Advanced Manufacturing of Kinase Inhibitor Intermediates via Safe Catalytic Hydrogenation and SNAr Coupling
Advanced Manufacturing of Kinase Inhibitor Intermediates via Safe Catalytic Hydrogenation and SNAr Coupling
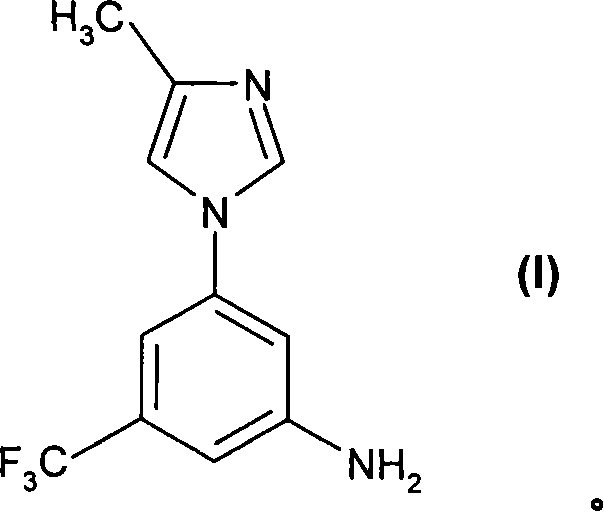
The pharmaceutical industry continuously seeks robust synthetic pathways for complex heterocyclic intermediates, particularly those serving as key building blocks for oncology therapeutics. Patent CN101193866A introduces a transformative methodology for the preparation of 5-(4-methyl-1H-imidazol-1-yl)-3-(trifluoromethyl)-aniline, designated as Formula (I). This compound acts as a critical precursor for substituted pyrimidinylaminobenzamides, which are potent inhibitors of tyrosine kinases such as c-Abl and Bcr-Abl, essential targets in leukemia treatment. The disclosed innovation addresses longstanding inefficiencies in prior art by replacing hazardous reagents and energy-intensive purification steps with a streamlined, scalable catalytic process. By leveraging nucleophilic aromatic substitution followed by catalytic hydrogenation, this technology offers a compelling value proposition for reliable pharmaceutical intermediate supplier networks aiming to enhance production safety and consistency.
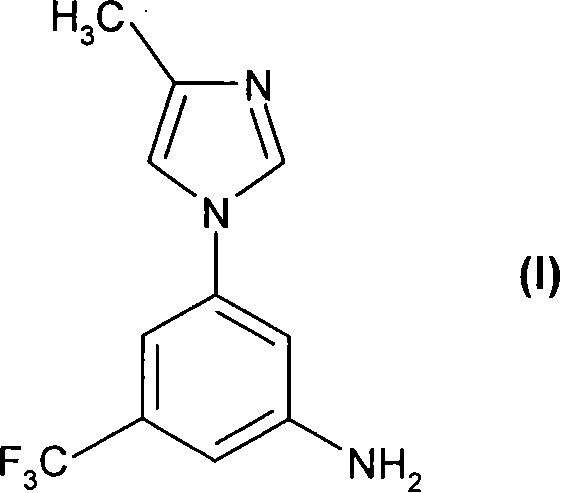
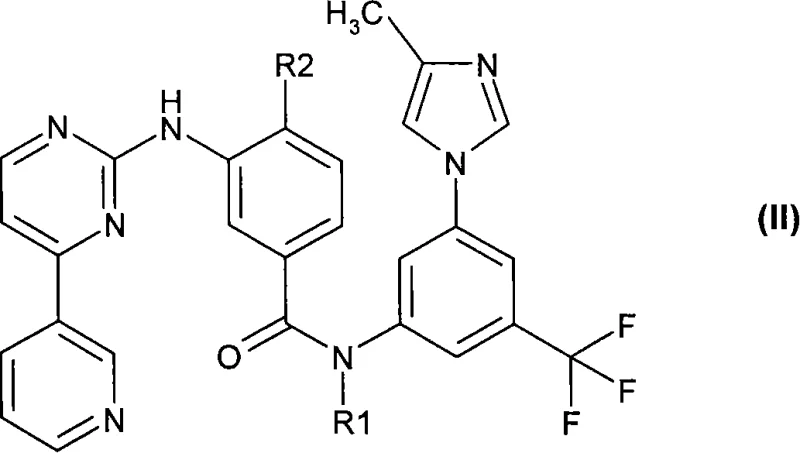
Furthermore, the strategic importance of Formula (I) extends to its role in generating Formula (II) compounds, which have demonstrated efficacy against receptor tyrosine kinases including PDGF-R, Flt3, VEGF-R, and c-Kit. The ability to manufacture this core scaffold efficiently directly impacts the supply chain continuity for downstream API production. As global demand for targeted cancer therapies rises, the adoption of this optimized synthetic route ensures that manufacturers can meet rigorous quality standards while mitigating the environmental and safety liabilities associated with older methodologies. This report analyzes the technical merits and commercial implications of this patent for R&D and procurement stakeholders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
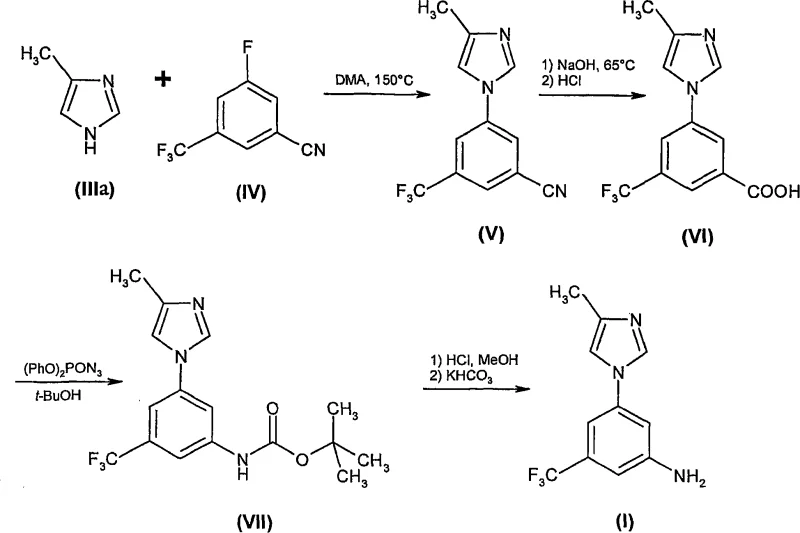
Prior to this invention, the synthesis of Formula (I) relied on a cumbersome four-step pathway that presented significant operational hazards and cost inefficiencies. The conventional route initiated with an aromatic substitution reaction requiring extreme thermal energy, typically around 150°C, to couple 4-methyl-1H-imidazole with a fluorinated benzene derivative. More critically, the subsequent transformation involved a Curtius rearrangement utilizing diphenylphosphoryl azide, a reagent known for its instability and toxicity. This step not only posed severe safety risks to plant personnel but also generated difficult-to-remove diphenylphosphate by-products, complicating waste management protocols.
