Advanced Manufacturing of Sitafloxacin Intermediate via Novel Metal-Mediated Coupling
Advanced Manufacturing of Sitafloxacin Intermediate via Novel Metal-Mediated Coupling
The pharmaceutical landscape for broad-spectrum antibacterial agents continues to evolve, with Sitafloxacin standing out as a potent fourth-generation quinolone capable of overcoming resistant bacterial strains. Central to the efficient production of this critical medication is the availability of high-quality precursors, specifically the chemical entity known as Sitafloxacin Intermediate II. A groundbreaking preparation method disclosed in patent CN109293513B offers a transformative approach to synthesizing this key building block. By leveraging a novel metal-mediated coupling strategy, this technology addresses longstanding inefficiencies in quinolone manufacturing, providing a pathway that combines high yield with operational simplicity. For global supply chain leaders and R&D directors, understanding the nuances of this patented process is essential for securing a competitive edge in the production of next-generation antibiotics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex quinolone intermediates has been plagued by convoluted reaction sequences that hinder scalability and economic viability. As illustrated in prior art references, such as the work by H. Liu et al., traditional routes often involve multiple protection and deprotection steps, leading to significant material loss at each stage. These legacy processes typically require harsh reaction conditions and expensive reagents, resulting in a cumulative yield that is often suboptimal for industrial applications. Furthermore, the post-treatment procedures associated with these older methods are frequently labor-intensive, involving complex chromatographic separations to remove stubborn impurities generated by side reactions. This not only drives up the cost of goods sold (COGS) but also creates bottlenecks in production capacity, making it difficult to respond rapidly to market demand fluctuations.
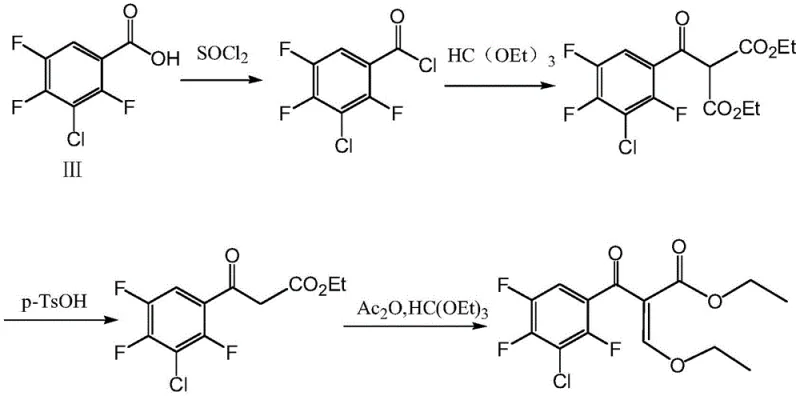
The Novel Approach
In stark contrast to the cumbersome legacy pathways, the methodology outlined in patent CN109293513B introduces a streamlined, direct coupling strategy that fundamentally simplifies the molecular construction of the intermediate. This innovative route bypasses unnecessary synthetic detours by directly reacting 3-chloro-2,4,5-trifluorobenzoyl chloride with 3-bromo-2-ethoxy ethyl acrylate in the presence of a low-valence metal. The elegance of this approach lies in its ability to forge the critical carbon-carbon bond in a single, highly efficient step under mild conditions. By eliminating the need for complex protecting group chemistry, the process drastically reduces the number of unit operations required, thereby minimizing solvent usage and waste generation. This shift represents a paradigm change in how fluoroquinolone intermediates are manufactured, prioritizing atom economy and process intensification without compromising on the structural integrity of the final product.
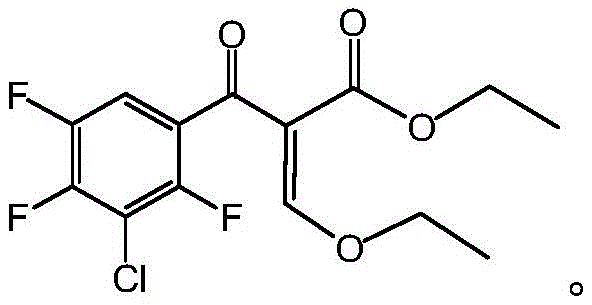
Mechanistic Insights into Metal-Mediated Acylation
The core of this technological advancement relies on the precise activation of the acyl chloride species through coordination with low-valence metals such as Zinc, Iron, or Magnesium. Mechanistically, the metal serves to generate an organometallic intermediate in situ from the bromo-acrylate species, which then acts as a potent nucleophile towards the electrophilic carbonyl carbon of the benzoyl chloride. This interaction facilitates a smooth addition-elimination sequence that constructs the beta-keto ester framework essential for the subsequent cyclization into the quinolone core. The choice of metal is critical; for instance, Zinc powder offers a balanced reactivity profile that minimizes homocoupling side reactions while ensuring rapid conversion. The reaction kinetics are further optimized by maintaining strict temperature control between -20°C and 10°C, which suppresses thermal degradation and ensures high regioselectivity during the bond-forming event.
