Revolutionizing N-Sulfonyl Amidine Production: A Safe, Metal-Free Strategy for Industrial Scale-Up
The landscape of organic synthesis for nitrogen-containing heterocycles is undergoing a significant transformation, driven by the urgent need for safer, more sustainable, and cost-effective manufacturing protocols. Patent CN114920616A, published in August 2022, introduces a groundbreaking methodology for the synthesis of amidine compounds, specifically N-sulfonyl amidines, which serve as critical building blocks in the development of antibiotics, anti-inflammatory agents, and acaricides. This innovation addresses long-standing industrial pain points by replacing hazardous reagents and expensive transition metal catalysts with a benign organophosphorus activation system. For R&D directors and process chemists, this represents a paradigm shift from high-risk, high-cost methodologies to a robust, scalable platform that utilizes commercially abundant triphenylphosphine and carbon tetrachloride. The strategic value of this patent lies not only in its chemical elegance but in its direct applicability to the commercial scale-up of complex pharmaceutical intermediates, offering a pathway to reduce production costs while enhancing operational safety profiles across the supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the amidine scaffold has relied heavily on methodologies that pose significant challenges for industrial implementation and environmental compliance. Traditional Route 1, for instance, employs zinc catalysts and diazonium esters under reflux conditions for extended periods, creating substantial explosion hazards and generating heavy metal waste streams that require expensive remediation. Similarly, Route 2 utilizes precious palladium chloride catalysts at elevated temperatures, driving up raw material costs and introducing the risk of toxic metal contamination in the final product, which is unacceptable for pharmaceutical applications. Other existing strategies involve the use of tert-butyl hydroperoxide, a first-class organic oxidant known for its thermal instability and potential for violent decomposition, or electrochemical methods that suffer from poor scalability and high equipment capital expenditure. These conventional approaches collectively suffer from harsh reaction conditions, limited substrate scope, and prohibitive safety risks, making them ill-suited for the rigorous demands of modern GMP manufacturing environments where consistency and safety are paramount.
The Novel Approach
In stark contrast to these legacy methods, the novel process disclosed in the patent leverages an in situ generated phosphonium salt activation system to drive the condensation of sulfonamides and formamides under remarkably mild conditions. By utilizing triphenylphosphine and carbon tetrachloride as activator precursors, the reaction proceeds efficiently at a low temperature of 40°C, eliminating the need for high-energy heating or dangerous oxidants. This metal-free strategy not only drastically reduces the environmental footprint by avoiding transition metals but also simplifies the reaction setup to a standard nitrogen-atmosphere protocol using common solvents like dichloromethane or DMF. The versatility of this approach is evidenced by its compatibility with a wide array of substrates, including electron-deficient and electron-rich aryl sulfonamides, heterocyclic variants, and diverse formamide derivatives, ensuring high yields without the need for specialized catalysts. 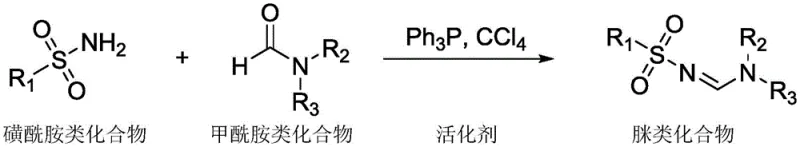
Mechanistic Insights into Organophosphorus-Mediated Condensation
The core mechanistic advantage of this synthesis lies in the generation of a highly reactive phosphonium intermediate, which acts as a potent dehydrating and activating agent for the nucleophilic attack of the sulfonamide nitrogen on the formamide carbonyl carbon. Unlike transition metal catalysis which often involves complex oxidative addition and reductive elimination cycles prone to side reactions, this organophosphorus mechanism operates through a straightforward activation-dehydration pathway that minimizes the formation of difficult-to-remove byproducts. The use of carbon tetrachloride in conjunction with triphenylphosphine facilitates the formation of the active species at ambient to mild thermal conditions, ensuring that sensitive functional groups on the aromatic rings or heterocyclic cores remain intact throughout the transformation. This mechanistic simplicity translates directly to process robustness, as the reaction kinetics are predictable and less susceptible to the trace impurities that often poison metal catalysts, thereby ensuring consistent batch-to-batch reproducibility essential for commercial production.
Furthermore, the absence of metal catalysts fundamentally alters the impurity profile of the crude reaction mixture, removing the burden of heavy metal scavenging from the downstream purification process. In traditional palladium or copper-catalyzed routes, residual metal levels must be reduced to parts-per-million (ppm) levels to meet regulatory standards for drug substances, often requiring additional chromatography or specialized resin treatments that lower overall yield and increase cycle time. By employing a purely organic activation system, this novel method inherently produces a cleaner crude product where the primary impurities are organic phosphine oxides, which are generally easier to separate via standard aqueous workup and crystallization techniques. This intrinsic purity advantage significantly streamlines the isolation of high-purity N-sulfonyl amidines, reducing the consumption of silica gel and solvents during purification and enhancing the overall mass balance efficiency of the manufacturing process.
