Revolutionizing Beta-Lactam Synthesis: Direct Acylation of Amine Salts for Commercial Scale Production
Revolutionizing Beta-Lactam Synthesis: Direct Acylation of Amine Salts for Commercial Scale Production
The pharmaceutical industry constantly seeks robust synthetic routes that maximize yield while minimizing degradation, particularly for sensitive beta-lactam scaffolds. Patent CN1019300B introduces a transformative methodology for the preparation of carboxylic acid amides, specifically targeting the critical acylation step in synthesizing 7-acylamino-cephalosporin derivatives. Unlike conventional protocols that necessitate the liberation of the free amine base prior to reaction, this innovation demonstrates that amine acid addition salts can react directly with carboxylic acid 2-benzothiazolylthioesters. This breakthrough is particularly significant for manufacturers aiming to become a reliable cephalosporin intermediate supplier, as it addresses the inherent instability of 7-amino-cephalosporin free bases. By bypassing the neutralization step, the process not only streamlines operations but also significantly enhances the purity profile of the resulting antibacterial agents, offering a distinct competitive edge in the global supply chain for high-value antibiotic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the acylation of amines with activated esters like 2-benzothiazolyl thioesters requires the amine to be in its free base form to act as an effective nucleophile. In standard practice, if the starting material is an acid addition salt, which is often the case for stability reasons during storage and transport, at least one equivalent of an organic base, typically a tertiary amine, must be added to the reaction mixture. This pre-neutralization step introduces several complications for cost reduction in pharmaceutical manufacturing. Firstly, the generation of the free base can trigger decomposition pathways, especially for fragile molecules like 7-amino-cephalosporins which are prone to beta-lactam ring opening or epimerization under basic conditions. Secondly, the addition of stoichiometric amounts of organic bases generates salt by-products that must be removed during workup, increasing solvent consumption and waste disposal costs. Furthermore, residual organic bases can be difficult to completely purge from the final product, posing potential toxicity risks that require rigorous and expensive purification steps to meet stringent regulatory standards for active pharmaceutical ingredients.
The Novel Approach
The methodology disclosed in CN1019300B fundamentally alters this paradigm by enabling the direct reaction of the amine acid addition salt with the thioester acylating agent. This unexpected finding eliminates the prerequisite for pre-neutralization with organic bases, thereby preserving the structural integrity of sensitive substrates. For the commercial scale-up of complex beta-lactam antibiotics, this translates to a remarkably cleaner reaction profile. The process operates effectively at mild temperatures, typically ranging from 15°C to 30°C, and utilizes common inert solvents such as dichloromethane, which is identified as the optimal medium. By removing the base-mediated degradation pathway, manufacturers can achieve substantially higher yields of high-purity 7-acylamino-cephalosporin derivatives. This approach not only simplifies the operational workflow by reducing the number of reagents but also mitigates the formation of difficult-to-remove impurities, directly addressing the pain points of R&D directors focused on impurity control and process robustness.
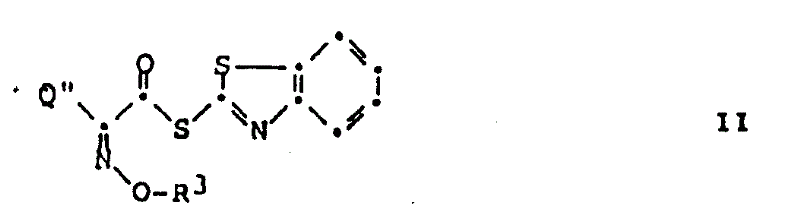
Mechanistic Insights into Direct Acylation of Amine Salts
The core mechanistic advantage of this process lies in the unique reactivity of the 2-benzothiazolyl thioester moiety combined with the specific conditions that allow the protonated amine salt to participate in the acylation. While classical organic chemistry suggests that a protonated amine is non-nucleophilic, the high electrophilicity of the thioester carbonyl carbon, activated by the electron-withdrawing benzothiazole sulfur system, facilitates the reaction even in the presence of the counter-ion. The reaction likely proceeds through a mechanism where the local environment or the specific solvation allows for a transient equilibrium or direct attack that bypasses the need for bulk deprotonation. This is critical for maintaining the stability of the beta-lactam ring, which is highly susceptible to nucleophilic attack by hydroxide ions or strong organic bases. By avoiding the introduction of external bases, the process minimizes the risk of beta-lactam hydrolysis, a common side reaction that leads to open-ring impurities which are notoriously difficult to separate from the desired product.
Furthermore, the choice of the 2-benzothiazolyl leaving group is instrumental in driving the reaction to completion with high selectivity. The leaving group ability of the benzothiazole thiolate is superior to many other ester leaving groups, ensuring rapid acylation kinetics at room temperature. This rapid kinetics is essential for reducing lead time for high-purity antibiotic intermediates, as it shortens the overall batch cycle time. From an impurity control perspective, the absence of tertiary amine salts in the reaction mixture simplifies the downstream processing. The workup typically involves simple aqueous washes to remove inorganic salts and unreacted starting materials, followed by crystallization. This streamlined purification strategy ensures that the final product meets rigorous quality specifications with minimal chromatographic intervention, which is a key factor in achieving cost-effective large-scale production.