Additionally, the legacy process suffered from inconsistent product yields and quality profiles, necessitating purification via column chromatography. In a commercial manufacturing context, chromatography is economically prohibitive due to high solvent consumption, low throughput, and significant downtime for column packing and regeneration. These factors collectively resulted in elevated production costs and extended lead times, making the conventional method unsuitable for large-scale commercial scale-up of complex pharmaceutical intermediates. The reliance on such inefficient techniques created a bottleneck in the supply chain, limiting the availability of high-purity materials needed for clinical and commercial drug development.
The Novel Approach
The patented methodology revolutionizes this landscape by condensing the synthesis into a highly efficient two-step sequence that eliminates the need for hazardous azides and chromatographic purification. The new approach begins with a base-mediated nucleophilic aromatic substitution between dinitrobenzotrifluoride and 4-methyl-1H-imidazole, conducted in polar aprotic solvents like DMA at moderate temperatures ranging from 90°C to 140°C. This coupling reaction is followed directly by a catalytic hydrogenation step using standard heterogeneous catalysts such as Pd/C or Raney Nickel. This strategic shift replaces the dangerous Curtius rearrangement with a safe, atom-economical reduction, fundamentally altering the risk profile of the manufacturing process.
Moreover, the novel route incorporates a sophisticated purification strategy based on salt formation and crystallization rather than chromatography. By converting the crude amine into a hydrochloride salt, impurities and isomers are effectively separated through filtration, allowing for the isolation of the free base with purity exceeding 99%. This modification drastically simplifies the downstream processing unit operations, reducing solvent usage and cycle times. For procurement managers, this translates to a more predictable supply of cost reduction in API manufacturing, as the elimination of chromatography and hazardous reagents lowers both raw material and waste disposal expenditures significantly.
