Advanced Metal-Free Synthesis of Aryne-Substituted Nitriles for Commercial Scale-Up
The landscape of organic synthesis for complex nitrile derivatives is undergoing a significant transformation, driven by the urgent need for cleaner, more cost-effective manufacturing routes in the pharmaceutical sector. Patent CN112159336B, titled "Preparation method of high-purity aryne substituted nitrile compound," introduces a groundbreaking methodology that addresses the longstanding challenges associated with traditional alkyne functionalization. This innovation centers on a robust nucleophilic substitution strategy that bypasses the need for expensive transition metal catalysts, thereby offering a streamlined pathway to high-value intermediates. For R&D directors and procurement specialists alike, this patent represents a pivotal shift away from legacy technologies that are plagued by high operational costs and stringent purification requirements. By leveraging a specific sequence of base activation and controlled temperature addition, the disclosed method achieves exceptional purity levels, often exceeding 99%, which is critical for downstream drug substance manufacturing. The technical implications of this disclosure extend far beyond the laboratory, promising substantial improvements in supply chain reliability and overall process economics for the global production of fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryne-substituted nitriles has relied heavily on methodologies that introduce significant complexity and cost into the manufacturing workflow. As illustrated in earlier literature, such as the 2016 reported method, the construction of the carbon-carbon triple bond linkage frequently necessitates the use of palladium and copper co-catalysts in cross-coupling reactions.
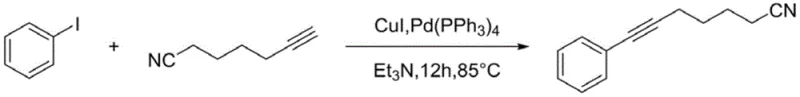
While chemically effective, these transition metal-catalyzed routes present severe drawbacks for industrial scale-up, primarily concerning the removal of trace metal impurities. In the context of Good Manufacturing Practice (GMP) for pharmaceuticals, residual palladium levels must be reduced to parts-per-million (ppm) or even parts-per-billion (ppb) levels, requiring additional processing steps such as silica gel treatment or specialized metal scavengers. Furthermore, earlier approaches dating back to 2002 utilized harsh conditions and difficult-to-source raw materials, resulting in prolonged reaction times and poor atom economy. These legacy processes create bottlenecks in production capacity and inflate the Cost of Goods Sold (COGS) due to the high price of noble metals and the labor-intensive purification protocols required to meet regulatory standards.
The Novel Approach
In stark contrast to the metal-dependent strategies of the past, the novel approach detailed in patent CN112159336B utilizes a direct nucleophilic substitution mechanism mediated by strong inorganic bases. This method fundamentally alters the reaction paradigm by generating a reactive acetylide anion in situ, which then attacks a haloalkane nitrile electrophile. The elimination of precious metal catalysts not only drastically reduces raw material costs but also simplifies the downstream workup procedure, as there is no need for complex metal removal steps. The process operates under relatively mild thermal conditions, utilizing common solvents like DMSO and THF, which are readily available in bulk quantities globally. This shift towards a base-mediated protocol ensures that the final product is free from heavy metal contamination from the outset, significantly enhancing the purity profile of the resulting aryne-substituted nitrile. Consequently, this approach offers a more sustainable and economically viable route for the commercial manufacture of these critical building blocks.
Mechanistic Insights into Base-Mediated Nucleophilic Substitution
The core chemical innovation of this patent lies in the precise control of the deprotonation and subsequent alkylation steps to maximize yield and minimize byproduct formation. The reaction initiates with the activation of a strong base, such as sodium hydride (NaH) or potassium hydride (KH), in a polar aprotic solvent like DMSO. Heating this mixture to 50-60°C is a critical preparatory step that ensures the effective formation of the active basic species, which is essential for the subsequent deprotonation of the terminal alkyne. Once the base is activated, the reaction mixture is cooled to low temperatures, typically around -10°C, before the introduction of the arylacetylene substrate. This temperature gradient is vital for managing the exothermic nature of the acetylide formation and preventing thermal degradation or polymerization of the sensitive alkyne species.
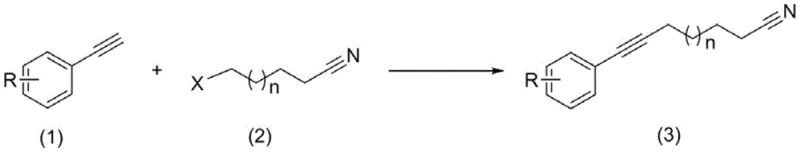
Following the generation of the nucleophilic acetylide anion, the haloalkane nitrile is introduced slowly while maintaining the low temperature regime between -10°C and -5°C. This controlled addition facilitates a clean SN2 substitution reaction where the acetylide anion displaces the halide leaving group on the alkyl chain. The patent emphasizes that maintaining this specific thermal window is crucial for suppressing side reactions, such as elimination or over-alkylation, which could compromise the purity of the final product. The reaction is allowed to proceed for 4 to 6 hours, ensuring complete conversion of the starting materials as monitored by GC analysis. The resulting crude mixture is then subjected to a straightforward aqueous workup and column chromatography, yielding the target aryne-substituted nitrile with high structural integrity and minimal impurity load, demonstrating the robustness of this mechanistic pathway.
