Advanced Continuous Flow Technology for Manufacturing High-Purity m-Trifluoromethyl Benzyl Chloride Intermediates
Advanced Continuous Flow Technology for Manufacturing High-Purity m-Trifluoromethyl Benzyl Chloride Intermediates
The chemical industry is currently witnessing a paradigm shift towards continuous manufacturing, particularly for hazardous and exothermic transformations such as chloromethylation. A pivotal development in this domain is documented in patent CN113200815A, which discloses a robust method for the continuous flow synthesis of m-trifluoromethyl benzyl chloride. This specific intermediate is a critical building block in the synthesis of various agrochemicals and pharmaceutical agents, necessitating a production protocol that balances high throughput with stringent safety standards. The disclosed technology leverages the inherent advantages of micro-reactor engineering to overcome the thermodynamic and kinetic limitations traditionally associated with batch-wise Friedel-Crafts type alkylations. By transitioning from conventional kettle reactors to a continuous flow architecture, the process achieves a dramatic reduction in reaction time, shifting from hours to mere seconds, while simultaneously enhancing the selectivity for the mono-chloromethylated product. This technological leap not only addresses the safety concerns related to handling corrosive chlorinating agents and strong acids but also provides a scalable pathway for reliable pharmaceutical intermediates supplier networks to meet global demand without compromising on quality or environmental compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of m-trifluoromethyl benzyl chloride has been plagued by significant inefficiencies and safety hazards inherent to batch processing. Traditional routes often rely on the chlorination of m-xylene followed by fluorination, a multi-step sequence characterized by prolonged reaction times, poor conversion rates, and low overall selectivity. Alternatively, direct chloromethylation using toxic reagents like chloromethyl methyl ether poses severe occupational health risks due to its carcinogenic nature. Furthermore, in conventional batch reactors, the mixing of organic and acidic phases is often inefficient, leading to localized hot spots that promote unwanted side reactions. Specifically, the lack of precise stoichiometric control frequently results in the formation of difficult-to-remove impurities, such as dichloromethylated derivatives and bimolecular substitution products. As illustrated in the structural analysis of potential byproducts, these impurities compromise the purity profile of the final API intermediate, necessitating costly and wasteful downstream purification steps.
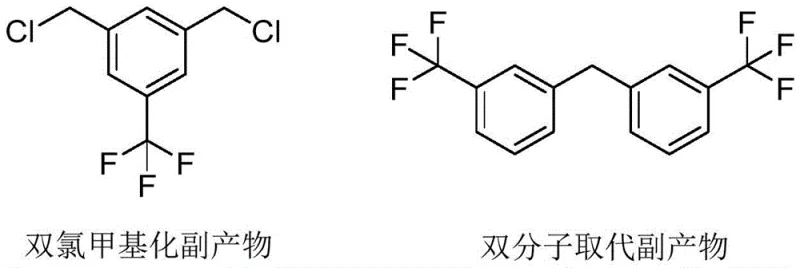
The accumulation of these byproducts is not merely a quality issue but a substantial economic burden, as separating structurally similar halogenated aromatics often requires energy-intensive distillation or chromatography. Moreover, the batch process suffers from a significant "amplification effect," where heat dissipation becomes increasingly difficult as the reactor volume increases, leading to thermal runaway risks and inconsistent batch-to-batch reproducibility. The release of hydrogen chloride gas in an uncontrolled manner further exacerbates equipment corrosion and environmental compliance challenges, making the conventional kettle-type reaction process unsuitable for modern, green chemical manufacturing standards. Consequently, the industry has long sought a methodology that can mitigate these risks while improving the atom economy and spatial-time yield of the transformation.
The Novel Approach
The innovative continuous flow methodology described in the patent data represents a decisive break from these legacy constraints by utilizing a plug-flow reactor system that ensures rapid and homogeneous mixing of reactants. In this advanced setup, the organic solution containing trifluoromethyl benzene and a formaldehyde source is precisely metered against an acidic solution containing the chlorinating agent and a phase transfer catalyst. This intimate contact between phases, facilitated by the high surface-area-to-volume ratio of the flow channels, allows for instantaneous heat exchange, effectively neutralizing the exotherm of the chloromethylation reaction. The result is a process that operates safely at elevated temperatures (50-70°C) with residence times as short as 180 seconds, achieving conversion rates and selectivities that are unattainable in batch mode. By strictly controlling the molar ratios and flow dynamics, the system suppresses the formation of poly-chlorinated species, thereby delivering a crude product stream with exceptional purity that simplifies the final isolation steps. This approach not only enhances the safety profile by minimizing the inventory of hazardous reagents at any given time but also aligns perfectly with the goals of cost reduction in pharmaceutical intermediates manufacturing through improved resource efficiency.
