Advanced Metal-Free Synthesis of Sulfonylcoumarins for Pharmaceutical Intermediates
Advanced Metal-Free Synthesis of Sulfonylcoumarins for Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign methodologies for constructing complex heterocyclic scaffolds. Patent CN114853721A introduces a groundbreaking synthetic strategy for sulfonylcoumarin compounds, a class of bioisosteres with significant therapeutic potential in treating conditions such as glaucoma, epilepsy, and hypertension. This innovation addresses critical bottlenecks in existing manufacturing processes by utilizing a mild, metal-free catalytic system that leverages beta-ketosulfonyl fluorides as key building blocks. By shifting away from hazardous reagents and extreme conditions, this technology offers a compelling value proposition for reliable pharmaceutical intermediate supplier networks aiming to enhance process safety and efficiency. The following analysis details the mechanistic advantages and commercial implications of this novel approach.
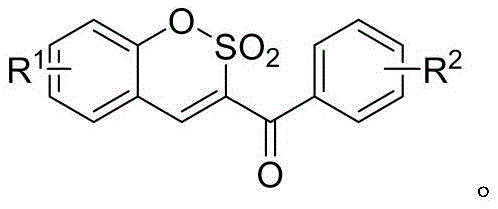
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of sulfonylcoumarin derivatives has relied heavily on methodologies that pose significant operational and environmental challenges. Traditional routes often involve the heating of 2-hydroxyaryl aldehydes with active sulfonyl chlorides in the presence of pyridine, a process that frequently requires harsh thermal conditions and generates stoichiometric amounts of corrosive byproducts. Alternatively, strategies employing Baylis-Hillman reactions necessitate the use of strong organic bases like DBU or toxic catalysts, which complicate downstream purification and increase the burden on waste management systems. These conventional pathways are further characterized by excessively long reaction times and poor atom economy, leading to inflated production costs and extended lead times for high-purity pharmaceutical intermediates. The reliance on moisture-sensitive reagents and the generation of acidic waste streams create substantial barriers to sustainable commercial scale-up of complex polymer additives and drug candidates alike.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a remarkably simple yet effective protocol centered on the reactivity of beta-ketosulfonyl fluorides. As illustrated by the reactant structure below, these fluorinated precursors enable a smooth cyclization under mild conditions without the need for transition metal catalysts or aggressive Lewis acids. 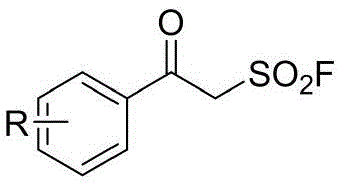 The process operates efficiently in a polar aprotic solvent system like DMF at moderate temperatures around 40°C, drastically reducing energy consumption compared to refluxing conditions. This novel approach not only simplifies the operational workflow by eliminating the need for inert atmosphere techniques often required for sensitive organometallic reagents but also ensures a cleaner reaction profile. The use of inexpensive inorganic bases like potassium fluoride further enhances the economic viability, making this a superior choice for cost reduction in electronic chemical manufacturing and pharmaceutical sectors where margin pressure is high.
The process operates efficiently in a polar aprotic solvent system like DMF at moderate temperatures around 40°C, drastically reducing energy consumption compared to refluxing conditions. This novel approach not only simplifies the operational workflow by eliminating the need for inert atmosphere techniques often required for sensitive organometallic reagents but also ensures a cleaner reaction profile. The use of inexpensive inorganic bases like potassium fluoride further enhances the economic viability, making this a superior choice for cost reduction in electronic chemical manufacturing and pharmaceutical sectors where margin pressure is high.
Mechanistic Insights into Fluorine-Sulfur Exchange Cyclization
The core of this synthetic breakthrough lies in the unique reactivity of the sulfur-fluorine bond within the beta-ketosulfonyl fluoride motif. Upon exposure to fluoride ions in a polar medium, the substrate undergoes a nucleophilic activation that facilitates an intramolecular cyclization with the aromatic coupling partner. This mechanism avoids the formation of unstable intermediates common in sulfonyl chloride chemistry, thereby minimizing side reactions such as hydrolysis or polymerization. The reaction proceeds through a concerted pathway where the fluoride acts as both a base and a leaving group promoter, ensuring high conversion rates even with sterically hindered substrates. This mechanistic elegance allows for the precise control of regioselectivity, which is paramount for maintaining the integrity of the bioactive pharmacophore in the final sulfonylcoumarin derivative.
