Advanced Synthesis of 3-Methylthiophene-2-Aldehyde for Commercial Scale-Up and High Purity
Advanced Synthesis of 3-Methylthiophene-2-Aldehyde for Commercial Scale-Up and High Purity
The global demand for high-performance heterocyclic intermediates continues to surge, driven by the relentless innovation in pharmaceutical and agrochemical sectors. Among these critical building blocks, 3-methylthiophene-2-aldehyde stands out as a versatile precursor for constructing complex molecular architectures found in life-saving drugs and crop protection agents. However, the historical challenge has always been balancing high purity with economic viability and operational safety. A pivotal breakthrough in this domain is documented in Chinese Patent CN102993163A, which discloses a robust, two-step synthesis method that fundamentally redefines the production landscape for this valuable compound. By shifting away from hazardous organolithium reagents and waste-intensive Vilsmeier reactions, this technology offers a pathway to >99% purity with exceptional yield efficiency. For R&D directors and supply chain leaders, understanding the nuances of this oxidative bromination and subsequent Grignard formylation strategy is essential for securing a reliable supply chain and optimizing manufacturing costs in an increasingly regulated environment.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-methylthiophene-2-aldehyde has been plagued by significant technical and safety hurdles that hinder scalable commercial production. Traditional literature, such as US 2008/21026 and WO2007/19884, describes routes relying on n-butyllithium for lithiation followed by DMF quenching. While chemically feasible, this approach presents severe safety liabilities because n-butyllithium is pyrophoric, requiring stringent inert atmosphere controls and cryogenic temperatures often below -78°C to prevent thermal runaway. Furthermore, the selectivity of lithiation is imperfect, frequently generating the 4-isomer (4-thiotolene-2-formaldehyde) in quantities around 20%. Since the boiling points of these isomers are remarkably close, fractional distillation becomes energy-intensive and often fails to achieve the high purity required for pharmaceutical applications. Alternative methods utilizing Vilsmeier reagents suffer from their own drawbacks, primarily the generation of massive volumes of acidic wastewater and difficult downstream purification processes, making them environmentally unsustainable for modern green chemistry standards.
The Novel Approach
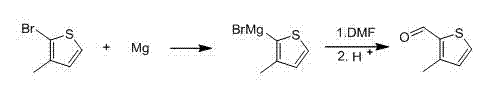
The methodology outlined in patent CN102993163A introduces a paradigm shift by employing a mild oxidative bromination strategy followed by a controlled Grignard reaction. Instead of aggressive lithiation, the process initiates with the reaction of 3-methylthiophene with a bromide source and hydrogen peroxide in an aqueous system. This electrophilic substitution is highly regioselective, favoring the 2-position due to the electronic activation of the thiophene ring and the steric guidance of the methyl group. The reaction proceeds smoothly at temperatures between 5°C and 10°C, eliminating the need for extreme cryogenics. Following the isolation of the intermediate 3-methyl-2-bromothiophene, the second step involves the formation of a Grignard reagent using magnesium turnings in tetrahydrofuran (THF). This organomagnesium species then reacts with N,N-dimethylformamide (DMF) to install the aldehyde functionality. This route not only circumvents the safety risks of butyllithium but also leverages the recyclability of the aqueous bromide layer, creating a closed-loop system that minimizes waste discharge and maximizes atom economy.
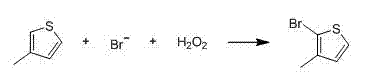
Mechanistic Insights into Oxidative Bromination and Grignard Formylation
The core of this technological advancement lies in the precise control of the electrophilic aromatic substitution during the bromination phase. In the presence of hydrogen peroxide and hydrobromic acid (or a bromide salt), an active brominating species is generated in situ, likely hypobromous acid or molecular bromine, which attacks the electron-rich thiophene ring. The methyl substituent at the 3-position exerts an ortho/para directing effect, but the 2-position is kinetically favored over the 5-position due to the proximity to the sulfur atom which stabilizes the cationic intermediate through resonance. The patent specifies maintaining the reaction temperature strictly between 5°C and 10°C during the addition of hydrogen peroxide. This thermal control is critical; it suppresses poly-bromination and prevents the formation of the undesired 4-bromo isomer. After the reaction reaches completion and warms to 20°C, the organic layer is separated and washed with sodium sulfite to quench residual oxidants, ensuring the stability of the brominated intermediate before vacuum rectification yields 3-methyl-2-bromothiophene with purity levels reaching 99.3%.
Following the successful isolation of the bromide, the mechanism transitions to organometallic chemistry via the Grignard formation. Magnesium metal inserts into the carbon-bromine bond of the 3-methyl-2-bromothiophene in anhydrous THF, generating the corresponding Grignard reagent. This step is initiated thermally and then maintained at 20°C to 30°C, a significantly milder range compared to traditional lithiation protocols. The nucleophilic carbon of the Grignard reagent subsequently attacks the carbonyl carbon of DMF, forming a stable tetrahedral intermediate. Upon acidic hydrolysis, this intermediate collapses to release the target aldehyde, 3-methylthiophene-2-aldehyde. The use of DMF as both a reactant and a solvent component allows for excellent solubility and reaction kinetics. The final workup involves extraction with toluene and washing with sodium bicarbonate to remove acidic byproducts, followed by high-vacuum distillation. This sequence ensures that the final product achieves a purity of over 99.1%, with yields consistently reported above 92%, demonstrating the robustness of the catalytic cycle and the efficiency of the purification steps.
