Optimizing Cephalosporin Intermediate Synthesis via Novel Anhydrous Aminothiazole Preparation
The pharmaceutical industry continuously seeks robust methodologies to enhance the efficiency of beta-lactam antibiotic synthesis, particularly for cephalosporin derivatives where intermediate purity dictates final drug efficacy. Patent CN1232512C introduces a transformative industrial process for the preparation of anhydrous 2-(2-aminothiazole-4-yl)-2-hydroxy compounds, addressing a longstanding bottleneck in the production of key antibiotic intermediates. Traditionally, these aminothiazole derivatives exist as hydrates containing one or two molecules of crystallization water, which poses significant challenges during subsequent acylation steps. The presence of this crystal water necessitates the use of excessive halogenating agents to compensate for water consumption, leading to increased operational costs and complex waste management issues. This novel method provides a streamlined pathway to convert these hydrates into high-purity anhydrous forms through a solvent-mediated dehydration process, ensuring that downstream reactions proceed with optimal stoichiometry and minimal side reactions. By implementing this technology, manufacturers can achieve a more sustainable and cost-effective production line for critical pharmaceutical building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
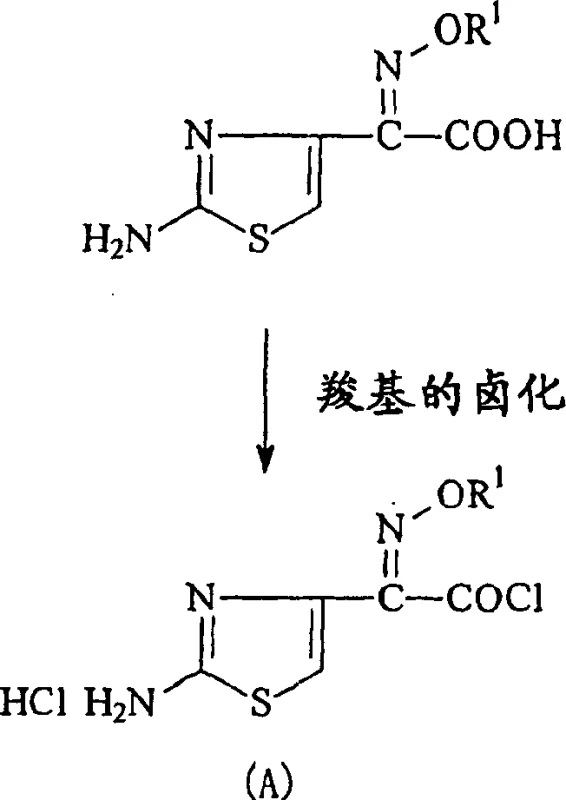
In conventional synthesis routes for cephalosporin side chains, the starting aminothiazole derivatives are typically isolated as hydrates, which inherently contain structural water molecules within their crystal lattice. When these hydrated compounds are directly subjected to chlorination using agents like phosphorus pentachloride to form acid chlorides, the crystallization water reacts competitively with the halogenating agent. This parasitic reaction consumes a significant portion of the reagent, historically requiring up to 3 equivalents of halogenating agent to ensure complete conversion of the carboxylic acid group. This excess usage not only inflates raw material costs substantially but also generates large volumes of hazardous acidic waste and phosphorus-containing byproducts that require rigorous treatment. Furthermore, the presence of water can lead to hydrolysis of the formed acid chloride, reducing overall yield and complicating the purification of the final intermediate. The environmental load associated with neutralizing and disposing of this excess reagent creates a significant compliance burden for large-scale manufacturing facilities aiming to meet modern green chemistry standards.
The Novel Approach
The innovative process described in patent CN1232512C circumvents these inefficiencies by introducing a dedicated dehydration step prior to halogenation, fundamentally altering the stoichiometry of the reaction. By suspending the hydrated crystals in specific ketone solvents such as acetone or methyl ethyl ketone and heating to reflux, the crystal water is effectively removed without degrading the sensitive aminothiazole structure. This pre-treatment yields an anhydrous intermediate with moisture content reduced to negligible levels, typically below 0.5 percent. Consequently, the subsequent reaction with halogenating agents requires only a near-stoichiometric amount, approximately 1 to 1.2 equivalents, to achieve complete conversion to the acid chloride. This drastic reduction in reagent consumption translates directly into lower material costs and a significantly simplified waste stream. The process ensures that the valuable halogenating agent is utilized exclusively for the intended transformation of the carboxyl group, maximizing atom economy and process safety.
Mechanistic Insights into Solvent-Mediated Dehydration
The core mechanism of this technology relies on the differential solubility and thermodynamic stability of the hydrate versus the anhydrous form in ketone solvents. When the hydrated 2-(2-aminothiazole-4-yl)-2-hydroxy compound is suspended in a solvent like acetone at elevated temperatures, the solvent molecules interact with the crystal surface, facilitating the release of water of crystallization into the bulk liquid phase. The reflux conditions provide the necessary thermal energy to break the hydrogen bonding network holding the water within the crystal lattice, while the solvent's affinity for water helps to drive the equilibrium towards the anhydrous state. Upon cooling, the anhydrous compound precipitates out of the solution as a stable crystal form, distinct from the original hydrate, as confirmed by powder X-ray diffraction patterns which show unique peaks at specific 2-theta angles. This phase transition is critical because it locks the compound in a water-free state that is kinetically stable under the drying conditions used, preventing re-absorption of moisture from the atmosphere before the next reaction step.
