Optimizing Palmatine Production: A Technical Analysis of High-Yield Synthetic Routes
Introduction to Advanced Palmatine Synthesis
The global demand for effective antibacterial and anti-inflammatory agents continues to drive innovation in pharmaceutical manufacturing, particularly for established alkaloids like Palmatine, also known commercially as Fibrauretin. Traditionally sourced from natural plant extracts such as Fibraurea recisa, the supply chain for this active ingredient has faced significant bottlenecks due to resource exhaustion and long growth cycles of the raw botanical material. Addressing these critical supply constraints, patent CN102532130A introduces a robust total chemical synthesis method that fundamentally restructures the production landscape. This novel approach replaces the erratic availability of natural extraction with a predictable, high-yield chemical process, utilizing readily available aromatic amines and aldehydes as starting materials. By shifting the production paradigm from agriculture-dependent extraction to precision chemical synthesis, manufacturers can secure a stable supply of high-purity Palmatine, ensuring continuity for downstream pharmaceutical formulations and reducing reliance on fluctuating natural markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this technological breakthrough, the synthetic landscape for Palmatine was dominated by cumbersome multi-step pathways that were economically and operationally inefficient. As documented in prior art such as CN 1687064A, traditional methods often required upwards of ten distinct reaction steps, beginning with basic feedstocks like methyl catechol and proceeding through methylation, chloromethylation, and cyanation. These legacy processes were plagued by harsh reaction conditions, requiring rigorous equipment specifications and complex operational protocols that increased the risk of safety incidents. Furthermore, the cumulative yield of these lengthy sequences was dismally low, often hovering around 20%, which rendered the final product prohibitively expensive for large-scale applications. The necessity for multiple purification stages between each step not only consumed vast quantities of solvents but also extended production lead times, making it difficult to respond agilely to market demands for this critical therapeutic agent.
The Novel Approach
In stark contrast to the convoluted legacy pathways, the methodology disclosed in CN102532130A achieves the target molecule through a highly efficient three-step sequence that maximizes atom economy and operational simplicity. The process initiates with a direct thermal condensation between 3,4-dimethoxyphenethylamine and 2,3-dimethoxybenzaldehyde, followed immediately by a reduction and a final catalytic cyclization. This streamlined architecture eliminates the need for protecting group strategies and complex intermediate isolations that characterize older routes. The strategic design allows for the telescoping of the first two steps, where the crude imine intermediate proceeds directly to reduction without purification, drastically cutting down processing time and waste generation. With a reported total recovery rate exceeding 85%, this route represents a quantum leap in manufacturing efficiency, offering a viable pathway for the cost reduction in API manufacturing that procurement teams have long sought.
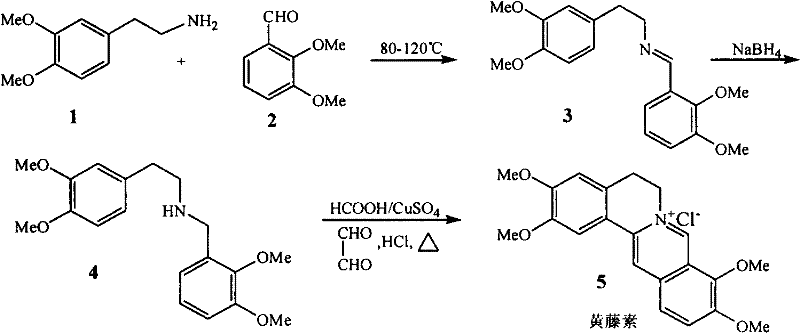
Mechanistic Insights into Formic Acid/Copper Sulfate Catalyzed Cyclization
The cornerstone of this synthetic route lies in the final cyclization step, which constructs the complex isoquinoline skeleton characteristic of the Palmatine structure. This transformation involves the reaction of the secondary amine intermediate with glyoxal (oxalic dialdehyde) in the presence of a specific catalytic system comprising formic acid and copper sulfate. The mechanism likely proceeds via a Pictet-Spengler-type condensation where the amine reacts with the dialdehyde to form an iminium ion, which subsequently undergoes electrophilic aromatic substitution to close the ring. The inclusion of copper sulfate acts as a Lewis acid catalyst, activating the carbonyl species and facilitating the dehydration steps necessary for aromatization. This catalytic synergy allows the reaction to proceed under relatively mild boiling water bath conditions, avoiding the extreme temperatures or strong acidic environments that often degrade sensitive organic molecules in traditional syntheses.