Mechanistic Insights into Nucleophilic Aromatic Substitution and Catalytic Hydrogenation
The core of this synthetic innovation lies in the optimization of the nucleophilic aromatic substitution (SNAr) mechanism in Step A. The reaction utilizes the strong electron-withdrawing nature of the nitro and trifluoromethyl groups on the benzene ring to activate the fluorine leaving group for displacement by the imidazole nitrogen. The patent highlights that the choice of base and solvent is critical; bases such as potassium carbonate or potassium phosphate in DMA facilitate rapid decomposition of the starting materials while minimizing side reactions. Careful control of temperature and heating cycles is essential, as the reaction mixture can be unstable; faster heating and cooling profiles or continuous flow equipment are recommended to suppress decomposition and ensure a cleaner reaction profile with reduced isomer formation.
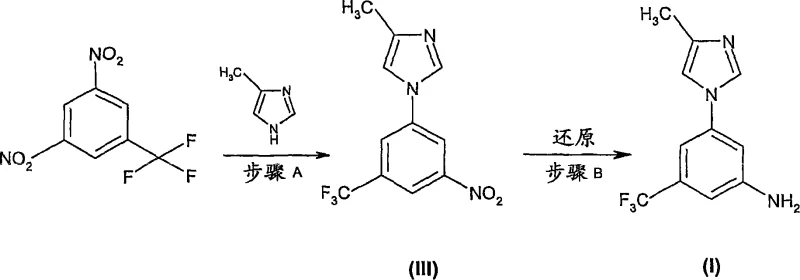
Following the coupling, Step B employs heterogeneous catalysis to reduce the nitro group to an amine. The mechanism involves the adsorption of hydrogen gas and the nitro-imidazole intermediate onto the metal surface of the catalyst (Pd or Ni), where sequential hydrogenation occurs. The patent specifies that this reduction can be performed at moderate pressures of 1-20 bar, preferably 5-10 bar, ensuring high conversion rates without compromising safety. The use of hydrogen transfer agents like formic acid is also noted as an alternative, providing flexibility in reactor setup. This mechanistic understanding allows R&D directors to fine-tune reaction parameters, such as catalyst loading (0.1-20 wt%) and solvent systems (methanol), to maximize yield and minimize the presence of the 1:9 error isomer mentioned in the examples.
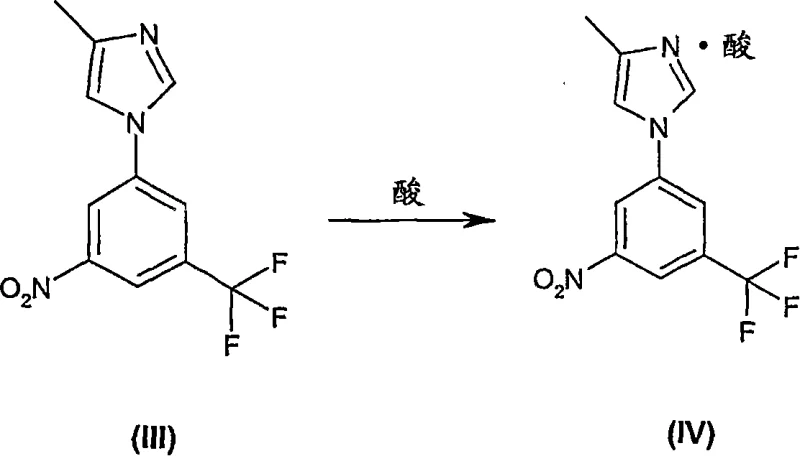
Crucially, the purification mechanism leverages the differential solubility of the amine salt versus neutral impurities. By treating the crude reaction mixture with hydrochloric acid in a solvent system like methanol and acetone, the desired aniline precipitates as a stable hydrochloride salt. This solid-state separation is far more efficient than liquid-liquid extraction or chromatography for removing non-basic organic impurities. Subsequent treatment with a base like sodium bicarbonate regenerates the free amine, which is then crystallized to final specifications. This robust purification loop ensures that the final product meets stringent purity specifications required for GMP manufacturing, validating the process's suitability for producing high-purity pharmaceutical intermediates.