From an impurity control perspective, this dehydration mechanism is vital for maintaining the integrity of the aminothiazole ring and the oxime functionality. Water presence during the chlorination step can lead to the hydrolysis of the activated acid chloride intermediate back to the carboxylic acid, creating a cycle of reagent consumption and yield loss. By ensuring the starting material is anhydrous, the reaction environment remains strictly non-aqueous, preventing hydrolytic degradation and minimizing the formation of difficult-to-remove impurities. This high level of purity is essential for the subsequent coupling reaction with 7-aminocephalosporanic acid derivatives, where even trace impurities can affect the stereochemistry and biological activity of the final antibiotic. The process effectively isolates the dehydration variable, allowing for precise control over the reaction parameters and ensuring consistent batch-to-batch quality that meets the stringent specifications required for pharmaceutical grade intermediates.
How to Synthesize 2-(2-Aminothiazole-4-yl)-2-hydroxy Compound Efficiently
Implementing this synthesis route requires careful attention to solvent selection and temperature control to ensure complete dehydration without thermal degradation of the sensitive oxime group. The process begins by suspending the hydrated starting material in a dry ketone solvent, followed by a controlled heating phase to reflux temperatures which typically range between 55 to 60 degrees Celsius depending on the specific solvent boiling point. Detailed standard operating procedures for filtration, washing, and vacuum drying are essential to maintain the anhydrous state of the product prior to its immediate use in chlorination. The following guide outlines the critical operational parameters derived from the patent examples to ensure successful replication of this high-yield process.
- Suspend the hydrated 2-(2-aminothiazole-4-yl)-2-hydroxy compound in a ketone solvent such as acetone or methyl ethyl ketone.
- Heat the suspension to reflux temperatures, typically around 55-56°C for acetone, to facilitate the removal of crystal water.
- Filter the resulting crystals and dry them under reduced pressure to obtain the anhydrous product with moisture content below 0.5%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this anhydrous preparation method offers substantial strategic advantages beyond mere technical feasibility. The primary benefit lies in the drastic reduction of halogenating agent consumption, which directly correlates to a significant decrease in raw material expenditure per kilogram of final product. By lowering the requirement from three equivalents to nearly one, the process effectively triples the efficiency of one of the most costly and hazardous reagents in the synthesis chain. This efficiency gain allows for more competitive pricing structures and improved margin protection against volatility in the global market for phosphorus-based chemicals. Additionally, the simplified waste profile reduces the operational burden on environmental treatment facilities, lowering the total cost of ownership for the manufacturing site.
- Cost Reduction in Manufacturing: The elimination of excess halogenating agent usage results in direct material savings and reduces the need for expensive neutralization chemicals. By avoiding the consumption of reagents on water removal, the process ensures that every dollar spent on chemicals contributes directly to product formation rather than waste generation. This optimization leads to a leaner cost structure that enhances competitiveness in the global supply of antibiotic intermediates without compromising on quality or yield.
- Enhanced Supply Chain Reliability: Reducing the dependency on large volumes of hazardous halogenating agents mitigates supply risk and logistical complexities associated with transporting and storing dangerous chemicals. A process that requires less reagent is inherently more resilient to supply chain disruptions, ensuring consistent production schedules and reliable delivery timelines for downstream pharmaceutical customers. This stability is crucial for maintaining the continuity of antibiotic production lines which are critical for public health.
- Scalability and Environmental Compliance: The reduced generation of acidic and phosphorus-containing waste simplifies the effluent treatment process, making it easier to scale production to meet commercial demand while adhering to strict environmental regulations. Facilities can expand capacity without proportionally increasing their environmental footprint, supporting sustainable growth and long-term regulatory compliance. This alignment with green chemistry principles enhances the corporate reputation and ensures future-proofing against tightening environmental laws.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this anhydrous preparation technology. These insights are derived directly from the experimental data and process descriptions within the patent documentation to provide clarity on feasibility and performance. Understanding these details is essential for technical teams evaluating the integration of this method into existing manufacturing workflows.
Q: Why is removing crystal water critical for aminothiazole derivatives?
A: Crystal water interferes with subsequent halogenation reactions, requiring excessive amounts of halogenating agents like phosphorus pentachloride. Removing water allows for stoichiometric usage, reducing cost and waste.
Q: What solvents are effective for this dehydration process?
A: Ketone solvents such as acetone, methyl ethyl ketone, and methyl isobutyl ketone, as well as acetonitrile, are highly effective for suspending and dehydrating the crystals via reflux.
Q: How does this process impact environmental compliance?
A: By reducing the equivalent of halogenating agents from approximately 3 to 1.2, the process significantly lowers the generation of acidic waste and hazardous byproducts, easing environmental treatment burdens.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2-Aminothiazole-4-yl)-2-hydroxy Compound Supplier
NINGBO INNO PHARMCHEM stands at the forefront of pharmaceutical intermediate manufacturing, leveraging advanced process technologies like the one described in CN1232512C to deliver superior value to our global partners. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are seamlessly translated into robust industrial realities. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of aminothiazole derivative meets the exacting standards required for cephalosporin synthesis. Our commitment to quality and process optimization ensures that our clients receive intermediates that facilitate high-yield downstream reactions and consistent final drug quality.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific production needs. By partnering with our technical procurement team, you can access specific COA data and route feasibility assessments that demonstrate the tangible economic and operational benefits of our optimized synthesis routes. Let us help you streamline your supply chain and enhance your manufacturing efficiency with our proven expertise in complex pharmaceutical intermediate production.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
