Advanced Synthesis of Side Fluorine Tolane Diluents for Next-Gen High Birefringence Liquid Crystals
The rapid evolution of Augmented Reality (AR) display systems demands liquid crystal materials that simultaneously possess high optical efficiency and ultra-fast response times, typically less than 1 millisecond. A critical challenge in this field is the inherent contradiction between achieving high birefringence and maintaining low rotational viscosity. Patent CN107628932B addresses this bottleneck by disclosing a novel class of side fluorine-containing diphenylacetylene diluents. These compounds are engineered to reduce the rotational viscosity of high-birefringence liquid crystal mixtures without compromising their optical anisotropy. Unlike traditional diluents that often rely on non-conjugated cyclohexane structures which drastically lower birefringence, this invention leverages a conjugated tolane core with specific lateral fluorine and terminal alkene substituents. This structural innovation allows for a significant enhancement in birefringence, reported to be 13 to 15 times higher than commercial benchmarks like 3HHV, while ensuring the mixture remains fluid at low temperatures.
The development of reliable electronic chemical supplier networks depends heavily on the adoption of synthetic routes that avoid legacy inefficiencies. Conventional methods for producing low-viscosity diluents frequently incorporate cyclohexane rings to manage melting points. However, these saturated structures disrupt the conjugated pi-system essential for high birefringence, forcing formulators to accept a severe penalty in optical performance. Furthermore, the synthesis of cyclohexane-based intermediates often necessitates energy-intensive hydrogenation and rigorous cis-trans isomer separation processes. These additional unit operations not only inflate manufacturing costs but also introduce complexity in impurity control, potentially affecting the voltage holding ratio and reliability of the final display panel. The reliance on such structurally compromised diluents limits the ability of R&D teams to push the boundaries of response speed in next-generation micro-displays.
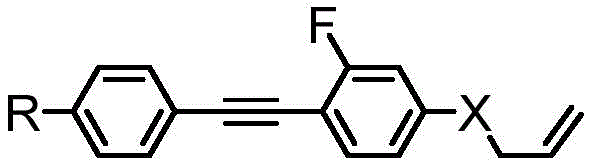
The novel approach detailed in the patent circumvents these limitations by utilizing a fully conjugated diphenylacetylene scaffold functionalized with a lateral fluorine atom. This design strategy capitalizes on the high polarizability of the triple bond and the aromatic rings to maximize birefringence, while the fluorine atom and terminal alkene chain serve to depress the melting point and rotational viscosity. The synthesis relies on robust carbon-carbon bond-forming reactions, specifically the Sonogashira coupling, which is well-established in the fine chemical industry. By replacing the bulky, non-conjugated cyclohexane moiety with a planar, fluorinated phenyl ring, the new diluent maintains the molecular linearity required for effective mesogen alignment. This results in a material that acts as a true high-birefringence dopant rather than a mere viscosity reducer, enabling the formulation of liquid crystals with delta-n values exceeding 0.25.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional strategies for lowering the viscosity of high-birefringence liquid crystals have predominantly focused on blending in non-conjugated cyclic compounds, such as cyclohexyl-benzenes or bicyclohexyls. While these additives successfully lower the melting point and viscosity due to their flexible ring structures, they act as optical dead weight. The lack of pi-conjugation in the cyclohexane ring significantly dilutes the overall birefringence of the mixture, requiring higher concentrations of expensive high-delta-n monomers to compensate. Moreover, the industrial preparation of these cyclohexane derivatives often involves the catalytic hydrogenation of aromatic precursors, a step that requires high-pressure equipment and precious metal catalysts like rhodium or ruthenium. The subsequent need to separate cis and trans isomers adds further downstream processing burden, increasing solvent consumption and waste generation. These factors collectively drive up the cost of goods sold and extend the lead time for high-purity electronic chemicals, creating a supply chain bottleneck for mass-market AR device production.
The Novel Approach
In stark contrast, the methodology presented in CN107628932B employs a convergent synthesis centered around the Sonogashira cross-coupling reaction to construct the rigid tolane core. This approach allows for the precise installation of a fluorine atom at the ortho-position relative to the ether or alkyl linkage, a modification proven to disrupt molecular packing and lower the clearing point without sacrificing optical anisotropy. The terminal functionalization is achieved through either a Williamson ether synthesis followed by allylation or a Wittig olefination, both of which are highly selective and scalable. By avoiding the saturation of aromatic rings, the process preserves the extended conjugation length, which is the fundamental driver of the material's high birefringence properties. This shift from saturated to unsaturated diluents represents a paradigm change in liquid crystal formulation, offering a pathway to achieve millisecond-level response times without the traditional trade-off in optical performance.
