Advanced Synthesis of 4-Fluoro-2,3-Dimethylphenyl Methanesulfonate for Commercial Scale-Up
The chemical industry is constantly evolving to meet the dual demands of economic efficiency and environmental sustainability, a challenge vividly addressed in the recent patent literature regarding the synthesis of specialized sulfonate esters. Specifically, patent number CN110776441B discloses a robust and innovative methodology for the preparation of 4-fluoro-2,3-dimethylphenyl methanesulfonate, a critical building block with extensive applications in the development of advanced agrochemicals and pharmaceutical active ingredients. This technical disclosure marks a significant departure from legacy synthetic pathways that have long plagued manufacturers with high operational costs and severe environmental liabilities. By re-engineering the molecular construction from the ground up, starting with the readily available and cost-effective 3-fluoroxylene, the inventors have established a sequence that integrates halogenation, coupling, hydrolysis, and esterification into a cohesive and commercially viable workflow. For R&D directors and procurement strategists alike, this patent represents more than just a new recipe; it offers a strategic pathway to secure supply chains for high-value fluorinated intermediates while simultaneously mitigating the risks associated with heavy metal catalysts and hazardous waste streams.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorinated phenyl sulfonates has been fraught with significant technical and economic hurdles that hinder efficient commercial scale-up. Prior art methods, such as those referenced in the background of the patent, typically initiate the synthesis from 4-nitro-2,3-dimethylphenol, necessitating a multi-step reduction process to convert the nitro group into an amine. This reduction step invariably requires the use of palladium-carbon, a noble metal catalyst that not only commands a high market price but also introduces complex purification challenges to remove trace metal residues from the final API intermediate. Furthermore, the subsequent conversion of the amine to the fluoride via diazotization is notoriously problematic, requiring harsh reagents like pyridine-hydrogen fluoride and generating substantial volumes of nitrogen-containing wastewater that impose a heavy burden on environmental treatment facilities. These conventional routes are characterized by low atom economy, high energy consumption due to hydrogenation requirements, and a safety profile that is less than ideal for large-scale continuous manufacturing, making them increasingly unattractive in a regulatory environment that demands greener chemistry.
The Novel Approach
In stark contrast to the cumbersome legacy processes, the novel approach detailed in CN110776441B leverages a clever retrosynthetic strategy that bypasses the need for nitro reduction and diazotization entirely. By selecting 3-fluoroxylene as the foundational raw material, the synthesis capitalizes on the inherent reactivity of the aromatic ring to introduce the necessary functional groups through a sequence of electrophilic substitution and nucleophilic aromatic substitution reactions. This paradigm shift eliminates the dependency on expensive noble metal catalysts and avoids the generation of hazardous diazonium intermediates, thereby streamlining the process flow and significantly lowering the barrier to entry for mass production. The integration of a copper-catalyzed coupling reaction allows for the precise installation of the oxygen functionality, which is subsequently revealed through a controlled hydrolysis step, demonstrating a level of chemoselectivity that preserves the integrity of the fluorine substituent throughout the synthesis. This methodology not only enhances the overall yield potential by reducing the number of purification bottlenecks but also aligns perfectly with modern principles of green chemistry by minimizing waste discharge and utilizing more benign reaction conditions.
Mechanistic Insights into the Four-Step Synthetic Sequence
The core of this technological advancement lies in the precise orchestration of four distinct chemical transformations, each optimized to maximize conversion while minimizing byproduct formation. The journey begins with the regioselective halogenation of 3-fluoroxylene, where the electronic effects of the existing substituents direct the incoming halogen atom to the para-position relative to the fluorine, yielding the key intermediate 4-fluoro-2,3-dimethyl halobenzene. This step is critical as it sets the stereochemical and electronic stage for the subsequent coupling reaction, which proceeds via a copper-mediated mechanism to form the aryl alkyl ether. Following the formation of the ether linkage, the process employs a robust alkaline hydrolysis to cleave the alkyl group, effectively unmasking the phenolic hydroxyl group without compromising the sensitive carbon-fluorine bond. Finally, the resulting phenol undergoes esterification with methylsulfonyl chloride in the presence of an acid-binding agent, locking in the final sulfonate structure with high fidelity.
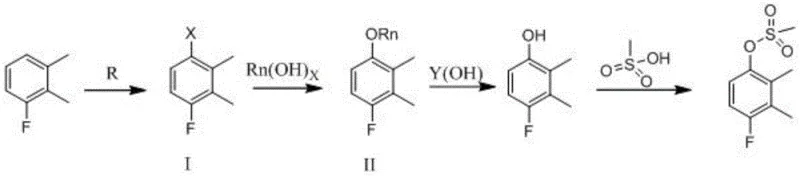
As illustrated in the comprehensive reaction scheme above, the transition from the halogenated intermediate to the final sulfonate ester is achieved through a logical progression of functional group interconversions that avoid redox-heavy steps. The use of copper bromide in the coupling phase facilitates the formation of the carbon-oxygen bond under relatively mild thermal conditions, while the subsequent hydrolysis in dimethyl sulfoxide with potassium hydroxide ensures complete conversion to the phenol. This mechanistic pathway is particularly advantageous for maintaining high purity profiles, as the intermediates formed are chemically distinct and easily separable via standard extraction and crystallization techniques. For the technical team, understanding this mechanism is vital for troubleshooting potential impurities, such as over-halogenated species or unreacted starting materials, ensuring that the final product meets the stringent specifications required for downstream drug synthesis.