From an impurity control perspective, this mechanism offers distinct advantages over acid-catalyzed or base-mediated alternatives. The mild nature of the metal-mediated coupling prevents the hydrolysis of the sensitive ester and acyl chloride functionalities, which are common failure points in traditional syntheses. Additionally, the use of dichloromethane as a solvent provides an ideal medium for solubilizing both the organic substrates and the metal surface, promoting homogeneous reaction progress. Any residual metal salts formed during the process are easily removed through a simple aqueous wash, leaving behind a product stream with a remarkably clean impurity profile. This inherent cleanliness reduces the reliance on energy-intensive recrystallization steps, allowing manufacturers to achieve purity specifications exceeding 99% with minimal downstream processing effort.
How to Synthesize Sitafloxacin Intermediate Efficiently
Implementing this synthesis requires careful attention to reagent stoichiometry and thermal management to maximize the benefits of the patented protocol. The process begins with the activation of the benzoic acid starting material, followed by the critical metal-mediated coupling step which dictates the overall success of the campaign. Operators must ensure that the acyl chloride is generated quantitatively before introduction to the metal slurry to prevent competitive hydrolysis. The following guide outlines the standardized operational parameters derived from the patent examples, serving as a foundational reference for process engineers aiming to replicate these results in a GMP environment.
- Convert 3-chloro-2,4,5-trifluorobenzoic acid to its corresponding acyl chloride using thionyl chloride or oxalyl chloride in dichloromethane at 0-10°C.
- Prepare a reaction mixture of 3-bromo-2-ethoxy ethyl acrylate and a low-valence metal (such as Zinc or Iron powder) in dichloromethane.
- Slowly add the acyl chloride solution to the metal/acrylate mixture at low temperature (-20 to 10°C), stir, and perform aqueous workup to isolate the intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this novel synthetic route translates into tangible operational improvements that extend far beyond the laboratory bench. The simplification of the chemical pathway directly correlates with a reduction in raw material inventory requirements, as fewer distinct reagents and solvents are needed to complete the synthesis. This consolidation of the supply base mitigates the risk of disruptions caused by the scarcity of specialized reagents, ensuring a more resilient production schedule. Furthermore, the high efficiency of the reaction means that less starting material is wasted, effectively lowering the input cost per kilogram of the final intermediate. These factors combine to create a manufacturing profile that is not only cost-effective but also robust against the volatility of the global chemical market.
- Cost Reduction in Manufacturing: The elimination of complex multi-step sequences significantly lowers the operational expenditure associated with labor, energy, and equipment usage. By removing the need for expensive transition metal catalysts often used in cross-coupling reactions and replacing them with abundant metals like Zinc or Iron, the direct material costs are substantially reduced. Additionally, the high solvent recovery rate mentioned in the patent allows for the recycling of dichloromethane, further diminishing the recurring cost of consumables. This lean manufacturing approach ensures that the cost of goods remains competitive even when scaling to multi-ton production volumes.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as thionyl chloride, zinc powder, and basic halogenated solvents ensures that the supply chain is not dependent on single-source suppliers of exotic reagents. This accessibility guarantees consistent availability of inputs, reducing lead times and preventing production delays. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in raw material quality, providing a buffer against supply chain fluctuations. Consequently, manufacturers can maintain steady output levels to meet the rigorous demands of pharmaceutical clients.
- Scalability and Environmental Compliance: The mild reaction temperatures and ambient pressure conditions make this process inherently safer and easier to scale from pilot plant to commercial manufacturing. The reduction in side reactions leads to a cleaner waste stream, simplifying effluent treatment and lowering the environmental compliance burden. High yields mean less chemical waste is generated per unit of product, aligning with green chemistry principles and corporate sustainability goals. This environmental efficiency is increasingly becoming a key differentiator in vendor selection processes for major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and advantages of the technology. Understanding these details is crucial for technical teams evaluating the integration of this route into their existing manufacturing portfolios.
Q: What is the purity profile of the Sitafloxacin intermediate produced via this method?
A: The patented process achieves a crude purity of approximately 90% which can be easily upgraded to >99% through conventional post-treatment, significantly reducing the burden on downstream purification compared to prior art methods.
Q: Which metal catalysts are compatible with this synthesis route?
A: The method is robust and flexible, accommodating various low-valence or transition metals including Magnesium, Copper, Zinc, Iron, and Cobalt, allowing manufacturers to select based on cost and availability.
Q: How does this process improve upon the traditional Liu et al. synthetic route?
A: Unlike the traditional route which suffers from long step counts and complex post-treatment, this novel approach utilizes a direct coupling strategy that simplifies the workflow, reduces energy consumption, and improves overall yield to over 96%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sitafloxacin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced antibiotics like Sitafloxacin depends on the reliability and quality of the supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of global pharmaceutical partners. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of Sitafloxacin Intermediate meets the highest industry standards. Our commitment to technical excellence allows us to navigate the complexities of fluoroquinolone synthesis with precision and consistency.
We invite you to collaborate with us to leverage this innovative manufacturing technology for your antibiotic programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain for Sitafloxacin production.
