Advanced Acylation Technology for Stable 7-Amino-Cephalosporin Derivatives Manufacturing
Advanced Acylation Technology for Stable 7-Amino-Cephalosporin Derivatives Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes that balance high purity with operational simplicity, particularly when handling sensitive beta-lactam scaffolds. Patent CN1019300B introduces a transformative methodology for the preparation of carboxylic acid amides, specifically targeting the acylation of amines that are unstable in their free base form. This technology is particularly relevant for the synthesis of 7-acylamino-cephalosporin derivatives, where traditional methods often suffer from degradation issues. By enabling the direct reaction of amine acid addition salts with carboxylic acid 2-benzothiazolylthioesters, this process bypasses the critical and often problematic neutralization step. For R&D directors and procurement specialists, this represents a significant opportunity to enhance the reliability of pharmaceutical intermediate supplier networks while ensuring the structural integrity of complex antibiotic precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the acylation of amines to form carboxylic acid amides requires the amine to be in its reactive free base form. In the context of cephalosporin chemistry, the 7-amino group is often supplied as a stable acid addition salt to prevent decomposition during storage and handling. Conventional protocols mandate the addition of at least one equivalent of an organic base, typically a tertiary amine like triethylamine or DBU, to the reaction mixture to liberate the free amine prior to acylation. This extra step introduces several inefficiencies: it increases the complexity of the reaction mixture, necessitates the removal of the resulting amine salts during workup, and can generate exothermic conditions that threaten the stability of the sensitive beta-lactam ring. Furthermore, the presence of excess organic base can sometimes catalyze unwanted side reactions or epimerization, leading to a broader impurity profile that complicates downstream purification and reduces overall yield.
The Novel Approach
The innovative method disclosed in the patent data fundamentally alters this paradigm by demonstrating that amine acid addition salts can react directly with 2-benzothiazolylthioesters without prior neutralization. This unexpected finding allows for the acylation to proceed efficiently even when the amine remains in its salt form. The core of this chemistry relies on the high reactivity of the 2-benzothiazolylthioester moiety, which acts as a potent acylating agent capable of overcoming the kinetic barrier presented by the protonated amine. 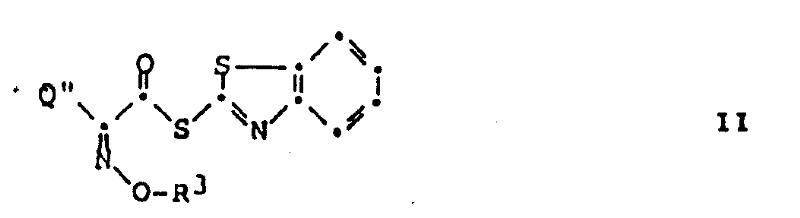 As illustrated in the structure above, the thioester linkage provides the necessary electrophilicity to drive the reaction forward at mild temperatures, typically between 15°C and 30°C. This elimination of the base neutralization step not only streamlines the process but also creates a milder reaction environment that is inherently safer for fragile molecular architectures like cephalosporins.
As illustrated in the structure above, the thioester linkage provides the necessary electrophilicity to drive the reaction forward at mild temperatures, typically between 15°C and 30°C. This elimination of the base neutralization step not only streamlines the process but also creates a milder reaction environment that is inherently safer for fragile molecular architectures like cephalosporins.
Mechanistic Insights into Direct Salt Acylation
From a mechanistic perspective, the ability of the acid addition salt to participate directly in nucleophilic attack suggests a dynamic equilibrium or a specific activation pathway facilitated by the thioester. While the amine is predominantly protonated, the high electrophilicity of the carbonyl carbon in the 2-benzothiazolylthioester likely allows for reaction with the small fraction of free amine present in equilibrium, or potentially through a concerted mechanism involving the counter-ion. This subtle interplay ensures that the concentration of the highly reactive free amine remains low throughout the process, which is a crucial factor in minimizing degradation pathways such as beta-lactam ring opening. By avoiding the sudden surge of free base concentration that occurs during traditional neutralization, the process maintains a controlled reaction rate that favors the formation of the desired amide bond over decomposition products.
