Breakthrough Total Synthesis of Luteoride A: Scalable Route for High-Purity Indole Alkaloids
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access complex bioactive molecules, and the recent disclosure in patent CN114890932A represents a significant milestone in the field of indole alkaloid synthesis. This patent details the first total synthesis process of Luteoride A, a valuable indole alkaloid, introducing a groundbreaking strategy that installs the critical isoprene group directly at the 7-position of the indole ring without the need for protecting groups. This innovation addresses long-standing challenges in synthetic organic chemistry by leveraging redox economy and atom economy, thereby offering a streamlined alternative to conventional multi-step sequences. For research and development directors and procurement specialists, this technology signals a shift towards safer, more cost-effective manufacturing protocols that minimize reliance on hazardous reagents while maximizing output quality. The methodology not only simplifies the synthetic tree but also enhances the environmental profile of the production process, aligning perfectly with modern green chemistry principles and regulatory demands for sustainable manufacturing practices in the pharmaceutical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
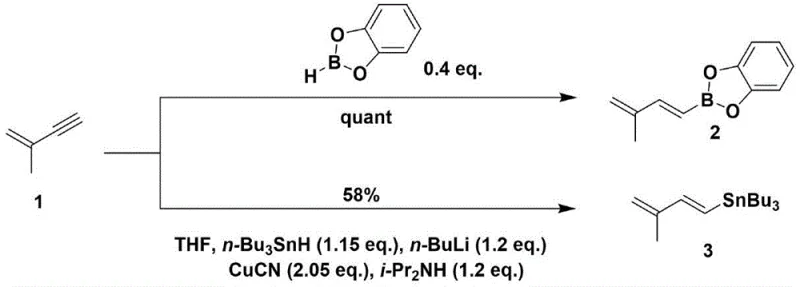
Historically, the introduction of prenyl or isoprene groups onto indole scaffolds has been fraught with significant technical and safety hurdles that complicate commercial scale-up. Traditional methodologies often rely on the use of highly toxic reagents such as trimethylsilyldiazomethane and cuprous cyanide, which pose severe health risks to operators and require specialized containment infrastructure to manage exposure. Furthermore, these legacy routes frequently necessitate the use of pyrophoric and highly flammable reagents like n-butyllithium and tri-n-butyltin hydride, which demand rigorous safety protocols, inert atmosphere handling, and expensive quenching procedures to prevent catastrophic incidents. The reliance on expensive organoboron reagents like catecholborane further inflates the raw material costs, rendering the final active pharmaceutical ingredient economically unviable for mass production. Additionally, conventional strategies often suffer from poor atom economy due to the necessity of multiple protection and deprotection steps, generating substantial chemical waste and lowering the overall process mass intensity, which is a critical metric for supply chain sustainability.
The Novel Approach
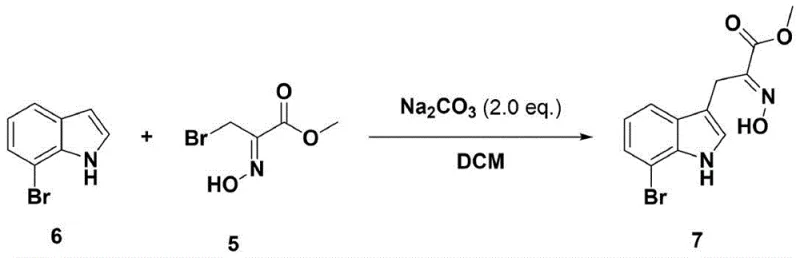
In stark contrast to these cumbersome legacy methods, the novel process disclosed in the patent utilizes a direct, protecting-group-free strategy that fundamentally reshapes the synthetic landscape for Luteoride A. By employing a heteroatom Diels-Alder reaction followed by aromatization, the process achieves the construction of the functionalized indole core with remarkable efficiency and selectivity. This approach completely bypasses the need for dangerous organometallic reagents, substituting them with stable, commercially available starting materials such as methyl bromopyruvate and hydroxylamine hydrochloride. The elimination of protection groups not only reduces the number of unit operations but also significantly improves the overall yield by minimizing material loss during intermediate purification stages. This streamlined workflow translates directly into reduced production timelines and lower capital expenditure on safety equipment, making it an attractive option for manufacturers aiming to optimize their cost structures while maintaining high standards of product purity and regulatory compliance.
Mechanistic Insights into the Protecting-Group-Free Synthesis
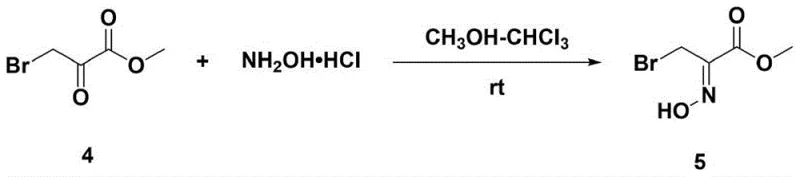
The core of this innovative synthesis lies in the strategic formation of the oxime ester intermediate, which serves as a versatile dipole in the subsequent cycloaddition reaction. In the initial step, methyl bromopyruvate reacts with hydroxylamine hydrochloride under mild conditions to generate the key oxime species (Compound 5) with an impressive yield of 87%. This transformation is critical as it activates the carbonyl functionality for the subsequent nucleophilic attack, setting the stage for the construction of the carbon-nitrogen framework essential for the indole scaffold. The reaction proceeds smoothly in a mixed solvent system of chloroform and methanol at room temperature, demonstrating excellent functional group tolerance and eliminating the need for cryogenic conditions often associated with sensitive intermediate formations. The stability of this oxime intermediate allows for straightforward isolation and purification, ensuring that the downstream reactions proceed with high fidelity and minimal impurity carryover, which is paramount for meeting stringent pharmaceutical quality standards.
