Advanced Oxidative Dehydration Strategy for High-Purity Ceramide 3 and Amide Manufacturing
The landscape of amide bond formation, a cornerstone reaction in both pharmaceutical and cosmetic chemistry, is undergoing a significant transformation driven by the need for greener and more cost-effective methodologies. Patent CN110078635B, published in early 2022, introduces a groundbreaking approach that challenges the dominance of traditional peptide coupling agents. This intellectual property discloses a novel synthetic method wherein the dehydration reaction between carboxylic acids and amines is promoted by a unique combination of common quaternary ammonium salts—specifically derived from imidazole, thiazole, or triazole—and fluoride ions, all under oxidative conditions. This technology is particularly impactful for the production of high-value lipids such as Ceramide 3, a critical active ingredient in the global skincare market known for its exceptional moisture-retaining properties. By shifting away from expensive, stoichiometric coupling reagents towards catalytic promoters and benign oxidants, this patent offers a compelling roadmap for manufacturers seeking to optimize their supply chains while maintaining rigorous purity standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of amide bonds, especially in complex molecules like ceramides, has relied heavily on activated ester methods using reagents such as carbodiimides (e.g., DIC, EDC), phosphonium salts, or uronium salts (e.g., HOBt, HOAt). While effective, these conventional protocols suffer from severe economic and operational drawbacks that hinder large-scale efficiency. Firstly, these coupling agents are inherently expensive fine chemicals, often costing significantly more than the substrates themselves, which drastically inflates the Cost of Goods Sold (COGS) for bulk manufacturing. Secondly, these reactions typically generate stoichiometric amounts of difficult-to-remove byproducts, such as urea derivatives from carbodiimides, which necessitate complex and solvent-intensive purification steps like column chromatography or repeated recrystallization. Furthermore, many of these traditional reagents pose safety hazards, including potential explosivity (as seen with some azole additives) and sensitization issues, creating substantial regulatory and handling burdens for industrial facilities aiming for green chemistry compliance.
The Novel Approach
In stark contrast, the methodology outlined in CN110078635B presents a paradigm shift by utilizing readily available, commodity-grade chemicals to drive the dehydration process. Instead of pre-activating the carboxylic acid with costly reagents, this system employs a quaternary ammonium salt of imidazole, thiazole, or triazole alongside a fluoride source and an oxidant like potassium persulfate. This combination effectively activates the carboxylic acid in situ, facilitating nucleophilic attack by the amine under mild conditions. The most striking advantage is the operational simplicity; the reaction proceeds efficiently at room temperature, eliminating the energy costs associated with heating or cryogenic cooling often required by sensitive coupling agents. Moreover, the patent highlights that the degradation products of the quaternary salts can be recovered and reused, introducing a circular economy element that is virtually absent in traditional peptide synthesis. This approach not only simplifies the workflow but also aligns perfectly with modern sustainability goals by reducing chemical waste and energy consumption.
Mechanistic Insights into Oxidative Dehydration Catalysis
The core innovation of this technology lies in the synergistic interaction between the quaternary ammonium salt, the fluoride ion, and the oxidant. The quaternary salts, structurally represented in the patent as imidazolium, thiazolium, or triazolium species, act as potent promoters that likely facilitate the formation of an activated acyl intermediate. The presence of fluoride ions, such as those from potassium fluoride, plays a critical role in enhancing the nucleophilicity or stabilizing transition states, potentially through hydrogen bonding interactions or by modifying the solubility of the ionic species in organic solvents. The oxidant, typically a persulfate, serves to regenerate the active catalytic species or drive the equilibrium forward by consuming reduced byproducts, ensuring the reaction proceeds to completion without the need for excess driving forces. This mechanistic pathway avoids the formation of stable, non-volatile urea byproducts typical of carbodiimide chemistry, thereby streamlining the downstream processing.
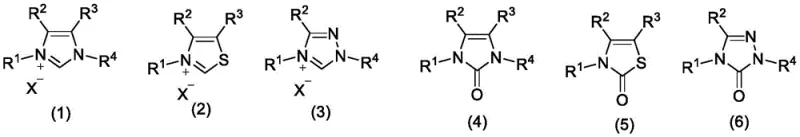
Understanding the structural versatility of the catalysts is crucial for R&D teams optimizing this process. As illustrated in the patent data, the quaternary ammonium salts can bear various alkyl, aryl, or heterocyclic substituents (R1-R4), allowing for fine-tuning of the electronic and steric properties to match specific substrate requirements. The degradation products, which are neutral imidazole, thiazole, or triazole derivatives, are not merely waste; the patent explicitly notes their potential for recovery and reduction back to the active quaternary form. This recyclability suggests a robust catalytic cycle that minimizes the accumulation of impurities in the reaction matrix. For the synthesis of sensitive molecules like phytosphingosine derivatives, this mild yet effective activation mechanism prevents racemization or decomposition of chiral centers, a common pitfall in harsh acidic or basic amidation conditions. The ability to control the impurity profile through catalyst selection makes this method particularly attractive for producing high-purity cosmetic and pharmaceutical intermediates.