How to Synthesize 4-Fluoro-2,3-Dimethylphenyl Methanesulfonate Efficiently
Implementing this synthesis in a pilot or production plant requires strict adherence to the reaction parameters outlined in the patent examples to ensure reproducibility and safety. The process begins with the controlled addition of halogenating agents to 3-fluoroxylene at low temperatures to manage exothermicity, followed by a high-temperature coupling step that requires pressure-rated equipment. Detailed standard operating procedures for workup, including specific solvent ratios for extraction and precise pH controls during hydrolysis, are essential for maximizing recovery at each stage. While the general pathway is robust, minor adjustments in stoichiometry and temperature may be required depending on the specific grade of raw materials sourced, necessitating a thorough process validation before full-scale deployment.
- Perform halogenation on 3-fluoroxylene using bromine or similar agents to obtain 4-fluoro-2,3-dimethyl halobenzene.
- Execute a copper-catalyzed coupling reaction with an alcohol to form the corresponding phenyl ether intermediate.
- Hydrolyze the ether under alkaline conditions to yield 4-fluoro-2,3-dimethylphenol, followed by esterification with methylsulfonyl chloride.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound advantages that extend far beyond the laboratory bench, directly impacting the bottom line and supply chain resilience for global buyers. By shifting the raw material base to 3-fluoroxylene, a commodity chemical with a stable and abundant supply, manufacturers can decouple their production costs from the volatile pricing of noble metals and specialized nitro-compounds. This strategic sourcing decision inherently de-risks the supply chain, ensuring that production schedules are not disrupted by shortages of exotic catalysts or regulatory crackdowns on hazardous reagents. Furthermore, the elimination of the diazotization step removes a major bottleneck related to wastewater treatment capacity, allowing facilities to operate at higher throughput without incurring prohibitive environmental compliance costs.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the complete removal of palladium-carbon catalysts from the process flow, which eradicates the need for expensive metal scavenging steps and reduces the cost of goods sold significantly. Additionally, the use of common solvents and reagents such as methanol, acetic acid, and triethylamine ensures that operational expenditures remain predictable and low, avoiding the premium pricing associated with specialized fluorinating agents or high-pressure hydrogenation services. The simplified purification train, which relies on standard crystallization rather than complex chromatography, further drives down processing costs by reducing solvent consumption and labor hours per kilogram of product.
- Enhanced Supply Chain Reliability: Utilizing 3-fluoroxylene as the starting material provides a distinct logistical advantage, as this feedstock is produced by multiple global suppliers, preventing single-source dependency. The robustness of the chemical steps, which do not rely on moisture-sensitive organometallics or unstable diazonium salts, translates to higher batch success rates and more consistent delivery timelines for customers. This reliability is crucial for pharmaceutical companies managing tight inventory windows, as it minimizes the risk of production delays caused by failed batches or extended quality control testing associated with metal impurities.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing unit operations that are standard in the fine chemical industry, such as stirred tank reactors and liquid-liquid extraction columns. By avoiding the generation of heavy metal waste and nitrogen-rich effluent, the synthesis aligns with increasingly stringent global environmental regulations, reducing the long-term liability and permitting hurdles for manufacturing sites. This 'green' profile not only lowers waste disposal costs but also enhances the corporate sustainability metrics of the supply chain, a factor that is becoming progressively important for end-users in the European and North American markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, derived directly from the comparative data and experimental examples provided in the patent documentation. These insights are intended to clarify the operational benefits and technical nuances for stakeholders evaluating this technology for potential licensing or procurement partnerships. Understanding these details is essential for making informed decisions about integrating this intermediate into broader drug substance manufacturing campaigns.
Q: Why is the new synthesis route preferred over the traditional nitro-reduction method?
A: The traditional method relies on expensive palladium-carbon catalysts for hydrogenation and hazardous diazotization steps that generate significant wastewater. The new route utilizes abundant 3-fluoroxylene and avoids noble metals entirely, drastically reducing both raw material costs and environmental compliance burdens.
Q: What represents the critical quality control point in this synthesis?
A: The regioselectivity during the initial halogenation of 3-fluoroxylene is crucial to ensure the formation of the 4-fluoro-2,3-dimethyl isomer. Additionally, the final recrystallization step is vital for achieving the high purity levels (over 98%) required for sensitive pharmaceutical applications.
Q: Can this process be scaled for industrial production?
A: Yes, the process utilizes standard unit operations such as halogenation, extraction, and distillation which are easily scalable. The avoidance of high-pressure hydrogenation and unstable diazonium salts significantly enhances operational safety and feasibility for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Fluoro-2,3-Dimethylphenyl Methanesulfonate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and a commitment to quality excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity profiles described in CN110776441B can be reliably replicated on an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch of 4-fluoro-2,3-dimethylphenyl methanesulfonate meets the exacting standards required for pharmaceutical and agrochemical applications.
We invite forward-thinking procurement leaders and R&D directors to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this greener, more efficient supply source. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of innovation, reliability, and cost-effectiveness.
