Scalable Synthesis of 3,3-Dimethylcyclopropane-1,2-dicarboxylic Anhydride for Pharmaceutical Manufacturing
The pharmaceutical industry's relentless pursuit of effective treatments for Hepatitis C has placed significant demand on the supply chain for high-quality intermediates, specifically 3,3-dimethylcyclopropane-1,2-dicarboxylic anhydride. Patent CN101863866B introduces a transformative synthesis methodology that addresses critical bottlenecks in yield, safety, and purification that have historically plagued the manufacturing of this vital scaffold. Unlike conventional approaches that rely on hazardous conditions and generate complex impurity profiles, this novel route utilizes dichloro chrysanthemic acid methyl ester as a robust starting material, undergoing a controlled potassium permanganate oxidation followed by a streamlined cyclization step. This technical breakthrough not only enhances the structural integrity of the final product but also aligns perfectly with modern Good Manufacturing Practice (GMP) standards required by global regulatory bodies. For R&D directors and procurement specialists alike, understanding the nuances of this patent is essential for securing a reliable supply of this high-purity API intermediate.
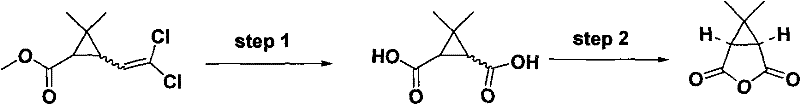
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,3-dimethylcyclopropane-1,2-dicarboxylic anhydride has been fraught with significant operational hazards and inefficiencies that deter large-scale commercialization. Traditional methods, such as those disclosed in earlier patents like CN1805931A, typically employ chrysanthemic acid ethyl ester as the raw material, which necessitates oxidation in pure acetone environments. This specific solvent choice creates a volatile scenario where the exothermic nature of the potassium permanganate oxidation reaction can lead to violent temperature spikes and the rapid evolution of oxygen gas. Such uncontrolled exothermicity poses a severe explosion risk, making industrial scale-up inherently dangerous and often requiring expensive, specialized containment infrastructure. Furthermore, these legacy processes suffer from incomplete oxidation reactions, leaving behind substantial amounts of unreacted starting material and generating a plethora of side products that are chemically similar to the target molecule. The difficulty in separating these impurities often results in lower overall yields and necessitates multiple, costly purification steps that erode profit margins and extend lead times for pharmaceutical manufacturers.
The Novel Approach
The methodology outlined in CN101863866B represents a paradigm shift by fundamentally altering the reaction engineering and raw material selection to mitigate these risks. By switching the substrate to dichloro chrysanthemic acid methyl ester, the reaction pathway becomes more selective, significantly reducing the formation of stubborn byproducts that complicate downstream processing. A critical innovation in this approach is the reversal of the addition order; instead of adding the oxidant to the substrate, the dichloro ester is added dropwise into a suspension of potassium permanganate. This simple yet profound change allows for precise thermal management, ensuring that the heat of reaction is dissipated effectively and preventing the runaway scenarios common in older protocols. Additionally, the introduction of water as a co-solvent or even the primary solvent medium dramatically reduces the flammability of the reaction mixture, effectively neutralizing the explosion hazard associated with pure organic solvents. This enhanced safety profile, combined with the ability to recycle acetone from the mother liquor, creates a manufacturing process that is not only safer for operators but also economically superior due to reduced solvent consumption and waste disposal costs.
Mechanistic Insights into Potassium Permanganate Oxidative Cleavage
At the heart of this synthesis lies a sophisticated oxidative cleavage mechanism driven by potassium permanganate, which acts as a powerful oxidizing agent to convert the dichloro alkene moiety into the corresponding dicarboxylic acid functionality. The reaction proceeds through the formation of a cyclic manganate ester intermediate, which subsequently decomposes to yield the carboxyl groups while maintaining the stereochemical integrity of the cyclopropane ring. The presence of the chlorine atoms on the starting material plays a crucial electronic role, potentially activating the double bond towards nucleophilic attack by the permanganate ion, thereby facilitating a more complete conversion compared to non-halogenated analogs. The solvent system, comprising a tunable ratio of water and acetone (ranging from 1:0 to 1:20), is critical for solubilizing both the organic substrate and the inorganic oxidant, creating a pseudo-homogeneous environment that maximizes collision frequency and reaction efficiency. Maintaining the reaction temperature between 10°C and 40°C is paramount; temperatures below this range slow the kinetics excessively, while higher temperatures risk decomposing the sensitive cyclopropane ring or promoting over-oxidation. This delicate balance ensures that the oxidation stops precisely at the diacid stage without degrading the core scaffold, which is essential for maintaining the biological activity of the downstream Hepatitis C inhibitor.
Controlling the impurity profile is perhaps the most significant advantage of this mechanistic approach, particularly when contrasted with reference examples from prior art. In traditional syntheses using chrysanthemic acid esters, LC-MS analysis often reveals persistent impurities such as Compound II (a mono-oxidized species) and Compound III (a degradation product), which are structurally analogous to the target and notoriously difficult to remove via crystallization. The new route minimizes the generation of these specific contaminants by ensuring a more thorough oxidation environment and avoiding the harsh acidic conditions that promote degradation in earlier steps. As illustrated in the impurity analysis, the new method achieves purities exceeding 98.5% for the diacid intermediate, whereas older methods struggle to reach 93% without extensive chromatography. This high level of chemical purity is transmitted through to the final anhydride product, which can be recrystallized to achieve greater than 99.5% purity. For pharmaceutical applications, where impurity thresholds are strictly regulated, this inherent selectivity reduces the burden on quality control laboratories and ensures a consistent, compliant supply of the active pharmaceutical ingredient intermediate.
