Advanced Synthetic Route for Novel Pleuromutilin Compounds Targeting Commercial Scale-up
Advanced Synthetic Route for Novel Pleuromutilin Compounds Targeting Commercial Scale-up
The pharmaceutical landscape is constantly evolving in the search for potent antibiotics that can combat resistant bacterial strains, and the recent disclosure in patent CN108929253B presents a significant breakthrough in the field of pleuromutilin derivatives. This intellectual property outlines a robust and highly efficient preparation method for a novel pleuromutilin compound, specifically characterized by the molecular architecture designated as Formula I. For research and development directors overseeing antibiotic pipelines, this patent offers a compelling alternative to traditional semi-synthetic routes that have historically been plagued by low conversion rates and complex purification challenges. The invention belongs to the domain of drug synthesis, focusing on modifying the diterpenoid parent nucleus to enhance antibacterial efficacy while simultaneously optimizing the manufacturing process for industrial viability. By targeting the 8th position side chain through a sequence of hydroxy mesylation and nucleophilic substitution, the inventors have unlocked a pathway that not only improves yield but also ensures high purity standards essential for regulatory approval. 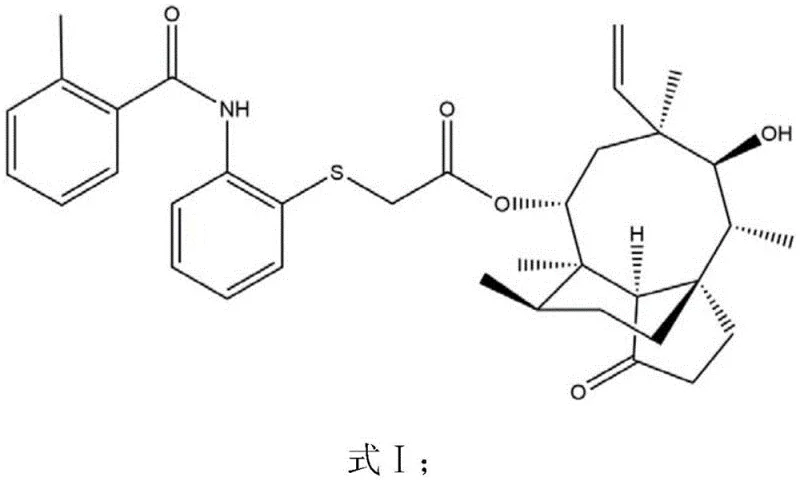
The strategic importance of this technology cannot be overstated for procurement managers and supply chain heads who are tasked with securing reliable sources of high-value active pharmaceutical ingredients (APIs) and intermediates. Pleuromutilins are renowned for their unique mechanism of action, inhibiting peptidyl transferase by binding to the 50s subunit of bacterial ribosomes, which makes them effective against pathogens that have developed resistance to macrolides and lincosamides. However, the commercialization of such compounds has often been hindered by the inherent difficulties in modifying the pleuromutilin core without degrading its biological activity. This patent addresses those pain points directly by introducing a streamlined synthetic protocol that utilizes readily available and cost-effective raw materials such as p-toluenesulfonyl chloride and o-methylbenzoic acid. The result is a process that aligns perfectly with the goals of green ecology and sustainable development, offering a tangible solution for cost reduction in antibiotic manufacturing without compromising on the quality or potency of the final therapeutic agent.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of pleuromutilin derivatives has faced substantial hurdles related to physicochemical properties and synthetic efficiency. Most existing derivatives suffer from poor water solubility, which leads to difficult absorption profiles, short biological half-lives, and low utilization rates in clinical and veterinary settings. From a process chemistry perspective, modifying the mother nucleus structure of pleuromutilin is notoriously challenging; more than 1000 derivatives have been synthesized to date, yet very few have progressed to become effective candidates or approved veterinary drugs. Conventional methods often involve harsh reaction conditions that can degrade the sensitive diterpenoid skeleton, resulting in yields that typically hover between 10% and 50%. Furthermore, traditional routes frequently require expensive transition metal catalysts or aggressive reagents that cause significant corrosion to production equipment, thereby increasing maintenance costs and shortening the operational lifespan of reactors. These factors collectively contribute to a high cost of goods sold (COGS) and create supply chain vulnerabilities that make it difficult to scale production to meet global demand for next-generation antibiotics.
