Advanced Photochemical Synthesis of Linagliptin Chloro Intermediate for Commercial Scale-Up
The global demand for effective Type 2 diabetes treatments continues to surge, driving intense innovation in the synthesis of DPP-IV inhibitors like linagliptin. As a critical building block in this therapeutic class, the linagliptin chloro intermediate requires a manufacturing process that balances high purity with economic viability. Patent CN112898303A introduces a groundbreaking synthetic method that addresses longstanding inefficiencies in producing this key pharmaceutical intermediate. By leveraging a novel photochemical oxidative coupling strategy, this technology offers a streamlined pathway that bypasses the hazardous reagents and complex multi-step sequences characteristic of legacy protocols. For R&D directors and supply chain leaders, this represents a significant opportunity to optimize the commercial scale-up of complex pharmaceutical intermediates while ensuring robust quality control.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of linagliptin intermediates has relied on routes that are fraught with safety hazards and operational inefficiencies. Traditional methodologies often utilize 6-amino-1-methyl uracil as a starting material, necessitating a dangerous diazotization step involving sodium nitrite. This reagent is not only explosive and difficult to handle on a large scale but also generates significant nitrogen oxide waste, complicating environmental compliance. Furthermore, the conventional pathway involves a tedious sequence of reduction, cyclization, iodination, and substitution reactions. This elongated process inherently accumulates impurities, leading to lower overall yields and requiring extensive purification efforts that drive up manufacturing costs. The reliance on such volatile chemistry creates bottlenecks in the supply chain, limiting the ability of manufacturers to respond rapidly to market demands for high-purity pharmaceutical intermediates.
The Novel Approach
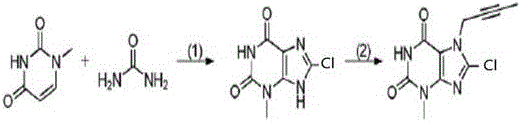
In stark contrast, the innovative method disclosed in the patent utilizes a direct and efficient photochemical strategy. The process begins with the readily available 1-methyluracil, reacting it with urea and N-chlorosuccinimide (NCS) under the irradiation of a high-pressure mercury lamp. This oxidative coupling cyclization occurs under mild acidic conditions, effectively constructing the core heterocyclic structure in a single pot. The subsequent step involves a straightforward nucleophilic substitution with 1-bromo-2-butyne to install the necessary alkyne functionality. This approach drastically reduces the number of unit operations, eliminates the need for explosive nitrites, and operates at moderate temperatures ranging from 50 °C to 85 °C. By simplifying the synthetic tree, this method not only enhances safety but also significantly improves the atom economy and throughput potential for industrial applications.
Mechanistic Insights into Photochemical Oxidative Coupling
The core of this technological breakthrough lies in the photochemically driven oxidative coupling mechanism. Under the influence of high-pressure mercury lamp irradiation, the reaction system activates the N-chlorosuccinimide (NCS) and the oxidant, typically tert-butyl hydroperoxide (TBHP) or hydrogen peroxide. The acid catalyst, preferably sulfuric acid at a molar ratio of 1.0:0.1 relative to the substrate, facilitates the protonation of the uracil ring, increasing its electrophilicity. Simultaneously, the urea acts as a nitrogen source, participating in the cyclization to form the xanthine-like core. The photo-energy input lowers the activation energy barrier for this oxidative transformation, allowing the reaction to proceed smoothly at 85 °C in solvents like dioxane. This precise control over the reaction environment minimizes side reactions, such as over-chlorination or polymerization, which are common pitfalls in thermal-only processes.
Following the formation of the chloro-intermediate (Compound A), the mechanism shifts to a classic SN2-type substitution. The addition of a sterically hindered base, such as N,N-diisopropylethylamine (DIPEA), serves to scavenge the hydrobromic acid generated during the reaction with 1-bromo-2-butyne. The use of DIPEA is critical as it prevents the degradation of the sensitive heterocyclic core while promoting the nucleophilic attack of the nitrogen atom on the propargyl bromide. Conducted in acetone at 65 °C, this step ensures high conversion rates without compromising the integrity of the triple bond. The result is a final product with demonstrated purity levels of 99%, indicating that the mechanistic pathway effectively suppresses the formation of regio-isomers and other structural impurities that often plague alternative synthetic routes.