How to Synthesize 3,3-Dimethylcyclopropane-1,2-dicarboxylic Anhydride Efficiently
The practical execution of this synthesis requires careful attention to the sequential addition of reagents and temperature control to maximize yield and safety. The process begins with the preparation of a potassium permanganate suspension in the chosen solvent system, cooled to between 10°C and 15°C to absorb the initial heat of reaction. The dichloro chrysanthemic acid methyl ester is then introduced slowly, maintaining the reaction temperature below 40°C throughout the addition and subsequent stirring period of 4 to 6 hours. Following the oxidation, the mixture is treated with sodium bisulfite to quench excess oxidant, followed by acidification to precipitate the dicarboxylic acid, which is then extracted and purified.
- Oxidize dichloro chrysanthemic acid methyl ester using potassium permanganate in a water/acetone solvent system at 10-40°C.
- Perform post-treatment with sodium bisulfite and acidification to isolate 3,3-dimethylcyclopropane-1,2-dicarboxylic acid.
- Cyclize the dicarboxylic acid in acetic anhydride under reflux to form the final anhydride product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic reduction of operational risk; by eliminating the potential for explosive exothermic reactions, facilities can operate with lower insurance premiums and reduced need for specialized blast-proof infrastructure, directly impacting the bottom line. Furthermore, the ability to utilize a water-acetone solvent system allows for the recovery and reuse of acetone, which constitutes a significant portion of the variable costs in traditional batch processing. This solvent recycling capability not only lowers raw material expenditure but also simplifies waste stream management, reducing the environmental footprint and associated disposal fees. The robustness of the reaction conditions means that batch-to-batch variability is minimized, ensuring a consistent supply of material that prevents production stoppages at the drug formulation stage. Consequently, this method supports a more resilient supply chain capable of meeting the rigorous demands of global pharmaceutical markets without the volatility associated with older, less stable chemical processes.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the elimination of expensive purification steps and the optimization of raw material usage. By achieving higher conversion rates and minimizing the formation of hard-to-remove impurities, the overall yield of the process is significantly improved, meaning less starting material is required to produce the same amount of final product. The removal of transition metal catalysts or complex reagents found in alternative routes further simplifies the bill of materials, reducing the cost of goods sold (COGS). Additionally, the simplified workup procedure, which avoids complex chromatographic separations in favor of straightforward extraction and crystallization, reduces labor hours and equipment occupancy time. These cumulative efficiencies translate into substantial cost savings that can be passed down the supply chain, offering a competitive pricing advantage for the final API.
- Enhanced Supply Chain Reliability: Supply continuity is critical for life-saving medications, and this synthesis method enhances reliability by utilizing readily available and stable raw materials. Dichloro chrysanthemic acid methyl ester is a commercially accessible feedstock, reducing the risk of supply bottlenecks that can occur with exotic or custom-synthesized precursors. The process tolerance to variations in solvent ratios (water to acetone) provides operational flexibility, allowing manufacturers to adapt to market fluctuations in solvent availability without compromising product quality. Moreover, the safety improvements mean that production schedules are less likely to be disrupted by safety incidents or regulatory shutdowns, ensuring a steady flow of material to downstream customers. This reliability makes the manufacturer a preferred partner for long-term contracts and strategic alliances in the pharmaceutical sector.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to multi-ton production is often where traditional methods fail, but this route is explicitly designed for industrial scalability. The use of water as a major component of the solvent system improves heat transfer characteristics, making it easier to manage thermal loads in large reactors compared to pure organic systems. The reduction in hazardous waste generation, particularly the avoidance of heavy metal residues and the minimization of organic solvent discharge, aligns with increasingly stringent environmental regulations globally. This compliance reduces the administrative burden of permitting and reporting, accelerating the time to market for new drug applications. The process is inherently greener, supporting corporate sustainability goals while maintaining high production throughput, making it an ideal candidate for green chemistry initiatives within the fine chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 3,3-dimethylcyclopropane-1,2-dicarboxylic anhydride, based on the detailed specifications provided in the patent literature. Understanding these aspects is crucial for technical teams evaluating the feasibility of integrating this intermediate into their existing manufacturing workflows.
Q: What are the primary safety advantages of this synthesis method compared to traditional routes?
A: The patented method eliminates the high explosion risk associated with traditional acetone-based oxidations by utilizing a water/acetone mixed solvent system and controlling the addition order of reagents to manage exothermicity.
Q: How does the new route improve impurity profiles for pharmaceutical applications?
A: By using dichloro chrysanthemic acid methyl ester as the starting material and optimizing the oxidation conditions, the formation of difficult-to-remove side products like Compound II and III is significantly minimized, ensuring higher purity.
Q: Is the solvent system recyclable for cost-effective large-scale production?
A: Yes, the process allows for the recovery and recycling of acetone from the reaction mixture, which drastically reduces raw material consumption and waste treatment costs compared to single-use solvent protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3-Dimethylcyclopropane-1,2-dicarboxylic Anhydride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful development of Hepatitis C therapeutics depends on the availability of high-quality, consistently produced intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch meets the exacting standards required for pharmaceutical use. Our commitment to quality assurance means that we not only deliver the product but also provide comprehensive documentation and support to facilitate your regulatory filings.
We invite you to collaborate with us to optimize your supply chain for this critical intermediate. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our advanced synthesis capabilities can enhance your project's timeline and budget. Let us be your partner in bringing innovative medicines to market faster and more efficiently.