The Novel Approach
In stark contrast to these legacy methods, the approach detailed in patent CN108929253B introduces a sophisticated yet operationally simple strategy that leverages mild reaction conditions to achieve superior outcomes. The novel route initiates with the activation of the pleuromutilin side chain via hydroxy mesylation, followed by a precise nucleophilic substitution with diaminobenzenethiol. This sequence is meticulously controlled, utilizing phase transfer catalysis with benzyltriethylammonium chloride (TEBAC) to facilitate the reaction between organic and aqueous phases efficiently. The final condensation step with o-methylbenzoic acid or its chloride derivative is performed at ambient temperatures ranging from 20°C to 30°C, eliminating the need for energy-intensive heating or cooling cycles. This methodological shift results in a dramatic improvement in production metrics, with reported yields exceeding 79%, which represents a 30-70% increase over existing technologies. By simplifying the synthetic route and minimizing the number of unit operations, this approach not only accelerates the production period but also significantly reduces the generation of hazardous waste, positioning it as an ideal candidate for modern, eco-conscious pharmaceutical manufacturing facilities.
Mechanistic Insights into Nucleophilic Substitution and Condensation
For the R&D director evaluating the technical feasibility of this process, understanding the mechanistic underpinnings is crucial for ensuring reproducibility and quality control. The synthesis begins with the activation of the hydroxyl group at the 8th position of the pleuromutilin scaffold using p-toluenesulfonyl chloride in the presence of a strong base, such as sodium hydroxide, maintaining a pH of 12-13. This step converts the hydroxyl into a good leaving group (tosylate), priming the molecule for subsequent nucleophilic attack. The reaction is conducted in organic solvents like ethyl acetate or dichloromethane, which provide a homogeneous medium for the initial dissolution while allowing for easy phase separation during workup. The careful control of temperature during this exothermic tosylation, typically kept between 20°C and 30°C after an initial ice bath, prevents side reactions such as elimination or rearrangement that could compromise the integrity of the complex polycyclic core. This precision in reaction parameter control is what allows the process to maintain high fidelity to the desired molecular structure throughout the synthesis.
Following the formation of the tosylated intermediate, the process advances to a critical nucleophilic substitution step involving diaminobenzenethiol. This reaction is facilitated by a cooled alkaline solution and enhanced by the addition of TEBAC, which acts as a phase transfer catalyst to shuttle the nucleophile into the organic phase where the substrate resides. The mixture is then heated to reflux at 75-85°C, providing the necessary activation energy to drive the substitution to completion within 2 to 3 hours. The final stage involves the condensation of the resulting Intermediate (I) with o-methylbenzoic acid derivatives using coupling agents like EDC/HOBt or HATU in the presence of organic bases such as N,N-diisopropylethylamine. This amide bond formation is executed under mild conditions (20-30°C) to preserve the stereochemistry of the chiral centers within the pleuromutilin framework. Purification is achieved through standard column chromatography using silica gel and a petroleum ether/ethyl acetate mobile phase, ensuring that the final product meets stringent purity specifications required for pharmaceutical applications.
How to Synthesize Pleuromutilin Compound Efficiently
The synthesis of this high-value pleuromutilin derivative is structured around a logical sequence of transformations that prioritize both yield and operational safety. The protocol begins with the preparation of p-toluenesulfonylated pleuromutilin, followed by the generation of Intermediate (I) through thiol substitution, and concludes with the final acylation to install the o-methylbenzoyl moiety. Each step has been optimized to minimize impurity formation and maximize throughput, making it suitable for translation from laboratory benchtop to pilot plant and eventually to full commercial scale. The detailed standardized synthetic steps, including specific molar ratios, solvent volumes, and workup procedures, are outlined below to guide process engineers in implementing this technology effectively.
- React pleuromutilin with p-toluenesulfonyl chloride in organic solvent under alkaline conditions (pH 12-13) at 20-30°C to form p-toluenesulfonylated pleuromutilin.
- Perform nucleophilic substitution by reacting the tosylated intermediate with diaminobenzenethiol in cooled alkaline solution, followed by reflux with benzyltriethylammonium chloride (TEBAC).
- Purify the resulting Intermediate (I) using column chromatography with petroleum ether and ethyl acetate to remove impurities before the final coupling step.