How to Synthesize Ceramide 3 Efficiently
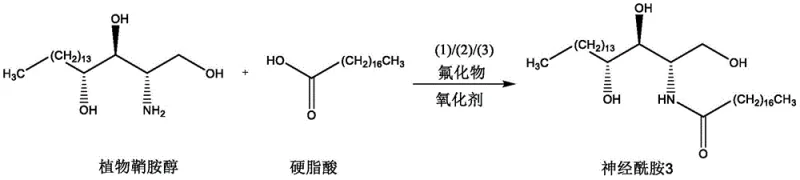
The practical application of this technology is best exemplified by the synthesis of Ceramide 3, a flagship molecule in the dermocosmetic industry. The protocol involves a straightforward one-pot procedure where phytosphingosine and stearic acid are combined with the quaternary salt promoter, fluoride, and oxidant in a solvent like t-butyl methyl ether. The reaction mixture is stirred at ambient temperature for an extended period, typically around 36 hours, allowing the slow but steady formation of the amide bond. Following the reaction, the workup is remarkably simple, involving basic aqueous extraction to remove inorganic salts and water-soluble byproducts, followed by drying and solvent removal. The crude product can then be purified via standard silica gel chromatography to yield the target ceramide with high purity. For detailed standardized operating procedures and specific molar ratios optimized for your facility, please refer to the technical guide below.
- Charge a reaction vessel with solvent (e.g., t-butyl methyl ether), carboxylic acid, quaternary ammonium salt (imidazole/thiazole/triazole), fluoride, amine, and oxidant.
- Stir the reaction mixture at room temperature for approximately 36 hours to allow the oxidative dehydration to proceed.
- Perform aqueous workup by extracting with water and ethyl acetate, dry the organic layer, remove solvent, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN110078635B offers tangible strategic benefits that extend beyond mere chemical curiosity. The primary driver for adoption is the drastic reduction in raw material costs. By replacing expensive, specialized coupling agents like HATU or PyBOP with commodity chemicals such as potassium fluoride and sodium persulfate, manufacturers can significantly lower the direct material input costs per kilogram of product. This shift transforms the cost structure of amide synthesis from a high-value fine chemical operation to a more commoditized process, improving margin potential in competitive markets like personal care. Furthermore, the reliance on widely available, non-hazardous reagents mitigates supply chain risks associated with the sourcing of niche reagents that may be subject to geopolitical shortages or strict transportation regulations.
- Cost Reduction in Manufacturing: The elimination of stoichiometric amounts of expensive coupling reagents represents a major opportunity for cost optimization. Traditional methods often require 1.2 to 1.5 equivalents of costly activators, whereas this oxidative system utilizes cheaper promoters that can potentially be recycled. Additionally, the simplified workup procedure reduces the consumption of solvents and silica gel during purification, leading to substantial savings in waste disposal and utility costs. The ability to run reactions at room temperature further contributes to energy efficiency, removing the need for expensive heating or cooling infrastructure and lowering the overall carbon footprint of the manufacturing site.
- Enhanced Supply Chain Reliability: The reagents specified in this patent, including stearic acid, phytosphingosine, and common inorganic salts, are globally sourced commodities with stable supply lines. Unlike specialized peptide coupling agents that may have limited suppliers and long lead times, the inputs for this process are readily available from multiple chemical distributors worldwide. This diversification of the supply base ensures business continuity and protects against price volatility. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, reducing the rate of batch failures and ensuring consistent delivery schedules to downstream customers.
- Scalability and Environmental Compliance: From a scale-up perspective, the exothermic profile of this reaction is manageable, and the absence of hazardous gas evolution (common in some activation methods) simplifies reactor design and safety protocols. The use of t-butyl methyl ether and water for extraction is a greener alternative to chlorinated solvents often used in traditional amide synthesis, facilitating easier compliance with increasingly stringent environmental regulations. The potential for recycling the catalyst degradation products adds another layer of sustainability, appealing to eco-conscious brands and helping manufacturers meet corporate social responsibility (CSR) targets regarding waste reduction and resource efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative dehydration technology. These insights are derived directly from the experimental data and claims within patent CN110078635B, providing a factual basis for evaluating its fit within your current manufacturing portfolio. Understanding the nuances of catalyst loading, solvent selection, and substrate scope is essential for successful technology transfer.
Q: What are the advantages of using quaternary ammonium salts over traditional coupling agents?
A: Traditional coupling agents like DIC or HOBt are often expensive and generate significant stoichiometric waste. The method in CN110078635B utilizes cheap, common quaternary salts and fluorides, significantly reducing raw material costs and simplifying the purification process.
Q: Can the catalyst degradation products be reused?
A: Yes, the patent explicitly states that the degradation products of the imidazole, thiazole, or triazole quaternary ammonium salts can be recovered and reused through reduction, enhancing the overall atom economy and sustainability of the process.
Q: Is this method suitable for large-scale Ceramide 3 production?
A: Absolutely. The process operates at room temperature, uses inexpensive oxidants like sodium persulfate, and involves a simple extraction workup. These factors make it highly scalable and suitable for industrial manufacturing of cosmetic ingredients like Ceramide 3.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ceramide 3 Supplier
As the global demand for bio-active lipids continues to surge, partnering with a technically proficient manufacturer is critical for securing a competitive edge. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced synthetic methodologies like the oxidative dehydration process to deliver superior quality Ceramide 3 and related amide intermediates. Our R&D team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the highest international standards for cosmetic and pharmaceutical applications.
We invite you to explore how our innovative manufacturing capabilities can drive value for your organization. By adopting these greener, more efficient synthetic routes, we can offer you a Customized Cost-Saving Analysis tailored to your specific volume needs. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples. Let us collaborate to build a resilient, cost-effective, and sustainable supply chain for your next generation of high-performance skincare formulations.
