which integrates a lateral fluorine substituent and a terminal butenyl group on a biphenyl scaffold. This structural design effectively disrupts molecular packing to enhance low-temperature fluidity without sacrificing the conjugated system necessary for high optical anisotropy.
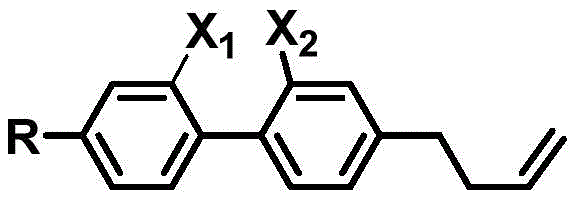
For R&D directors and procurement specialists in the electronic chemicals sector, this patent represents a pivotal shift away from legacy cyclohexane-based diluents like 3HHV, which typically exhibit birefringence values as low as 0.02. The disclosed compounds demonstrate birefringence indices 6 to 6.5 times higher than these commercial standards, enabling the formulation of mixed crystal systems that meet the rigorous speed requirements of modern optoelectronic devices. By leveraging industrially mature synthetic methodologies, specifically Suzuki coupling and Wittig olefination, the patent outlines a pathway that is not only chemically elegant but also commercially viable for mass production. This report analyzes the technical merits and supply chain implications of adopting this advanced diluent technology for next-generation display manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the formulation of high-birefringence liquid crystals has been plagued by a fundamental trade-off between optical performance and physical fluidity. Conventional diluents frequently rely on non-conjugated cyclohexane structures to manage viscosity, but this comes at the severe expense of birefringence, often dragging the overall mixture performance below the threshold required for fast-response applications. Furthermore, the synthesis of cyclohexane-based intermediates often involves complex hydrogenation steps that introduce cis-trans isomerization challenges, complicating purification and reducing overall yield. These structural limitations mean that formulators must add larger quantities of high-viscosity monomers to compensate for the low birefringence of the diluent, inadvertently increasing the rotational viscosity of the final mixture and slowing down response times. This creates a vicious cycle where improving one parameter degrades another, hindering the development of sub-millisecond switching displays.
The Novel Approach
The novel approach detailed in the patent circumvents these issues by utilizing a fully conjugated biphenyl core substituted with a lateral fluorine atom. This design strategy capitalizes on the high polarizability of the aromatic system to maintain exceptional birefringence while the fluorine atom acts as a steric disruptor to lower melting points and viscosity. The synthetic route bypasses the need for difficult ring hydrogenations entirely, opting instead for a convergent assembly of aromatic building blocks. By incorporating a terminal butenyl chain, the molecule gains additional conformational flexibility, further enhancing its ability to function as a low-viscosity solvent for high-performance liquid crystal monomers. This dual-functional design ensures that the diluent actively contributes to the optical goals of the mixture rather than merely acting as an inert spacer, representing a significant leap forward in material science for the display industry.
Mechanistic Insights into Suzuki-Wittig Cascade Synthesis
The synthetic elegance of this process lies in its reliance on two of the most robust carbon-carbon bond-forming reactions in organic chemistry: the Suzuki-Miyaura cross-coupling and the Wittig olefination. The sequence begins with the coupling of a phenylboronic acid derivative with a fluorinated bromo-acetal precursor. This step is catalyzed by tetrakis(triphenylphosphine)palladium in a biphasic system of DMF and water, facilitated by potassium carbonate as a base. The presence of the acetal protecting group is crucial here, as it masks the reactive aldehyde functionality during the harsh conditions of the coupling reaction, preventing unwanted side reactions or polymerization. The lateral fluorine atom on the aromatic ring exerts an electron-withdrawing effect that can influence the oxidative addition step of the catalytic cycle, yet the optimized conditions ensure high conversion rates, yielding the biphenyl acetal intermediate with excellent purity.
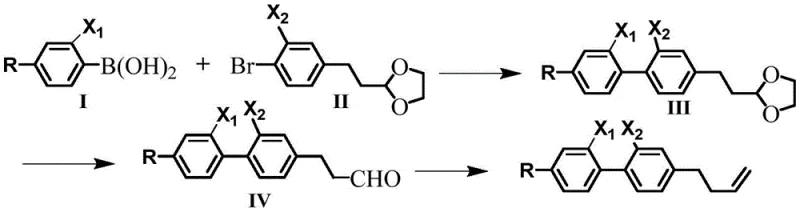
Following the construction of the biphenyl skeleton, the acetal group is hydrolyzed under mild acidic conditions using formic acid to reveal the aldehyde moiety. This intermediate then undergoes a Wittig reaction with bromomethyl triphenylphosphine salt in the presence of potassium tert-butoxide. This final transformation installs the critical terminal double bond, completing the butenyl side chain. The choice of reagents and the low-temperature initiation (-10°C) of the Wittig step are vital for controlling the E/Z selectivity and minimizing impurities. From a quality control perspective, this linear sequence avoids the generation of difficult-to-remove metal residues often associated with more exotic catalytic methods, simplifying the downstream purification process. The result is a highly pure diluent that meets the stringent electrical and optical specifications required for premium liquid crystal applications, with the process inherently designed to minimize hazardous waste generation.