- Condense Intermediate (I) with o-methylbenzoic acid or o-methylbenzoyl chloride using EDC/HOBt or HATU coupling agents and organic bases at 20-30°C to yield the final compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis route offers profound advantages that directly address the pain points of procurement managers and supply chain executives. The primary benefit lies in the substantial cost optimization achieved through the use of inexpensive and readily available starting materials. Unlike processes that rely on precious metal catalysts or exotic reagents, this method utilizes commodity chemicals like p-toluenesulfonyl chloride and o-toluic acid, which are stable in supply and low in cost. Furthermore, the reaction conditions are remarkably gentle on infrastructure; the reaction liquid exhibits minimal corrosivity to equipment, which extends the service life of reactors and piping systems, thereby reducing capital expenditure on maintenance and replacement. The ability to recycle solvents such as ethyl acetate and dichloromethane further contributes to a lower environmental footprint and reduced raw material consumption, aligning with corporate sustainability goals while driving down the overall cost of production.
- Cost Reduction in Manufacturing: The economic viability of this process is underscored by its high yield and simplified operational requirements. By achieving yields over 79%, the process significantly reduces the amount of raw material needed per kilogram of final product, directly lowering the variable cost of goods. The elimination of harsh conditions and expensive catalysts removes the need for specialized containment systems or extensive downstream purification steps to remove trace metals. Additionally, the short production period and simple operation reduce labor costs and increase facility throughput, allowing manufacturers to produce larger batches in less time. These efficiencies combine to create a highly competitive cost structure that can withstand market fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the synthetic route and the availability of its inputs. Because the reagents are common industrial chemicals rather than bespoke synthons, the risk of supply disruption is minimized. The process tolerance for mild conditions (20-30°C) means that it can be executed in standard multipurpose reactors without requiring specialized cryogenic or high-pressure equipment, increasing the number of potential contract manufacturing organizations (CMOs) capable of producing the material. This flexibility ensures continuity of supply even in the face of unexpected demand surges or logistical challenges, providing a secure foundation for long-term product planning and inventory management.
- Scalability and Environmental Compliance: Scaling this process from grams to tons is facilitated by its inherent safety and environmental profile. The use of recyclable solvents and the generation of minimal hazardous waste simplify the permitting process and reduce the burden on waste treatment facilities. The low corrosion rate of the reaction mixture protects the integrity of large-scale stainless steel reactors, ensuring consistent performance over thousands of batches. Moreover, the alignment with green chemistry principles—such as atom economy and energy efficiency—makes this process attractive for companies seeking to reduce their carbon footprint. This environmental compliance not only mitigates regulatory risk but also enhances the brand value of the final pharmaceutical product in markets that prioritize sustainable sourcing.
Frequently Asked Questions (FAQ)
To assist stakeholders in making informed decisions regarding the adoption of this technology, we have compiled a set of frequently asked questions based on the technical disclosures and experimental data within the patent. These inquiries address critical aspects of the synthesis, including yield optimization, antibacterial efficacy, and scalability concerns. The answers provided are derived directly from the patent specifications and comparative examples, ensuring accuracy and relevance for technical evaluation.
Q: How does this new synthesis method improve upon traditional pleuromutilin modification routes?
A: Traditional methods often suffer from low yields (10-50%) and complex purification due to poor water solubility and short half-lives of derivatives. This patented route achieves yields over 79% by utilizing mild reaction conditions (20-30°C) and efficient phase transfer catalysis, significantly simplifying the operational workflow.
Q: What represents the key antibacterial advantage of this specific pleuromutilin derivative?
A: The compound exhibits potent activity against Gram-positive bacteria, including methicillin-resistant Staphylococcus aureus (MRSA), with an MIC value of 0.25 μg/mL. Its unique binding mechanism to the bacterial ribosome 50s subunit minimizes cross-resistance with existing antibiotic classes.
Q: Is the manufacturing process environmentally sustainable for large-scale production?
A: Yes, the process is designed for green chemistry compliance. It utilizes recyclable organic solvents like ethyl acetate and dichloromethane, operates at low temperatures to reduce energy consumption, and employs reagents that minimize equipment corrosion, thereby extending reactor lifespan and reducing waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pleuromutilin Compound Supplier
As the global demand for effective antibiotics continues to rise, partnering with a knowledgeable and capable chemical manufacturer is essential for bringing novel therapeutics to market. NINGBO INNO PHARMCHEM stands at the forefront of fine chemical synthesis, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our team of expert chemists is well-versed in the intricacies of pleuromutilin chemistry and is equipped to handle the specific challenges associated with this class of compounds, ensuring that every batch meets stringent purity specifications. With our rigorous QC labs and state-of-the-art analytical capabilities, we guarantee the quality and consistency of our intermediates, providing our partners with the confidence they need to advance their drug development programs.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthetic technology for their antibiotic pipelines. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your path to commercial success while optimizing your supply chain efficiency.