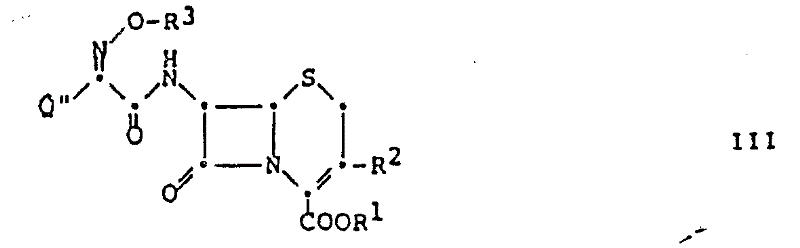
How to Synthesize 7-Acylamino-Cephalosporin Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this technology in a GMP environment. The process begins with the suspension of the 7-amino-cephalosporin acid addition salt, such as the hydrochloride or p-toluenesulfonate, in a suitable solvent like dichloromethane or acetone. The acylating agent, a carboxylic acid 2-benzothiazolyl thioester bearing the desired side chain (e.g., a 2-amino-4-thiazolyl glyoxylic acid derivative), is then added directly to the mixture. The reaction is maintained at ambient temperature, typically between 20°C and 25°C, with stirring continued for a period ranging from 1.5 to 3 hours depending on the specific substrate. Following the reaction, the mixture is subjected to a straightforward workup involving washing with aqueous sodium acetate or water to remove acidic by-products, followed by concentration and crystallization from solvents like isopropanol or ether. Detailed standardized synthesis steps see the guide below.
- Suspend the 7-amino-cephalosporin acid addition salt (e.g., hydrochloride or p-toluenesulfonate) in an inert organic solvent such as dichloromethane or acetone.
- Add the carboxylic acid 2-benzothiazolyl thioester acylating agent directly to the suspension at a controlled temperature between 15°C and 30°C.
- Stir the reaction mixture for 1.5 to 3 hours to ensure complete conversion, followed by standard workup procedures including washing, filtration, and crystallization to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this direct acylation technology offers profound strategic benefits beyond mere technical elegance. The elimination of the neutralization step represents a significant reduction in raw material costs, as expensive organic bases like DBU or triethylamine are no longer required in stoichiometric quantities. This directly contributes to cost reduction in pharmaceutical manufacturing by lowering the bill of materials and reducing the volume of chemical waste generated per kilogram of product. Moreover, the simplified workup procedure reduces solvent consumption and energy usage associated with distillation and drying, further enhancing the economic viability of the process. The ability to use stable acid addition salts as starting materials also improves supply chain reliability, as these salts are generally more stable and easier to store and transport than their free base counterparts, reducing the risk of raw material degradation before production.
- Cost Reduction in Manufacturing: The removal of the organic base neutralization step eliminates the cost of purchasing these reagents and the subsequent cost of disposing of the resulting salt waste. Additionally, the high purity of the crude product reduces the need for extensive recrystallization or chromatography, leading to substantial cost savings in downstream processing. The process operates at room temperature, eliminating the energy costs associated with heating or cooling reactors to extreme temperatures, which is a critical factor for energy-intensive chemical manufacturing.
- Enhanced Supply Chain Reliability: Utilizing acid addition salts as starting materials ensures greater stability during storage and logistics, minimizing the risk of raw material spoilage. The robustness of the reaction conditions, which tolerate a wide range of solvents and operate at ambient temperatures, reduces the dependency on specialized equipment or strict environmental controls. This flexibility allows for more agile production scheduling and reduces the likelihood of batch failures due to minor deviations in process parameters, ensuring a consistent and reliable supply of critical antibiotic intermediates to downstream customers.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the patent examples which cover gram to multi-gram scales without loss of efficiency. The use of common solvents like dichloromethane and acetone, which are widely available and have established recovery protocols, facilitates easy integration into existing manufacturing infrastructure. Furthermore, the reduction in chemical waste and the avoidance of heavy metal catalysts align with modern green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations and sustainability goals without compromising on productivity or product quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this direct acylation technology. These insights are derived directly from the experimental data and claims presented in patent CN1019300B, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating the feasibility of transferring this process to pilot or commercial scale, as well as for procurement teams assessing the long-term value proposition of suppliers utilizing this methodology.
Q: Why is reacting amine acid salts directly advantageous for cephalosporins?
A: 7-Amino-cephalosporins are often unstable in their free base form. This method avoids the need for organic base neutralization, preventing degradation and ensuring higher yields and purity of the final antibiotic derivative.
Q: What solvents are compatible with this acylation process?
A: The process is versatile and works in various inert organic solvents, with dichloromethane, acetone, and lower alcohols being particularly effective for dissolving reactants and facilitating the reaction at room temperature.
Q: Does this method eliminate the need for expensive catalysts?
A: Yes, the reaction proceeds efficiently without the need for additional organic bases or transition metal catalysts, simplifying the purification process and reducing the risk of heavy metal contamination in the final API.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Acylamino-Cephalosporin Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and robust synthetic routes in the production of life-saving antibiotics. Our technical team has extensively analyzed the advancements presented in CN1019300B and possesses the expertise to implement this direct acylation technology at an industrial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of high-quality intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 7-acylamino-cephalosporin derivatives meets the highest international standards for safety and efficacy.
We invite global pharmaceutical partners to collaborate with us to leverage this innovative synthesis method for your next project. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific production volumes and requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our adoption of this advanced acylation protocol can optimize your supply chain and enhance your product's market competitiveness. Let us be your trusted partner in delivering high-purity pharmaceutical intermediates with unmatched reliability and technical excellence.