How to Synthesize Aryne-Substituted Nitrile Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for reproducing these high-purity results in a pilot or production setting. The process begins with the careful preparation of the base-solvent system under an inert nitrogen atmosphere to exclude moisture and oxygen, which could quench the reactive intermediates. Operators must strictly adhere to the temperature profiles specified, particularly the initial heating phase for base activation followed by the rapid cooling prior to substrate addition. The sequential addition of reagents—first the arylacetylene to form the anion, and subsequently the halo-nitrile electrophile—is designed to maintain a steady concentration of the nucleophile, thereby optimizing the reaction kinetics. Detailed standardized operating procedures regarding stoichiometry, specifically a molar ratio of approximately 1:1:2 for alkyne, halide, and base, are essential for achieving the reported yields. For a comprehensive breakdown of the specific operational parameters and safety considerations, please refer to the technical guide below.
- Activate the base by heating sodium hydride (NaH) or potassium hydride (KH) with a polar aprotic solvent like DMSO under nitrogen protection at 50-60°C.
- Cool the mixture to -10°C and slowly add the terminal arylacetylene compound to generate the reactive acetylide anion intermediate.
- Maintain low temperature (-10 to -5°C) while adding the haloalkane nitrile electrophile, stirring for 4-6 hours to complete the substitution.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this metal-free synthesis route offers compelling advantages that directly impact the bottom line and supply chain resilience. The most significant benefit is the complete elimination of palladium and copper catalysts, which are subject to volatile market pricing and supply constraints. By removing these expensive inputs from the bill of materials, manufacturers can achieve a substantial reduction in raw material costs without compromising on quality. Furthermore, the simplified purification process reduces the consumption of auxiliary materials such as metal scavengers and specialized filtration media, leading to lower operational expenditures. This efficiency gain translates into a more competitive pricing structure for the final intermediate, allowing downstream partners to optimize their own manufacturing budgets while securing a reliable source of high-quality material.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the removal of noble metal catalysts and the associated purification burdens. Traditional cross-coupling methods require significant investment in metal scavengers and extensive analytical testing to verify residual metal levels, both of which add time and cost to the production cycle. By utilizing inexpensive inorganic bases like sodium hydride instead, the process drastically lowers the input cost per kilogram. Additionally, the higher selectivity of the reaction reduces the formation of difficult-to-separate byproducts, minimizing yield losses during purification. This streamlined workflow ensures that resources are focused on value-added production rather than waste management and remediation, resulting in a leaner and more cost-effective manufacturing operation.
- Enhanced Supply Chain Reliability: Supply chain stability is significantly improved by relying on commodity chemicals rather than specialized catalytic systems. Sodium hydride, DMSO, and THF are produced at massive scales globally, ensuring consistent availability and shielding the production schedule from the supply shocks often associated with rare earth or precious metal markets. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further reducing the risk of batch failures. For supply chain managers, this translates to predictable lead times and the ability to scale production volumes rapidly in response to market demand without being bottlenecked by the sourcing of exotic reagents.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is markedly lower than that of metal-catalyzed alternatives, aligning with modern green chemistry principles and regulatory expectations. The absence of heavy metals simplifies waste stream management, as the effluent does not require specialized treatment for metal recovery or disposal. This ease of waste handling facilitates smoother regulatory approvals and reduces the environmental compliance costs associated with hazardous waste disposal. Moreover, the reaction's compatibility with standard industrial reactors and its exothermic profile, which is manageable through standard cooling protocols, makes it highly scalable from kilogram to multi-ton production. This scalability ensures that the technology can grow with the commercial needs of the drug development pipeline, from early clinical trials to full-scale commercial launch.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical application of the method. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers highlight the specific operational parameters that differentiate this method from conventional approaches, ensuring that stakeholders have a accurate understanding of the process capabilities and limitations.
Q: Why is the metal-free approach superior for pharmaceutical intermediates?
A: Traditional methods often utilize palladium or copper catalysts which leave toxic metal residues. Removing these to meet ICH Q3D guidelines requires expensive scavengers and complex purification. The patented base-mediated method eliminates heavy metals entirely, simplifying compliance and reducing purification costs.
Q: What represents the critical control point in this synthesis?
A: Temperature control during the addition of the arylacetylene and the haloalkane nitrile is critical. The patent specifies cooling to -10°C to manage the exothermic formation of the acetylide anion and prevent side reactions such as polymerization or over-alkylation, ensuring high purity (>99%).
Q: Can this process be scaled for industrial production?
A: Yes, the process uses common industrial solvents like DMSO and THF and avoids sensitive transition metal catalysts. The reaction conditions (ambient to mild heating, standard inert gas protection) are highly amenable to large-scale batch reactors, ensuring supply chain continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryne-Substituted Nitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain a competitive edge in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the potential of patent CN112159336B and is fully equipped to translate this laboratory-scale innovation into robust commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are designed to handle reactive chemistries safely, with rigorous QC labs dedicated to maintaining stringent purity specifications that exceed industry standards. By partnering with us, you gain access to a supply chain that is not only cost-effective but also technically superior, guaranteeing the consistent delivery of high-performance intermediates for your drug development programs.
We invite you to engage with our technical procurement team to discuss how this metal-free synthesis route can be tailored to your specific project requirements. Whether you are looking to reduce the cost of goods for an existing API or secure a new source for a complex intermediate, we are prepared to provide a Customized Cost-Saving Analysis that quantifies the potential benefits for your organization. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to technical excellence can drive value for your business.