Furthermore, the control of impurities in this process is inherently superior due to the high selectivity of the condensation and reduction steps. The initial formation of the Schiff base (imine) is highly specific, minimizing the formation of side products that typically arise from non-selective alkylations in older methods. The subsequent reduction with sodium borohydride is a clean transformation that selectively targets the carbon-nitrogen double bond without affecting the methoxy groups on the aromatic rings. In the final stage, the use of concentrated hydrochloric acid serves a dual purpose: it facilitates the cyclization and simultaneously forms the stable hydrochloride salt of the product, which precipitates out of the solution as high-purity yellow needle-like crystals. This crystallization behavior acts as a built-in purification mechanism, ensuring that the final API meets stringent purity specifications without the need for extensive chromatographic separation.
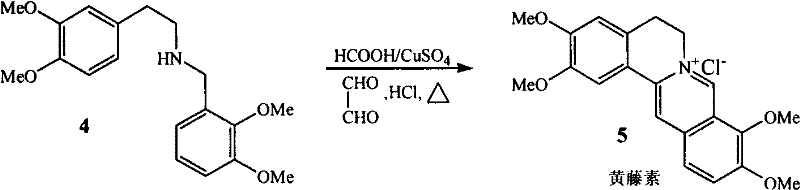
How to Synthesize Palmatine Chloride Efficiently
Executing this synthesis requires precise control over reaction parameters to maintain the high yields reported in the patent literature. The process begins with the careful heating of the amine and aldehyde mixture to between 80°C and 120°C to drive the condensation to completion, followed by a controlled addition of the reducing agent to manage exothermicity. The final cyclization step demands strict adherence to the molar ratios of the catalyst system to ensure complete ring closure. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below which outlines the critical process controls.
- Condense 3,4-dimethoxyphenethylamine with 2,3-dimethoxybenzaldehyde at 80-120°C to form the imine intermediate.
- Reduce the crude imine using sodium borohydride in methanol to obtain the secondary amine without intermediate purification.
- Perform catalytic cyclization with glyoxal, formic acid, and copper sulfate under reflux to yield Palmatine Chloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this three-step synthesis offers transformative benefits that extend far beyond simple yield improvements. The reduction from a ten-step process to a three-step process fundamentally alters the cost structure of the molecule by drastically reducing labor hours, energy consumption, and solvent usage. The ability to telescope the first two steps without intermediate purification is a significant logistical advantage, as it removes an entire unit operation from the production schedule, thereby increasing throughput capacity without additional capital investment. This efficiency translates directly into a more competitive pricing structure, allowing buyers to secure high-purity Palmatine at a fraction of the cost associated with natural extraction or legacy synthetic methods.
- Cost Reduction in Manufacturing: The elimination of complex intermediate isolation and purification steps results in substantial savings on solvents and consumables. By avoiding the use of expensive transition metal catalysts often required in cross-coupling reactions found in other routes, and instead utilizing inexpensive formic acid and copper sulfate, the raw material cost profile is significantly optimized. The high overall yield means that less starting material is wasted, further driving down the cost per kilogram of the final active ingredient.
- Enhanced Supply Chain Reliability: Dependence on agricultural sources for natural alkaloids introduces volatility due to weather patterns and harvest cycles; this synthetic route decouples production from these environmental variables. The starting materials, 3,4-dimethoxyphenethylamine and 2,3-dimethoxybenzaldehyde, are commodity chemicals available from multiple global suppliers, ensuring that production is never halted due to a single source failure. This diversification of the supply base guarantees consistent delivery schedules and protects downstream manufacturers from raw material shortages.
- Scalability and Environmental Compliance: The reaction conditions described, such as refluxing in methanol and boiling water baths, are easily scalable from pilot plants to multi-ton commercial reactors without requiring specialized high-pressure equipment. The simplified waste stream, resulting from fewer reaction steps and the absence of heavy metal contaminants like cyanide used in older methods, simplifies effluent treatment and ensures compliance with increasingly strict environmental regulations. This ease of scale-up facilitates the rapid expansion of production capacity to meet surging market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for technical teams evaluating this technology for adoption.
Q: What are the key advantages of this new Palmatine synthesis route?
A: The new route reduces the synthesis from 10 steps to just 3 steps, significantly increasing total yield from approximately 20% to over 85% while eliminating harsh reaction conditions.
Q: Is purification required between the first two reaction steps?
A: No, the process allows for telescoping; the crude imine product from the first step can be directly reduced in the second step without isolation, saving time and solvents.
Q: What catalysts are used in the final cyclization step?
A: The final ring-closure reaction utilizes a catalytic system comprising formic acid and copper sulfate to facilitate the formation of the isoquinoline core.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Palmatine Supplier
As the pharmaceutical industry increasingly prioritizes sustainable and efficient manufacturing pathways, NINGBO INNO PHARMCHEM stands at the forefront of implementing these advanced synthetic technologies. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and robust. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of Palmatine Chloride we produce meets the highest international standards for safety and efficacy required by regulatory bodies worldwide.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to our synthetically derived Palmatine. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a partnership built on transparency, quality, and mutual growth.
