Advanced Synthesis of Perfluoroalkylsulfonyl Isoflavones for Anticancer Drug Development
Advanced Synthesis of Perfluoroalkylsulfonyl Isoflavones for Anticancer Drug Development
The pharmaceutical industry is constantly seeking novel scaffolds that can overcome drug resistance and improve therapeutic indices in oncology. Patent CN101003530B introduces a groundbreaking methodology for the preparation of perfluoroalkylsulfonyl isoflavones, a class of compounds exhibiting profound anticancer properties. This technology addresses the historical challenge of effectively incorporating fluorine-containing groups into the isoflavone backbone, a modification known to enhance metabolic stability and membrane permeability. By utilizing perfluoroalkylsulfonyl fluorides as key reagents, the disclosed process offers a streamlined route to high-value intermediates that serve as potent inhibitors of human leukemia cells. For R&D directors and procurement specialists, understanding the nuances of this synthesis is critical for securing a reliable supply of next-generation anticancer leads.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the modification of isoflavones to include fluorine atoms has been fraught with synthetic difficulties and suboptimal outcomes. Traditional approaches often relied on the introduction of trifluoromethyl or difluoromethyl groups, which required harsh reaction conditions and complex multi-step sequences that were difficult to scale. Furthermore, previous studies indicated that while these modifications altered physicochemical properties, they did not significantly enhance the anticancer activity or solubility profiles necessary for effective drug development. The low absorption of native isoflavones in the intestinal tract further compounded these issues, rendering many conventional derivatives less viable as oral therapeutic agents. Consequently, the industry faced a bottleneck in producing fluorinated isoflavone analogs that possessed both synthetic accessibility and robust biological efficacy.
The Novel Approach
The methodology outlined in patent CN101003530B represents a paradigm shift by employing perfluoroalkylsulfonyl fluorides to functionalize the isoflavone core. This approach leverages the high reactivity of the sulfonyl fluoride group towards phenoxide ions, allowing for a direct and efficient substitution reaction under relatively mild conditions. Unlike previous methods that struggled with regioselectivity and yield, this novel route utilizes simple base catalysis in polar aprotic solvents to achieve high conversion rates. The resulting perfluoroalkylsulfonyl isoflavones not only possess improved solubility characteristics due to the lipophilic perfluoro chain but also demonstrate exceptional potency against cancer cell lines. This strategic modification transforms a naturally occurring scaffold into a highly specialized pharmaceutical intermediate suitable for advanced drug discovery programs.
Mechanistic Insights into Perfluoroalkylsulfonylation
The core of this synthesis lies in a nucleophilic substitution mechanism where the hydroxyl groups of the isoflavone substrate act as nucleophiles. Initially, the hydroxyisoflavone is dissolved in anhydrous N,N-dimethylformamide (DMF) and treated with potassium carbonate, which serves to deprotonate the phenolic hydroxyl groups and generate the reactive phenoxide species. Upon the addition of the perfluoroalkylsulfonyl fluoride reagent, the electron-rich oxygen atom of the phenoxide attacks the electrophilic sulfur atom of the sulfonyl fluoride. This results in the displacement of the fluoride ion and the formation of a stable sulfonate ester linkage. The reaction is typically conducted under a nitrogen atmosphere to prevent moisture interference, ensuring the integrity of the sensitive sulfonyl fluoride reagent throughout the process.
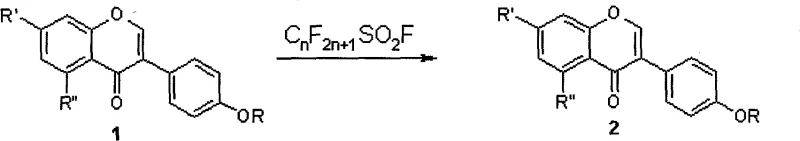
A critical aspect of this mechanism is the remarkable regioselectivity observed during the sulfonylation process. When the isoflavone substrate contains multiple hydroxyl groups, such as at the 7 and 4' positions, the reaction preferentially occurs at the 4'-hydroxyl group. This selectivity is driven by the electronic environment and steric factors inherent to the isoflavone structure, where the 4'-position is more accessible and nucleophilic under the specified basic conditions. For substrates with hydroxyl groups at the 7, 4', and 5 positions, the selectivity remains consistent, targeting the 4'-position exclusively. This intrinsic selectivity simplifies the purification process significantly, as it minimizes the formation of regioisomeric byproducts, thereby enhancing the overall purity of the final pharmaceutical intermediate without the need for complex chromatographic separations.