How to Synthesize Side-Fluorine Biphenyl Diluent Efficiently
The synthesis of these high-value electronic chemicals follows a streamlined three-step protocol that balances reaction efficiency with operational safety. The process initiates with the palladium-catalyzed coupling of specific boronic acids and brominated acetals, followed by a straightforward deprotection and a final olefination step. This modular approach allows for the easy variation of the alkyl chain (R group) to fine-tune the physical properties of the final diluent for specific customer formulations. The detailed standardized synthesis steps, including precise molar ratios, temperature profiles, and workup procedures, are outlined in the guide below to ensure reproducibility and scalability.
- Perform Suzuki coupling between a phenylboronic acid derivative and a fluorinated bromo-acetal using Pd(PPh3)4 catalyst in DMF/Water at 70-90°C.
- Hydrolyze the resulting acetal intermediate using formic acid in tetrahydrofuran at 50-70°C to generate the aldehyde precursor.
- Execute a Wittig reaction with bromomethyl triphenylphosphine salt and potassium tert-butoxide at -10°C to room temperature to install the terminal butenyl group.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers tangible strategic benefits rooted in chemical efficiency and raw material availability. The reliance on Suzuki coupling means that the starting materials—phenylboronic acids and brominated aromatics—are commodity chemicals available from a wide global network of suppliers, mitigating the risk of single-source bottlenecks. Furthermore, the elimination of complex cyclohexane synthesis steps, such as high-pressure hydrogenation and isomer separation, drastically simplifies the manufacturing workflow. This simplification translates directly into reduced processing time and lower energy consumption per kilogram of product, providing a clear pathway for cost optimization without compromising on the high purity standards demanded by the electronics sector.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive and specialized catalysts often required for asymmetric hydrogenation or complex ring-closing metathesis found in alternative routes. By utilizing standard palladium catalysts and common phosphonium salts, the direct material costs are significantly lowered. Additionally, the high yields reported in the patent examples suggest that raw material utilization is highly efficient, minimizing waste disposal costs. The avoidance of cis-trans isomerization issues inherent in cyclohexane chemistry further reduces the burden on purification resources, allowing for a leaner, more cost-effective production model that enhances overall margin potential.
- Enhanced Supply Chain Reliability: The synthetic pathway relies on robust, well-understood chemistry that is easily transferable between different manufacturing sites. This flexibility ensures that supply continuity can be maintained even if one production facility faces disruptions. The starting materials are stable and have long shelf lives, allowing for strategic stockpiling to buffer against market volatility. Moreover, the reaction conditions are relatively mild, reducing the wear and tear on reactor equipment and extending asset life, which contributes to a more reliable and predictable production schedule for long-term supply agreements.
- Scalability and Environmental Compliance: The use of aqueous workups and standard organic solvents like ethyl acetate and heptane aligns well with modern environmental, health, and safety (EHS) guidelines. The process generates fewer hazardous byproducts compared to routes involving heavy metal hydrogenation or aggressive halogenation. This environmental compatibility facilitates easier regulatory approval and permitting for scale-up activities. The linear nature of the synthesis allows for straightforward telescoping of steps or continuous flow processing opportunities, making the transition from pilot plant to multi-ton commercial production seamless and compliant with increasingly strict global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this side-fluorine biphenyl diluent technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the fit of this material within your specific liquid crystal formulation strategy.
Q: How does the side-fluorine substitution improve liquid crystal performance?
A: The lateral fluorine atom introduces a strong dipole moment and alters the molecular aspect ratio, which significantly reduces rotational viscosity while maintaining high birefringence, crucial for millisecond response times in AR displays.
Q: What are the key advantages of this synthesis route over cyclohexane-based diluents?
A: Unlike cyclohexane-based diluents that suffer from cis-trans isomerization issues and lower birefringence, this biphenyl route avoids complex hydrogenation steps, offers higher optical anisotropy (6-6.5 times higher than 3HHV), and utilizes mature, scalable coupling chemistry.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process relies on industrially mature Suzuki and Wittig reactions with readily available starting materials like phenylboronic acids and brominated acetals, ensuring robust supply chain continuity and ease of scale-up from kilograms to tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Side-Fluorine Biphenyl Diluent Supplier
As the demand for high-speed, high-resolution displays continues to surge, the need for specialized intermediates like side-fluorine biphenyl diluents becomes increasingly critical. NINGBO INNO PHARMCHEM stands ready to support your R&D and production needs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the precise requirements of Suzuki and Wittig chemistries, ensuring that every batch meets stringent purity specifications through our rigorous QC labs. We understand that consistency is key in the electronic materials sector, and our dedicated process engineering team works tirelessly to optimize yields and minimize impurities for every project we undertake.
We invite you to collaborate with us to unlock the full potential of this advanced diluent technology for your next-generation liquid crystal formulations. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and supply chain constraints. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample availability. Let us partner with you to drive innovation and efficiency in the rapidly evolving landscape of optoelectronic materials.
