Scalable Electrooxidative Cyclization for High-Purity Pyridazine Intermediates Production
The pharmaceutical and agrochemical industries are increasingly demanding sustainable synthetic routes that minimize environmental impact while maintaining high efficiency. Patent CN113981477A introduces a groundbreaking one-pot two-step electrooxidation cyclization method for preparing pyridazine compounds, addressing critical pain points in heterocyclic synthesis. This technology leverages constant current electrolysis to drive the cyclization of 2-phenylethynyl acetophenone derivatives with benzenesulfonyl hydrazides, effectively bypassing the need for stoichiometric chemical oxidants. By utilizing electricity as a clean reagent, this process aligns perfectly with the global shift towards green manufacturing, offering a robust alternative for producing bioactive scaffolds found in numerous herbicides and anticancer agents.
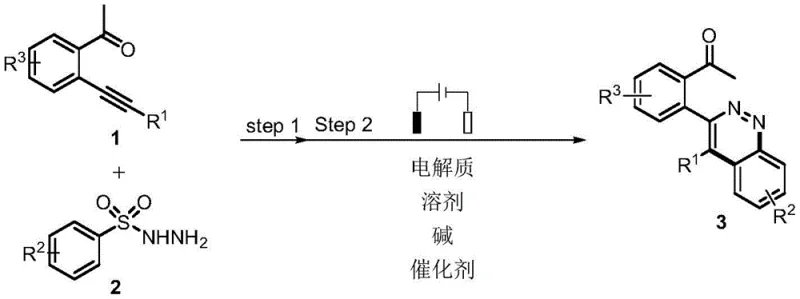
For R&D directors evaluating new pathways, the elimination of hazardous reagents is a primary concern. Traditional methods often rely on harsh conditions that generate significant waste streams, whereas this electrochemical approach operates under mild parameters, typically between 30°C and 60°C. The versatility of the substrate scope allows for the introduction of various functional groups, including halogens and alkyl chains, without compromising the integrity of the pyridazine ring. This level of control is essential for developing reliable pyridazine intermediate supplier capabilities, ensuring that diverse molecular architectures can be accessed efficiently for drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the pyridazine ring has been fraught with challenges related to reagent toxicity and operational complexity. Conventional synthetic strategies frequently employ transition metal catalysts such as copper or palladium, which not only increase raw material costs but also necessitate rigorous purification steps to remove trace metal residues that are strictly regulated in pharmaceutical products. Furthermore, these traditional routes often require strong chemical oxidants to drive the reaction forward, leading to the generation of substantial amounts of hazardous by-products that complicate waste management and increase the overall environmental footprint of the manufacturing process.
The Novel Approach
In stark contrast, the novel electrooxidative cyclization method described in the patent utilizes electrons as the primary oxidant, fundamentally changing the economic and ecological equation of pyridazine synthesis. By employing a simple undivided cell setup with a carbon cloth anode and a platinum cathode, the reaction proceeds smoothly under constant current conditions without the need for external oxidizing agents. This shift not only simplifies the post-reaction workup but also drastically reduces the consumption of auxiliary chemicals, representing a significant advancement in cost reduction in pharmaceutical intermediate manufacturing where reagent costs and waste disposal fees are major budget drivers.
Mechanistic Insights into Electrooxidative Cyclization
The core of this innovation lies in the synergistic interaction between the electrochemical potential and the specific organic catalysts employed. The patent highlights the use of phenothiazine-derived catalysts, specifically Cat 1 through Cat 6, which act as redox mediators to facilitate the electron transfer process. These catalysts, particularly those with electron-withdrawing groups like the cyano-substituted Cat 1, lower the oxidation potential required for the reaction, thereby enhancing energy efficiency and selectivity. The mechanism likely involves the anodic oxidation of the hydrazide or the alkyne substrate to generate reactive radical intermediates that subsequently undergo cyclization to form the stable pyridazine core.