How to Synthesize Perfluoroalkylsulfonyl Isoflavone Efficiently
The synthesis protocol described in the patent provides a robust framework for producing these high-value intermediates with consistency and reliability. The process begins with the precise dissolution of the hydroxyisoflavone starting material in dry DMF, followed by the addition of dried potassium carbonate powder to initiate the deprotonation phase at room temperature. Once the phenoxide is generated, the perfluoroalkylsulfonyl fluoride is introduced, and the temperature is elevated to facilitate the coupling reaction. The detailed operational parameters, including specific molar ratios and thermal profiles, are essential for maximizing yield and minimizing side reactions. For a comprehensive breakdown of the standardized operating procedures and safety protocols required for this synthesis, please refer to the technical guide below.
- Dissolve hydroxyisoflavone substrate in dry DMF and treat with potassium carbonate powder at room temperature to generate the phenoxide nucleophile.
- Add perfluoroalkylsulfonyl fluoride under nitrogen protection and heat the mixture to 40-120°C to facilitate the sulfonylation reaction.
- Quench the reaction in ice water, filter the precipitate, and purify the crude product via recrystallization to obtain high-purity isoflavone derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers substantial benefits for procurement managers and supply chain leaders focused on cost efficiency and reliability. The primary advantage lies in the simplicity of the reaction setup, which utilizes commodity chemicals such as potassium carbonate and DMF, alongside commercially available hydroxyisoflavone precursors. This reliance on widely sourced raw materials mitigates the risk of supply chain disruptions often associated with exotic or proprietary reagents. Furthermore, the workup procedure involves a straightforward precipitation into ice water followed by filtration and recrystallization, eliminating the need for expensive and time-consuming purification technologies. This streamlined downstream processing translates directly into reduced manufacturing costs and faster turnaround times for batch production.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and complex purification steps significantly lowers the operational expenditure associated with producing these intermediates. By avoiding the use of precious metals, manufacturers save on both reagent costs and the expensive remediation processes required to remove trace metal impurities to meet pharmaceutical standards. Additionally, the high regioselectivity of the reaction reduces waste generation, as fewer byproducts are formed that would otherwise require disposal or recycling. This efficiency allows for a more lean manufacturing model, where resources are utilized optimally to produce high-purity outputs with minimal environmental footprint.
- Enhanced Supply Chain Reliability: The use of stable and shelf-stable reagents ensures that production schedules can be maintained without the volatility often seen with moisture-sensitive or unstable intermediates. Since the starting hydroxyisoflavones can be sourced from established suppliers or synthesized via well-documented literature methods, the supply chain remains resilient against market fluctuations. The robustness of the reaction conditions, which tolerate a range of temperatures and times without significant degradation of yield, further adds to the reliability of the process. This stability is crucial for long-term supply agreements, providing partners with confidence in continuous availability.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory gram-scale experiments to multi-kilogram commercial production without requiring fundamental changes to the reaction engineering. The absence of hazardous heavy metals and the use of standard organic solvents simplify waste management and regulatory compliance, aligning with modern green chemistry initiatives. The ability to recrystallize the product from common solvents like ethanol ensures that the final material meets stringent purity specifications required for clinical applications. This combination of scalability and compliance makes the technology an attractive asset for companies looking to expand their portfolio of oncology intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of perfluoroalkylsulfonyl isoflavones. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and advantages of this technology. Understanding these details is essential for stakeholders evaluating the integration of this synthesis route into their existing manufacturing pipelines or drug discovery workflows.
Q: What is the regioselectivity of the perfluoroalkylsulfonylation reaction on isoflavones?
A: The reaction exhibits high regioselectivity. For isoflavones with multiple hydroxyl groups, such as those at the 7 and 4' positions, the perfluoroalkylsulfonyl group selectively substitutes the hydroxyl group at the 4' position, leaving other positions intact.
Q: What are the biological activities of the synthesized perfluoroalkylsulfonyl isoflavones?
A: These compounds demonstrate potent anticancer activity, specifically showing strong inhibitory effects against HL-60 human leukemia cell lines, with some derivatives exhibiting nearly complete inhibition at low micromolar concentrations.
Q: Why is this synthesis method preferred over traditional trifluoromethylation?
A: Traditional methods for introducing fluorine groups into isoflavones are often complex and yield products with limited solubility improvements. This method uses readily available perfluoroalkylsulfonyl fluorides to achieve simpler reaction conditions and significantly enhanced biological potency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Perfluoroalkylsulfonyl Isoflavone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technology disclosed in patent CN101003530B for the development of next-generation anticancer therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from benchtop discovery to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of perfluoroalkylsulfonyl isoflavone meets the exacting standards required for preclinical and clinical evaluation. We are committed to delivering high-quality intermediates that empower your research teams to push the boundaries of oncology treatment.
We invite you to collaborate with us to leverage this innovative synthesis route for your specific drug development needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and timeline constraints. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a supply chain that prioritizes quality, efficiency, and scientific excellence, ensuring your anticancer programs remain on the cutting edge of innovation.