Furthermore, the tolerance of this system towards various functional groups on the aromatic ring is a testament to its mild nature. Whether the aromatic partner bears electron-donating groups like methoxy or bulky substituents like tert-butyl, the cyclization proceeds with consistent efficiency. This broad substrate scope is critical for medicinal chemists exploring structure-activity relationships, as it allows for the rapid generation of diverse libraries without redesigning the synthetic route for each analog. The absence of heavy metal residues in the final product is another significant advantage, as it alleviates the need for costly scavenging steps to meet stringent regulatory limits on elemental impurities in active pharmaceutical ingredients. Consequently, this method represents a paradigm shift towards greener, more efficient heterocycle synthesis.
How to Synthesize Sulfonylcoumarin Efficiently
The practical implementation of this synthesis is straightforward and amenable to standard laboratory and pilot plant equipment. The process begins with the preparation of a homogeneous mixture of potassium fluoride in dry DMF, followed by the controlled addition of the beta-ketosulfonyl fluoride and the specific aromatic precursor. Reaction monitoring is easily achieved via thin-layer chromatography, allowing operators to determine the optimal endpoint without complex analytical instrumentation. For a comprehensive understanding of the specific molar ratios, temperature profiles, and workup procedures detailed in the patent examples, please refer to the standardized protocol below.
- Charge a reaction vessel with dry DMF and add potassium fluoride, followed by the sequential addition of beta-ketosulfonyl fluoride and the aromatic coupling partner.
- Heat the reaction mixture to approximately 40°C and monitor progress via TLC until the starting materials are fully consumed.
- Quench the reaction with deionized water, extract the organic phase with ethyl acetate, dry over anhydrous sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this synthetic route offers transformative benefits that directly impact the bottom line and supply chain resilience. By replacing expensive and hazardous catalysts with commodity chemicals like potassium fluoride, manufacturers can achieve substantial cost savings in raw material procurement. The simplified workup procedure, which involves a basic aqueous quench and extraction, reduces the consumption of specialized solvents and minimizes the volume of hazardous waste requiring disposal. These factors collectively contribute to a leaner manufacturing process that is less susceptible to fluctuations in the pricing of specialty reagents, thereby stabilizing the cost structure for long-term supply agreements.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and toxic organic bases removes the necessity for expensive purification steps such as metal scavenging or complex distillation sequences. This streamlining of the downstream processing significantly lowers the overall cost of goods sold, allowing for more competitive pricing in the global market for high-purity OLED material and pharmaceutical intermediates. Additionally, the mild reaction conditions reduce energy overheads associated with heating and cooling cycles, further enhancing the economic efficiency of the production facility.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as beta-ketosulfonyl fluorides and simple aromatic compounds mitigates the risk of supply disruptions often associated with proprietary or single-source reagents. Since the reaction does not require sensitive organometallic species that demand cold chain logistics or strict moisture control during transport, the inbound supply chain becomes more robust and flexible. This reliability ensures consistent delivery schedules and reduces the likelihood of production stoppages due to raw material shortages, a critical factor for maintaining continuity in the manufacture of vital therapeutic agents.
- Scalability and Environmental Compliance: The inherent safety of operating at low temperatures with non-pyrophoric reagents makes this process highly scalable from kilogram to multi-ton quantities without significant engineering hurdles. The reduced generation of corrosive waste streams aligns with increasingly stringent environmental regulations, lowering the compliance burden and potential liability for chemical manufacturers. This eco-friendly profile not only facilitates smoother regulatory approvals but also enhances the brand reputation of suppliers committed to sustainable chemistry practices, appealing to environmentally conscious partners in the fine chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this sulfonylcoumarin synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the method's capabilities and limitations. Understanding these aspects is essential for technical teams evaluating the feasibility of adopting this route for their specific project requirements.
Q: What are the primary advantages of this sulfonylcoumarin synthesis method over traditional routes?
A: This method eliminates the need for toxic, corrosive catalysts and harsh conditions typically required in conventional Baylis-Hillman or sulfonyl chloride reactions, operating instead under mild temperatures with simple workup procedures.
Q: Is this synthetic route suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes readily available reagents like potassium fluoride and operates at low temperatures (40°C), which significantly reduces energy consumption and safety risks associated with exothermic reactions, making it highly scalable.
Q: What is the substrate scope for the aromatic coupling partners in this reaction?
A: The methodology demonstrates broad compatibility with various substituted aromatic compounds, including those with methyl, methoxy, and tert-butyl groups, allowing for the diversification of the final sulfonylcoumarin library.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfonylcoumarin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthetic route in advancing the production of high-value heterocyclic intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of sulfonylcoumarin delivered meets the exacting standards required for pharmaceutical and agrochemical applications. Our commitment to quality and consistency makes us the preferred partner for organizations seeking to secure their supply chain for these critical building blocks.
We invite you to collaborate with us to leverage this innovative technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can drive value and efficiency in your supply chain. Let us help you navigate the complexities of chemical sourcing with confidence and precision.