How to Synthesize N-Sulfonyl Amidines Efficiently
The practical implementation of this synthesis protocol is designed for ease of operation, requiring standard laboratory glassware and readily available reagents that do not demand specialized storage or handling precautions beyond standard inert atmosphere techniques. The process begins with the preparation of a dry reaction vessel purged with nitrogen, into which the sulfonamide substrate, triphenylphosphine, and the chosen solvent are sequentially added to form a homogeneous mixture. Following this, carbon tetrachloride and the formamide coupling partner are introduced to initiate the activation sequence, after which the reaction mixture is gently heated to maintain a steady temperature of 40°C for a duration typically ranging from 8 to 18 hours depending on the specific electronic nature of the substrates. Detailed standardized synthetic steps see the guide below.
- Prepare the reaction vessel under nitrogen atmosphere and add sulfonamide, triphenylphosphine, and solvent (DCM or DMF).
- Introduce carbon tetrachloride and formamide to the mixture to generate the active phosphonium species in situ.
- Heat the reaction mixture to 40°C for approximately 12 hours, then quench with water and purify via extraction and chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this metal-free synthesis route offers compelling economic and logistical benefits that extend far beyond the laboratory bench. The elimination of precious metal catalysts such as palladium and the avoidance of hazardous oxidants like peroxides directly translate to a significant reduction in raw material procurement costs and insurance premiums associated with storing dangerous chemicals. Moreover, the reliance on commodity chemicals like triphenylphosphine and carbon tetrachloride ensures a stable and resilient supply chain, mitigating the risks of price volatility and availability shortages that frequently plague the market for specialized transition metal complexes and ligands. This stability allows for more accurate long-term budget forecasting and reduces the administrative burden of sourcing multiple specialized reagents from different vendors.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and the associated heavy metal removal steps results in substantial cost savings throughout the production lifecycle. By simplifying the purification workflow and eliminating the need for metal scavengers, manufacturers can reduce solvent consumption and waste disposal costs, leading to a more economically efficient process that improves overall profit margins without compromising product quality.
- Enhanced Supply Chain Reliability: Utilizing widely available, non-proprietary reagents ensures that production schedules are not held hostage by the lead times of niche catalyst suppliers. The robustness of the reaction conditions means that manufacturing can proceed with minimal downtime or batch failures due to catalyst deactivation, thereby guaranteeing consistent delivery timelines and strengthening the reliability of the supply chain for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: The mild reaction temperature of 40°C and the absence of explosive reagents make this process inherently safer and easier to scale from kilogram to multi-ton production volumes. This safety profile aligns perfectly with increasingly stringent environmental and safety regulations, reducing the regulatory hurdles for plant approval and minimizing the environmental impact of chemical manufacturing operations through greener chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology, providing clarity on its operational parameters and strategic benefits for potential partners. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, ensuring that decision-makers have access to accurate and actionable information. Understanding these details is crucial for evaluating the feasibility of integrating this process into existing manufacturing portfolios.
Q: What are the primary safety advantages of this new amidine synthesis method compared to traditional routes?
A: Unlike traditional methods that utilize explosive diazonium esters or hazardous organic peroxides like tert-butyl hydroperoxide, this process employs stable, commercially available reagents (triphenylphosphine and carbon tetrachloride) at mild temperatures (40°C), significantly reducing explosion risks and thermal hazards.
Q: Does this metal-free protocol support a broad range of substrate functional groups?
A: Yes, the method demonstrates excellent functional group tolerance, successfully accommodating aryl sulfonamides with electron-withdrawing or donating groups, heterocyclic sulfonamides (such as thiophene and isoxazole derivatives), and various formamides including cyclic and benzyl-substituted variants.
Q: How does eliminating transition metal catalysts impact the purification process for pharmaceutical grades?
A: By avoiding expensive and toxic transition metals like palladium, zinc, or copper, the process eliminates the need for complex and costly heavy metal scavenging steps, simplifying the downstream purification workflow and ensuring easier compliance with strict residual metal limits in API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Sulfonyl Amidines Supplier
As the global demand for high-quality pharmaceutical intermediates continues to rise, NINGBO INNO PHARMCHEM stands ready to leverage this cutting-edge synthesis technology to deliver superior value to our clients. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of N-sulfonyl amidines meets the exacting standards required for drug substance manufacturing, providing our partners with peace of mind and regulatory confidence.
We invite forward-thinking organizations to collaborate with us to explore the full potential of this metal-free synthesis route for their specific pipeline needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your project, and to obtain specific COA data and route feasibility assessments that demonstrate how we can optimize your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to not just a chemical supplier, but a strategic ally dedicated to driving innovation and efficiency in your drug development journey.