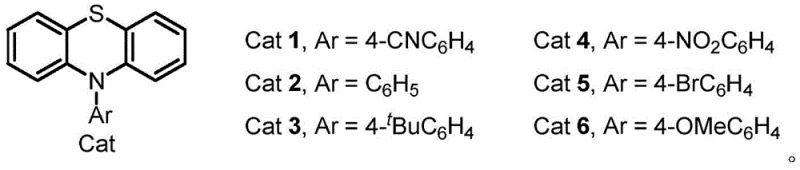
From an impurity control perspective, the mild nature of the electrochemical environment plays a pivotal role in ensuring product quality. Unlike thermal methods that might promote side reactions such as polymerization or decomposition of sensitive functional groups, the controlled potential electrolysis minimizes these pathways. The use of a mixed solvent system comprising acetonitrile, trifluoroethanol, and water further stabilizes the ionic species involved in the catalytic cycle. This precise control over the reaction milieu ensures that the resulting high-purity pyridazine compounds meet the stringent specifications required for downstream applications in medicinal chemistry and crop protection.
How to Synthesize Pyridazine Compounds Efficiently
Implementing this electrochemical protocol requires careful attention to the composition of the electrolyte and the choice of electrode materials to maximize yield and reproducibility. The process begins with dissolving the substrates in a optimized solvent mixture containing a supporting electrolyte like tetrabutylammonium tetrafluoroborate and a base such as potassium carbonate. Detailed standardized synthesis steps see the guide below.
- Dissolve 2-phenylethynyl acetophenone and benzenesulfonyl hydrazide in a mixed solvent system containing electrolyte, base, and catalyst.
- Insert carbon cloth anode and platinum cathode into the solution and apply constant current (5-15mA) at 30-60°C under open system conditions.
- Upon completion, wash with saturated NaHCO3, extract with ethyl acetate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to electrochemical synthesis offers tangible benefits beyond mere technical novelty. The removal of expensive noble metal catalysts and toxic oxidants directly translates to a leaner bill of materials, reducing the volatility associated with sourcing specialized reagents. Additionally, the simplified workup procedure, which avoids complex extraction protocols needed to remove metal residues, shortens the production cycle time and lowers labor costs, contributing to substantial cost savings in the overall manufacturing budget.
- Cost Reduction in Manufacturing: The elimination of stoichiometric oxidants and the use of low-loading organic catalysts significantly decrease raw material expenses. Furthermore, the avoidance of heavy metals removes the need for costly scavenging resins and extensive analytical testing for metal limits, streamlining the quality control process and reducing overhead.
- Enhanced Supply Chain Reliability: The starting materials, such as substituted acetophenones and sulfonyl hydrazides, are commodity chemicals with robust global supply chains, ensuring consistent availability. The reliance on electricity as a reagent rather than finite chemical oxidants insulates the production process from supply disruptions related to specialty chemical shortages.
- Scalability and Environmental Compliance: The reaction conditions are inherently safer, operating at near-ambient temperatures and pressures, which facilitates easier scale-up from laboratory to pilot and commercial plants. The green nature of the process, generating minimal hazardous waste, simplifies regulatory compliance and reduces the burden on wastewater treatment facilities, aligning with modern sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this electrochemical technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity for potential adopters.
Q: What are the advantages of using electrochemical oxidation over traditional metal catalysts for pyridazine synthesis?
A: The electrochemical method eliminates the need for stoichiometric toxic oxidants and expensive transition metal catalysts, significantly reducing heavy metal residue risks and waste treatment costs while adhering to green chemistry principles.
Q: Can this electrooxidative cyclization process be scaled for industrial production?
A: Yes, the process utilizes mild reaction conditions (30-60°C) and simple undivided cells with robust electrodes like carbon cloth and platinum, making it highly amenable to scale-up for commercial manufacturing of complex heterocycles.
Q: What is the typical yield and purity profile for these pyridazine derivatives?
A: Experimental data indicates yields ranging from 54% to 77% depending on substituents, with high purity achievable through standard workup and chromatography due to the selectivity of the electro-catalytic cycle.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyridazine Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modernizing the production of complex heterocycles. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of pyridazine intermediates meets the highest international standards for pharmaceutical and agrochemical applications.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our electrochemical capabilities can optimize your supply chain and enhance your product competitiveness in the global market.