Mechanistic Insights into Sonogashira Coupling and Functionalization
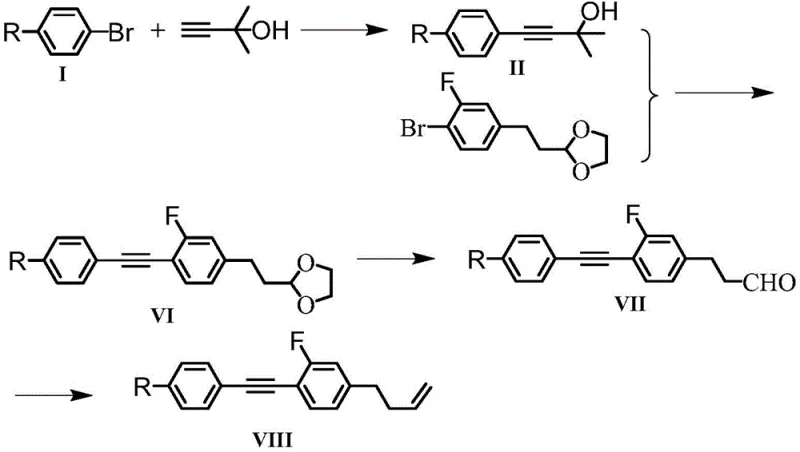
The core of this synthetic strategy lies in the palladium-catalyzed Sonogashira coupling, a reaction that efficiently joins terminal alkynes with aryl halides. In the first stage, para-alkyl bromobenzene reacts with 2-methyl-3-butyn-2-ol in the presence of tetrakis(triphenylphosphine)palladium(0) and cuprous iodide. The mechanism proceeds through the oxidative addition of the aryl bromide to the Pd(0) species, followed by transmetallation with the copper-acetylide complex formed in situ. This step is critical for establishing the diphenylacetylene backbone. The use of triethylamine as a base and solvent facilitates the deprotonation of the alkyne and stabilizes the catalytic cycle. The subsequent coupling of this intermediate with a fluorinated aryl bromide, such as 4-bromo-2-fluorophenol derivative, extends the conjugation further. The presence of the fluorine substituent on the coupling partner requires careful tuning of the ligand environment to ensure high turnover numbers, as electron-deficient aryl halides can sometimes slow down the oxidative addition step.
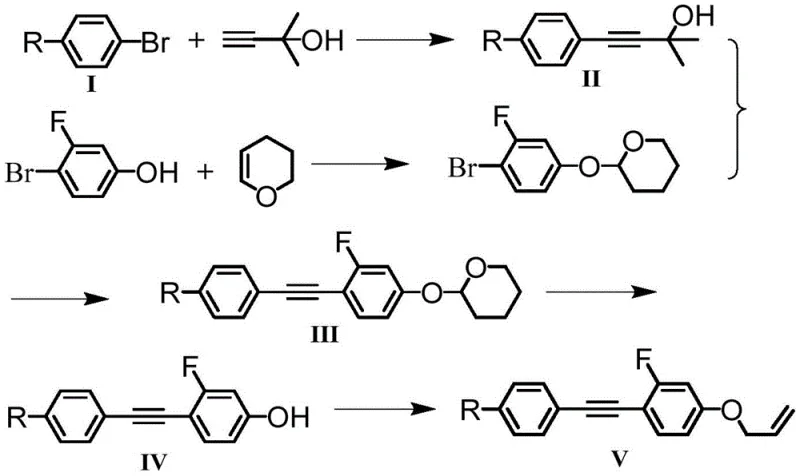
Following the construction of the carbon skeleton, the final steps involve installing the terminal alkene tail, which is crucial for modulating the viscosity. For the ether-linked variants (where X is O), the phenolic hydroxyl group generated after deprotection is alkylated with 3-bromopropylene under basic conditions. This nucleophilic substitution introduces the allyl group, adding conformational freedom to the molecular terminus. Alternatively, for the alkyl-linked variants (where X is CH2), the synthesis proceeds via a Wittig reaction. An aldehyde intermediate, protected as an acetal during the coupling steps to prevent side reactions, is deprotected and then reacted with a phosphonium ylide. This olefination step creates the terminal double bond with high stereoselectivity. The entire sequence is designed to minimize protecting group manipulations; for instance, the use of tetrahydropyranyl (THP) ethers allows for mild deprotection conditions that do not compromise the sensitive alkyne linkage. This mechanistic elegance ensures high purity and yield, which are paramount for electronic grade materials.