Mechanistic Insights into Phase Transfer Catalyzed Chloromethylation
To fully appreciate the technical superiority of this continuous flow protocol, one must examine the mechanistic role of the phase transfer catalyst (PTC) within the heterogeneous reaction mixture. The reaction involves the interaction of an organic substrate, m-trifluoromethyl benzene, with an electrophilic chloromethylating species generated in the aqueous acid phase. Without effective mass transfer, the reaction would be limited by the diffusion of ions across the phase boundary. The inclusion of a quaternary ammonium salt, such as cetyltrimethylammonium bromide, acts as a molecular shuttle, transporting the active chlorinating complex into the organic phase where the aromatic substitution occurs. This catalytic cycle is significantly accelerated in the turbulent flow regime of the reactor, ensuring that the concentration of the active electrophile remains optimal throughout the reaction channel. The precise temperature control afforded by the flow system prevents the decomposition of the unstable chloromethyl intermediates, which is a common failure mode in batch reactors where thermal gradients exist. Consequently, the reaction proceeds with high fidelity towards the desired mono-substituted product, preserving the integrity of the sensitive trifluoromethyl group.
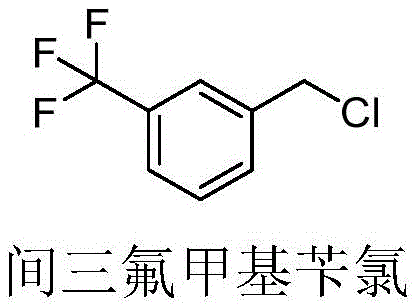
Furthermore, the choice of chlorinating agent plays a pivotal role in the mechanistic pathway and the resulting impurity profile. The patent highlights the efficacy of using chlorosulfonic acid or phosphorus trichloride in conjunction with concentrated sulfuric acid. These reagents generate highly reactive species that, under the controlled conditions of the flow reactor, selectively attack the meta-position of the trifluoromethyl benzene ring. The electron-withdrawing nature of the trifluoromethyl group deactivates the ring, requiring potent electrophiles and optimized conditions to achieve reasonable reaction rates. The continuous flow environment provides exactly these conditions by maintaining a high local concentration of reactants without allowing the bulk temperature to spike. This delicate balance ensures that the reaction kinetics favor the formation of the target high-purity OLED material precursor or agrochemical intermediate over thermodynamic byproducts. The ability to fine-tune these parameters in real-time offers R&D teams unprecedented control over the synthetic outcome, facilitating the rapid optimization of reaction conditions for diverse substrate scopes.
How to Synthesize m-Trifluoromethyl Benzyl Chloride Efficiently
Implementing this continuous flow synthesis requires a systematic approach to reagent preparation and equipment configuration to ensure optimal performance and safety. The process begins with the preparation of two distinct feed streams: an organic phase consisting of trifluoromethyl benzene dissolved with a polymerized formaldehyde source like trioxymethylene, and an acidic phase comprising the chlorinating agent and phase transfer catalyst dissolved in concentrated sulfuric acid. These streams are then independently preheated to a specific temperature range, typically between 50°C and 70°C, before being introduced into the continuous flow reactor via precision metering pumps. The synchronization of flow rates is critical, with the patent specifying a molar ratio optimization that ensures the chlorinating agent is present in slight excess to drive the reaction to completion without promoting over-chlorination. Detailed standardized synthesis steps for replicating this high-efficiency protocol are provided in the technical guide below.
- Prepare the organic phase by dissolving formaldehyde sources like trioxymethylene into trifluoromethyl benzene.
- Prepare the acid phase by dissolving a chlorinating agent and a quaternary ammonium phase transfer catalyst into concentrated sulfuric acid.