Furthermore, this mechanism offers superior control over the impurity profile, a key metric for high-purity pharmaceutical intermediates. The absence of stoichiometric organic bases means there are no corresponding amine salts to separate during the isolation phase, significantly simplifying the crystallization process. The final products, such as the 7-acylamino-cephalosporin derivatives shown below, are obtained with exceptional purity directly from the reaction mixture after simple aqueous washes. 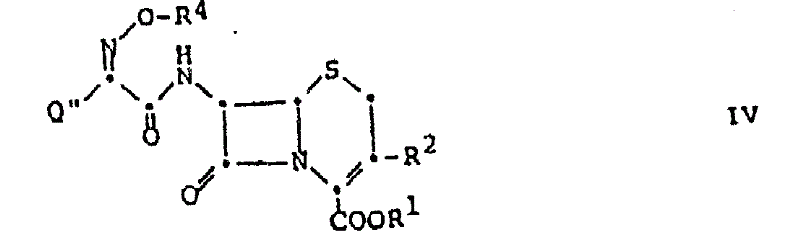 This structural integrity is vital for ensuring the biological efficacy of the final antibiotic drug substance. The method's versatility extends to various substituents at the 3-position of the cephalosporin nucleus, including heterocyclic groups like tetrazoles and thiadiazoles, proving its robustness across a wide range of commercially valuable antibiotic candidates.
This structural integrity is vital for ensuring the biological efficacy of the final antibiotic drug substance. The method's versatility extends to various substituents at the 3-position of the cephalosporin nucleus, including heterocyclic groups like tetrazoles and thiadiazoles, proving its robustness across a wide range of commercially valuable antibiotic candidates.
How to Synthesize 7-Acylamino-Cephalosporin Derivatives Efficiently
Implementing this synthesis route requires careful attention to solvent selection and temperature control to maximize the benefits of the direct salt reaction. The patent specifies that the process can be conducted in a wide variety of inert organic solvents where the starting materials are at least partially soluble, with dichloromethane being identified as the optimal choice for many embodiments. The operational simplicity allows for the direct suspension of the amine salt, followed by the addition of the thioester acylating agent, eliminating the need for precise pH monitoring or base dosing systems. Detailed standardized synthesis steps see the guide below.
- Suspend the 7-amino-cephalosporin acid addition salt in an inert organic solvent such as dichloromethane or acetone at room temperature.
- Add the carboxylic acid 2-benzothiazolylthioester acylating agent directly to the suspension without adding any organic base.
- Stir the mixture at 15°C to 30°C until reaction completion, followed by standard aqueous workup and crystallization to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible operational efficiencies and risk mitigation strategies. The primary advantage lies in the drastic simplification of the manufacturing workflow, which directly impacts the cost structure of producing complex antibiotic intermediates. By removing the requirement for expensive and hazardous organic bases like DBU, manufacturers can achieve cost reduction in pharmaceutical intermediate manufacturing through both raw material savings and reduced waste disposal costs. Additionally, the simplified workup procedure reduces the consumption of solvents and energy required for extensive purification steps, further enhancing the economic viability of large-scale production runs without compromising on quality standards.
- Cost Reduction in Manufacturing: The elimination of stoichiometric organic bases removes a significant line item from the bill of materials, while simultaneously reducing the load on wastewater treatment systems due to the absence of amine salts. This leaner chemical footprint allows for more competitive pricing models for bulk buyers seeking reliable sources of cephalosporin side chains. The process efficiency also means higher throughput per batch, as time-consuming neutralization and salt filtration steps are completely bypassed, leading to substantial overall cost savings in the production lifecycle.
- Enhanced Supply Chain Reliability: Operating at mild temperatures between 15°C and 30°C reduces the energy demand and thermal stress on equipment, facilitating easier commercial scale-up of complex pharmaceutical intermediates. The robustness of the reaction against variations in amine stability ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply lines to downstream API manufacturers. This reliability minimizes the risk of production delays caused by failed batches or extensive reprocessing, securing a steady flow of materials for global antibiotic markets.
- Scalability and Environmental Compliance: The use of common solvents like dichloromethane and the generation of fewer byproducts align well with modern environmental regulations and green chemistry principles. The process generates less hazardous waste compared to traditional base-mediated methods, simplifying compliance with strict environmental discharge standards. This environmental compatibility makes the technology attractive for facilities aiming to reduce their carbon footprint while maintaining high production volumes, ensuring long-term sustainability for the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this direct acylation technology. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into existing production lines. The answers are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: Why is reacting amine acid salts directly advantageous for cephalosporins?
A: Direct reaction eliminates the need for unstable free base formation, significantly reducing degradation of the sensitive beta-lactam ring and improving overall yield.
Q: What solvents are compatible with this novel acylation method?
A: The process is highly versatile, working effectively in halogenated hydrocarbons like dichloromethane, as well as lower alcohols and amides, providing flexibility for scale-up.
Q: Does this method require expensive organic bases like DBU?
A: No, the innovation specifically removes the requirement for stoichiometric organic bases, thereby simplifying purification and reducing raw material costs substantially.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Amino-Cephalosporin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the direct salt acylation process are translated into reliable industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of cephalosporin intermediate meets the highest international standards for safety and efficacy, providing our partners with absolute confidence in our supply.
We invite you to engage with our technical procurement team to discuss how this technology can optimize your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits of switching to this streamlined process for your projects. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being your strategic partner in high-quality chemical manufacturing.