How to Synthesize 3-Methylthiophene-2-Aldehyde Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and thermal management to replicate the high yields reported in the patent data. The process begins with the preparation of the bromination mixture, where the molar ratio of 3-methylthiophene to hydrobromic acid and hydrogen peroxide is optimized at approximately 1:1.2:1. Operators must ensure efficient stirring and heat exchange capabilities to manage the exotherm during peroxide addition. Once the intermediate is secured, the Grignard step demands strictly anhydrous conditions to prevent premature quenching of the organomagnesium species. The addition of the bromide to the magnesium slurry should be controlled to maintain a steady reflux without runaway heating. Finally, the formylation with DMF is conducted at ambient to slightly elevated temperatures (20-30°C), followed by a standard aqueous workup. For detailed operational parameters, equipment specifications, and safety protocols required for GMP manufacturing, please refer to the standardized synthesis guide below.
- Perform oxidative bromination of 3-methylthiophene using hydrobromic acid and hydrogen peroxide at 5-10°C to obtain 3-methyl-2-bromothiophene.
- Generate the Grignard reagent by reacting 3-methyl-2-bromothiophene with magnesium chips in tetrahydrofuran (THF).
- React the Grignard intermediate with N,N-dimethylformamide (DMF) at 20-30°C, followed by hydrolysis and vacuum distillation to isolate the final aldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented synthesis method represents a strategic opportunity to de-risk the supply of critical thiophene intermediates. The elimination of n-butyllithium removes a major bottleneck associated with the storage and handling of hazardous materials, thereby reducing insurance premiums and facility compliance costs. Furthermore, the ability to recycle the aqueous bromide solution directly impacts the cost of goods sold (COGS) by minimizing raw material consumption. The high selectivity of the reaction reduces the burden on downstream purification units, leading to significant energy savings and increased throughput capacity. These factors collectively enhance the reliability of supply, ensuring that production schedules are not disrupted by safety incidents or complex separation challenges.
- Cost Reduction in Manufacturing: The economic benefits of this route are derived primarily from the substitution of expensive and hazardous reagents with commodity chemicals like hydrogen peroxide and hydrobromic acid. By avoiding the use of n-butyllithium, manufacturers eliminate the need for specialized cryogenic reactors and the associated high energy costs for maintaining sub-zero temperatures. Additionally, the patent highlights the recyclability of the bromide-containing aqueous phase, which significantly lowers the consumption of bromine sources over multiple batches. This closed-loop approach not only reduces raw material expenses but also minimizes waste disposal fees, contributing to a substantially lower overall production cost structure compared to conventional lithiation or Vilsmeier methods.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the logistical complexities of sourcing pyrophoric reagents and managing their short shelf-lives. This new method relies on stable, shelf-stable reagents that are readily available in the global chemical market, reducing the risk of supply disruptions. The simplified process flow, characterized by fewer unit operations and milder reaction conditions, allows for faster batch turnover times. Consequently, manufacturers can respond more agilely to fluctuations in market demand, ensuring a steady flow of high-purity 3-methylthiophene-2-aldehyde to downstream API producers without the delays typically associated with complex isomer separations or hazardous material transport regulations.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental risks, but this technology is inherently designed for scalability. The use of aqueous oxidative systems and standard Grignard conditions translates well from pilot plants to multi-ton commercial reactors. From an environmental perspective, the drastic reduction in wastewater generation addresses one of the most pressing concerns in fine chemical manufacturing. By recycling the bromide stream and avoiding the heavy metal catalysts or chlorinated solvents often found in alternative routes, the process aligns with strict international environmental regulations. This compliance facilitates smoother regulatory approvals and enhances the sustainability profile of the final pharmaceutical or agrochemical products, a key metric for modern ESG-focused supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 3-methylthiophene-2-aldehyde using this advanced oxidative bromination technology. These insights are derived directly from the experimental data and beneficial effects described in patent CN102993163A, providing clarity on yield expectations, purity profiles, and operational safety. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for large-scale manufacturing.
Q: What are the primary safety advantages of this synthesis method compared to traditional butyllithium routes?
A: Traditional methods often utilize n-butyllithium, which is pyrophoric and requires extreme cryogenic conditions (-78°C), posing significant fire hazards and high energy costs. The patented method utilizes hydrogen peroxide and hydrobromic acid under mild temperatures (5-10°C), drastically reducing operational risks and eliminating the need for specialized cryogenic infrastructure.
Q: How does this process address the issue of isomer separation and purity?
A: Conventional Vilsmeier-Haack reactions often produce difficult-to-separate isomers like 4-thiotolene-2-formaldehyde due to similar boiling points. This oxidative bromination route exhibits high regioselectivity for the 2-position, effectively suppressing isomer formation and allowing for the production of 3-methylthiophene-2-aldehyde with purity exceeding 99% after simple vacuum rectification.
Q: Is the aqueous waste stream manageable in this synthetic route?
A: Yes, a key feature of this technology is the recyclability of the bromide-containing aqueous solution. Unlike methods generating large volumes of acidic wastewater, the bromide ions remaining after extraction can be recovered and reused, significantly reducing three-waste discharge and lowering raw material consumption costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Methylthiophene-2-Aldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity levels demonstrated in patent literature are faithfully reproduced on an industrial scale. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 3-methylthiophene-2-aldehyde meets the exacting standards required for pharmaceutical and agrochemical applications. Our commitment to quality assurance ensures that your supply chain remains uninterrupted and compliant with global regulatory frameworks.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific project needs. Whether you require custom synthesis services or bulk supply of this critical intermediate, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing processes can drive value and efficiency for your organization.