How to Synthesize Linagliptin Chloro Intermediate Efficiently
Implementing this synthesis requires careful attention to the photochemical parameters and stoichiometric ratios to maximize efficiency. The process is designed to be robust, utilizing common industrial solvents and reagents that are easily sourced from global chemical suppliers. The initial cyclization step demands a controlled environment to manage the exothermic nature of the oxidation, while the subsequent alkylation requires precise temperature control to prevent alkyne polymerization. Detailed standard operating procedures for this specific photochemical transformation are essential for maintaining batch-to-batch consistency. For a comprehensive guide on the exact operational parameters and workup procedures, please refer to the standardized synthesis steps outlined below.
- Perform photochemical reaction on 1-methyluracil, urea, and NCS with an oxidant under acid catalysis and high-pressure mercury lamp irradiation to obtain Compound A.
- Carry out substitution reaction on Compound A with 1-bromo-2-butyne using a base like DIPEA in acetone solvent to obtain the final linagliptin intermediate (Compound B).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers transformative benefits regarding cost structure and logistical reliability. By eliminating the dependency on hazardous sodium nitrite and reducing the total number of reaction steps, the process inherently lowers the consumption of raw materials and utilities. The shift to a shorter, safer workflow reduces the burden on waste treatment facilities and minimizes the risk of production stoppages due to safety incidents. This translates directly into a more resilient supply chain capable of meeting the rigorous delivery schedules of multinational pharmaceutical clients. Furthermore, the use of stable, non-explosive reagents simplifies storage and transportation logistics, removing significant regulatory hurdles associated with dangerous goods.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like sodium nitrite, combined with a reduction in the number of isolation and purification steps, leads to substantial cost savings. The high yields demonstrated in the patent examples suggest a significant improvement in material throughput, reducing the cost of goods sold (COGS) per kilogram of active intermediate. Additionally, the use of common solvents like acetone and dioxane, which are easily recoverable and recyclable, further enhances the economic viability of the process on a commercial scale.
- Enhanced Supply Chain Reliability: Sourcing 1-methyluracil and urea is far more stable and predictable than managing the supply of explosive nitrites, which are subject to strict regulatory controls. This stability ensures a continuous flow of raw materials, reducing lead time for high-purity pharmaceutical intermediates and mitigating the risk of supply disruptions. The robustness of the reaction conditions also means that the process is less susceptible to variations in raw material quality, ensuring consistent output even when sourcing from diverse global suppliers.
- Scalability and Environmental Compliance: The mild reaction conditions (50-85 °C) and the absence of heavy metal catalysts make this process highly amenable to scale-up from pilot plant to full commercial production. The simplified waste stream, devoid of heavy metals and explosive residues, aligns perfectly with modern green chemistry principles and stringent environmental regulations. This ease of compliance reduces the administrative and financial burden on manufacturing sites, facilitating faster approval for cost reduction in pharmaceutical intermediates manufacturing projects.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photochemical synthesis route. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and advantages of adopting this technology for large-scale production. Understanding these details is crucial for technical teams evaluating the transition from legacy processes to this more efficient methodology.
Q: How does this new synthesis route improve safety compared to traditional methods?
A: The novel route eliminates the use of explosive sodium nitrite reagents required in conventional processes, significantly enhancing operational safety and reducing hazardous waste handling costs.
Q: What are the typical yields and purity levels achieved with this photochemical method?
A: Laboratory examples demonstrate yields of up to 82% for the intermediate cyclization step and 96% for the final substitution, with product purity reaching 99% after recrystallization.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method utilizes easily available raw materials, mild reaction conditions (50-85°C), and common solvents like dioxane and acetone, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Linagliptin Intermediate Supplier
As the pharmaceutical industry evolves, the need for advanced, scalable, and compliant synthesis routes becomes paramount. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, offering deep expertise in translating complex laboratory innovations like the photochemical route for linagliptin intermediates into commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the highest global standards, providing a secure foundation for your downstream drug development.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and detailed route feasibility assessments to demonstrate how our optimized manufacturing capabilities can drive value and efficiency for your organization.