How to Synthesize 5-(4-Methyl-1H-Imidazol-1-Yl)-3-(Trifluoromethyl)-Aniline Efficiently
To implement this synthesis effectively, manufacturers must adhere to precise operational parameters regarding reagent stoichiometry and thermal management. The process begins with the careful addition of dinitrobenzotrifluoride and base to the solvent, followed by the controlled introduction of the imidazole component to manage exotherms. The subsequent hydrogenation requires rigorous inertization and pressure control to ensure complete reduction while maintaining catalyst activity. For a comprehensive guide on the specific weights, temperatures, and workup procedures detailed in the patent examples, please refer to the standardized synthesis protocol below.
- Perform nucleophilic aromatic substitution between dinitrobenzotrifluoride and 4-methyl-1H-imidazole using potassium carbonate in DMA solvent at 90-140°C.
- Reduce the resulting nitro-imidazole intermediate using hydrogen gas and a palladium or Raney nickel catalyst in methanol at 4-7.5 bar pressure.
- Purify the final aniline product via hydrochloride salt formation, filtration, and subsequent free-basing crystallization to achieve >99% purity without chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
The transition to this novel synthetic route offers profound commercial advantages that extend beyond mere technical feasibility, directly addressing key pain points in pharmaceutical supply chains. By eliminating the requirement for diphenylphosphoryl azide, the process removes a significant safety hazard and the associated regulatory burdens of handling toxic azides. This change simplifies the environmental health and safety (EHS) compliance landscape, reducing the need for specialized containment equipment and hazardous waste disposal services. Consequently, the overall operational expenditure is lowered, not through arbitrary percentage claims, but through the tangible removal of costly safety infrastructure and waste treatment protocols.
- Cost Reduction in Manufacturing: The most significant economic driver is the complete removal of column chromatography from the purification workflow. Chromatography is inherently batch-limited, solvent-intensive, and labor-heavy; replacing it with crystallization and filtration allows for continuous or larger-batch processing with significantly lower solvent recovery costs. Furthermore, the use of commodity chemicals like potassium carbonate and Raney Nickel instead of specialized phosphorous reagents reduces raw material procurement costs. This structural change in the bill of materials ensures a more competitive pricing structure for the final intermediate without compromising quality.
- Enhanced Supply Chain Reliability: The robustness of the hydrogenation step and the crystallization purification enhances the predictability of production schedules. Unlike the old method, which suffered from inconsistent yields and difficult by-product removal, this new route delivers consistent purity levels greater than 99%. Reliable output quality minimizes the risk of batch failures and reprocessing, which are major causes of supply delays. For supply chain heads, this means reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream API synthesis lines remain operational without interruption due to raw material shortages.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations like stirred tank reactors and filter dryers that are common in multi-purpose chemical plants. The avoidance of azides and the reduction in solvent volume per kilogram of product align with green chemistry principles, lowering the facility's environmental footprint. This alignment facilitates easier permitting and regulatory approval for commercial scale-up of complex pharmaceutical intermediates, allowing manufacturers to ramp up production volumes rapidly to meet market demand for oncology drugs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and claims within CN101193866A, providing clarity on safety, catalyst selection, and purification efficiency. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this process for their own manufacturing portfolios.
Q: How does this new synthesis route improve safety compared to previous methods?
A: The novel process eliminates the use of diphenylphosphoryl azide, a hazardous reagent required in the traditional Curtius rearrangement pathway, thereby significantly reducing operational risks and waste disposal costs associated with toxic by-products.
Q: What catalysts are suitable for the reduction step in this manufacturing process?
A: The patent specifies that both supported Group VIII transition metals like Palladium on Carbon (Pd/C) and Raney Nickel are effective for the hydrogenation step, allowing manufacturers to optimize based on catalyst availability and cost structures.
Q: How is high purity achieved without using column chromatography?
A: High purity (>99%) is attained through a robust crystallization protocol involving the formation of a hydrochloride salt intermediate, followed by filtration and regeneration of the free base, which effectively removes organic impurities and isomers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-(4-Methyl-1H-Imidazol-1-Yl)-3-(Trifluoromethyl)-Aniline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving oncology therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and compliant. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 5-(4-methyl-1H-imidazol-1-yl)-3-(trifluoromethyl)-aniline meets the exacting standards required by global pharmaceutical regulators.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this safer, chromatography-free method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project timelines are met with the highest level of quality and reliability.