- Pump both phases into a preheated continuous flow reactor at controlled flow rates (e.g., 50-70°C) for rapid reaction and subsequent purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this continuous flow technology offers compelling strategic advantages that extend beyond simple yield improvements. The primary value driver is the substantial reduction in operational costs associated with waste management and energy consumption. By eliminating the need for prolonged heating and cooling cycles typical of batch processes, the facility's energy footprint is drastically lowered, directly impacting the cost of goods sold. Additionally, the enhanced selectivity of the flow process means that raw materials are converted more efficiently into the desired product, reducing the volume of hazardous waste that requires expensive disposal. This efficiency gain translates into a more competitive pricing structure for the final intermediate, allowing downstream partners to optimize their own manufacturing margins. The elimination of carcinogenic reagents like chloromethyl methyl ether also reduces regulatory compliance costs and insurance premiums, further contributing to the overall economic viability of the supply chain.
- Cost Reduction in Manufacturing: The implementation of continuous flow chemistry fundamentally alters the cost structure of producing m-trifluoromethyl benzyl chloride by maximizing atom economy and minimizing solvent usage. Unlike batch processes that require large volumes of solvent to manage heat and viscosity, flow reactors operate with high concentrations, significantly reducing the load on solvent recovery systems. The precise control over reaction parameters eliminates the need for extensive rework or reprocessing of off-spec batches, ensuring that nearly every kilogram of input material contributes to the final revenue-generating product. Furthermore, the use of robust, non-precious metal catalysts and readily available acid reagents ensures that the raw material basket remains stable and affordable, shielding the supply chain from volatility in specialty chemical markets.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the unpredictability of batch manufacturing, where equipment failures or safety incidents can halt production for extended periods. The continuous flow system mitigates these risks by operating in a closed, automated environment that requires minimal human intervention, thereby reducing the likelihood of operator error. The modular nature of flow chemistry also allows for "numbering up" rather than scaling up, meaning that capacity can be increased by adding parallel reactor units without the need for massive capital investment in new infrastructure. This flexibility ensures that suppliers can respond rapidly to fluctuations in market demand, providing a reliable source of commercial scale-up of complex pharmaceutical intermediates even during periods of high global demand.
- Scalability and Environmental Compliance: Environmental regulations are becoming increasingly stringent, particularly regarding the emission of volatile organic compounds and acidic gases. The continuous flow process addresses these concerns by containing the reaction within sealed tubing, effectively capturing hydrogen chloride gas and preventing fugitive emissions. The improved heat exchange efficiency also means that the process generates less thermal waste, aligning with corporate sustainability goals. From a scalability perspective, the linear relationship between reactor volume and throughput in flow chemistry simplifies the path from pilot plant to full commercial production. This predictability allows supply chain planners to forecast production timelines with greater accuracy, reducing lead time for high-purity agrochemical intermediates and ensuring that downstream manufacturing schedules are met without delay.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the continuous flow synthesis of m-trifluoromethyl benzyl chloride, based on the detailed specifications provided in the patent literature. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this technology for their own production lines or for procurement specialists assessing supplier capabilities. The answers provided reflect the specific advantages of the flow chemistry approach over traditional batch methods, focusing on safety, purity, and scalability metrics that are critical for decision-making in the fine chemical sector.
Q: Why is continuous flow preferred over batch reactors for this chloromethylation?
A: Continuous flow offers superior heat exchange and mixing efficiency, preventing the thermal runaway and byproduct formation common in batch processes.
Q: What are the primary impurities avoided by this new method?
A: The process significantly reduces dichloromethylation and bimolecular substitution byproducts through precise stoichiometric control.
Q: Is this process scalable for industrial production?
A: Yes, the patent demonstrates successful scale-up from gram to kilogram levels with consistent yield and purity, suitable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable m-Trifluoromethyl Benzyl Chloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced intermediates requires more than just a patented process; it demands a partner with the technical expertise to execute it flawlessly at scale. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify that every batch of m-trifluoromethyl benzyl chloride meets the exacting standards required by the global pharmaceutical and agrochemical industries. Our dedication to continuous improvement means that we are constantly optimizing our flow chemistry platforms to deliver higher yields and lower costs for our clients.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage potential partners to contact us directly to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven performance. Let us help you secure a sustainable and cost-effective supply of this critical intermediate, driving innovation and efficiency in your own manufacturing operations.