How to Synthesize Side Fluorine Tolane Diluent Efficiently
The synthesis of these advanced liquid crystal diluents requires precise control over reaction parameters to ensure the removal of trace metal catalysts and halogenated impurities. The process begins with the rigorous exclusion of oxygen and moisture, typically under a nitrogen atmosphere, to prevent the homocoupling of the alkyne intermediates (Glaser coupling), which is a common side reaction in Sonogashira protocols. Detailed standardized operating procedures for the preparation of the key intermediates and the final purification steps are essential for reproducibility. The following guide outlines the critical operational phases derived from the patent examples, focusing on the optimization of molar ratios and temperature profiles to maximize yield and purity.
- Perform Sonogashira coupling between para-alkyl bromobenzene and 2-methyl-3-butyn-2-ol using Pd/Cu catalysts.
- Couple the resulting alkyne intermediate with a fluorinated phenol or benzaldehyde derivative via a second Sonogashira reaction.
- Finalize the structure through deprotection and allylation or Wittig olefination to introduce the terminal alkene group.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the transition to this side fluorine tolane architecture offers substantial strategic benefits regarding raw material security and cost structure. The starting materials, such as para-alkyl bromobenzenes and fluorinated phenols, are commodity chemicals produced on a massive scale for the agrochemical and pharmaceutical industries. This abundance ensures a stable supply chain with minimal risk of shortage-driven price volatility. Furthermore, the elimination of the cyclohexane hydrogenation step removes the need for high-pressure reactors and specialized heterogeneous catalysts, significantly reducing capital expenditure requirements for manufacturing facilities. The simplified workflow, which avoids the difficult separation of cis-trans isomers, translates directly into higher throughput and reduced cycle times. These efficiencies allow for cost reduction in electronic chemical manufacturing, making high-performance AR display components more economically viable for consumer electronics markets.
- Cost Reduction in Manufacturing: The synthetic route eliminates the need for expensive high-pressure hydrogenation equipment and precious metal catalysts typically required for saturating aromatic rings. By relying on solution-phase palladium catalysis with recoverable ligands, the process reduces both energy consumption and catalyst loading costs. Additionally, the avoidance of isomer separation steps simplifies the purification train, leading to significant savings in solvent usage and waste disposal fees. The overall atom economy is improved by focusing on direct coupling reactions rather than multi-step functional group interconversions.
- Enhanced Supply Chain Reliability: The reliance on widely available petrochemical derivatives like bromobenzene and allyl bromide ensures that the supply chain is not dependent on niche intermediates with long lead times. The robustness of the Sonogashira reaction means that production can be easily scaled across multiple contract manufacturing organizations without extensive requalification. This flexibility mitigates the risk of single-source bottlenecks and ensures consistent delivery schedules for downstream liquid crystal blenders. The stability of the intermediates also allows for strategic stockpiling, further buffering against market fluctuations.
- Scalability and Environmental Compliance: The process operates under relatively mild conditions compared to high-pressure hydrogenation, reducing the safety footprint of the manufacturing plant. The use of standard organic solvents like toluene and THF, which can be efficiently recycled, aligns with modern green chemistry principles. The absence of heavy metal residues from hydrogenation catalysts simplifies the wastewater treatment process, ensuring compliance with stringent environmental regulations. This streamlined approach facilitates the commercial scale-up of complex display materials from pilot batches to multi-ton annual production capacities with minimal environmental impact.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this new diluent technology in liquid crystal formulations. The answers are derived from the specific experimental data and beneficial effects reported in the patent documentation, providing clarity on performance metrics and synthesis feasibility. Understanding these details is crucial for R&D teams evaluating the material for next-generation display projects.
Q: How does the side fluorine substituent affect liquid crystal performance?
A: The lateral fluorine substituent effectively improves the low-temperature performance of the liquid crystal mixture while maintaining high birefringence, addressing the trade-off between viscosity and optical properties.
Q: What is the birefringence advantage of this tolane diluent over commercial 3HHV?
A: According to patent data, the synthesized diluent exhibits a birefringence value 13 to 15 times higher than the commercial 3HHV diluent, enabling faster response times in AR displays.
Q: Is the synthesis scalable for industrial production?
A: Yes, the process utilizes industrially mature Sonogashira coupling and Wittig reactions with readily available starting materials, avoiding complex cis-trans isomerization steps associated with cyclohexane derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Side Fluorine Tolane Diluent Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis for advanced electronic materials, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing palladium-catalyzed coupling reactions to meet the stringent purity specifications required for liquid crystal applications, ensuring that trace metal levels are kept below ppb thresholds. With our rigorous QC labs and state-of-the-art purification capabilities, we guarantee the high optical quality and batch-to-batch consistency necessary for high-birefringence display formulations. We understand the critical nature of supply continuity in the fast-paced consumer electronics sector and are committed to being a long-term strategic partner.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your product roadmap. By leveraging our expertise in fluoro-chemistry and conjugated systems, we can help you accelerate the development of faster, more efficient AR displays while optimizing your total cost of ownership.