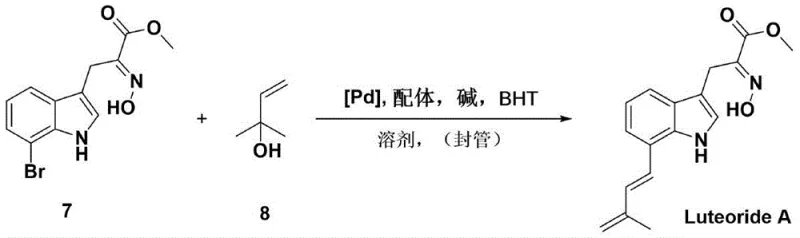
Following the formation of the oxime, the process advances through a sophisticated palladium-catalyzed Heck coupling to install the final isoprene moiety. This step involves the reaction of the brominated indole intermediate with 2-methyl-3-buten-2-ol in the presence of a palladium catalyst and a phosphine ligand. The mechanism facilitates the formation of a carbon-carbon bond at the 7-position of the indole ring, followed by an in situ elimination of the tertiary hydroxyl group to yield the conjugated diene system characteristic of Luteoride A. This tandem coupling-elimination sequence is highly efficient, achieving yields of up to 92-94%, and avoids the formation of regioisomers that typically plague electrophilic aromatic substitutions on indole rings. The use of BHT as an additive further stabilizes the reaction mixture against radical polymerization of the diene product, ensuring high purity and consistent batch-to-batch reproducibility, which are critical factors for commercial viability.
How to Synthesize Luteoride A Efficiently
The synthesis of Luteoride A via this patented route offers a robust and reproducible protocol that can be adapted for both laboratory-scale discovery and industrial-scale production. The process is divided into three distinct stages, beginning with the preparation of the oxime ester, followed by the heteroatom Diels-Alder cyclization with 7-bromoindole, and concluding with the palladium-catalyzed functionalization. Each step has been optimized to maximize yield and minimize waste, utilizing common solvents and reagents that are easily sourced from global chemical suppliers. The detailed standardized synthesis steps outlined below provide a clear roadmap for chemists to replicate this high-efficiency pathway, ensuring that the critical quality attributes of the final API intermediate are met with precision. By following these guidelines, manufacturing teams can achieve consistent results while adhering to safety and environmental best practices.
- React methyl bromopyruvate with hydroxylamine hydrochloride in a methanol-chloroform solvent system at room temperature to form the key oxime intermediate (Compound 5) with high yield.
- Perform a heteroatom Diels-Alder reaction between the oxime intermediate and 7-bromoindole in dichloromethane using sodium carbonate as a base to construct the functionalized indole core (Compound 7).
- Execute a palladium-catalyzed Heck coupling reaction between the indole intermediate and 2-methyl-3-buten-2-ol, followed by in situ elimination, to finalize the isoprene installation and yield Luteoride A.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis route presents a compelling value proposition driven by significant reductions in operational complexity and raw material costs. The elimination of exotic and hazardous reagents such as n-butyllithium and trimethylsilyldiazomethane removes the need for specialized storage facilities and expensive hazard mitigation protocols, thereby lowering the total cost of ownership for the manufacturing process. Furthermore, the use of commodity chemicals like methyl bromopyruvate and 7-bromoindole ensures a stable and resilient supply chain, reducing the risk of production delays caused by the scarcity of niche reagents. The high atom economy of the process also translates to reduced waste disposal costs and a smaller environmental footprint, aligning with corporate sustainability goals and regulatory requirements for green manufacturing. Overall, this technology enables a more agile and cost-competitive production model for high-value indole alkaloids.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by replacing expensive organoboron and organotin reagents with inexpensive, commercially available alternatives, drastically reducing the bill of materials for each production batch. The high yields observed in each step, particularly the 92% yield in the final coupling reaction, minimize material loss and maximize the output per unit of input, further driving down the cost per kilogram of the final product. Additionally, the avoidance of protection-deprotection sequences reduces solvent consumption and energy usage associated with additional reaction and purification steps, contributing to a leaner and more economical manufacturing operation.
- Enhanced Supply Chain Reliability: By relying on widely available starting materials rather than custom-synthesized specialty reagents, the supply chain becomes significantly more robust against market fluctuations and geopolitical disruptions. The simplified reaction conditions, which do not require extreme temperatures or pressures, allow for production in a broader range of manufacturing facilities, increasing supply flexibility and reducing lead times for high-purity indole alkaloids. This reliability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of downstream pharmaceutical customers who depend on consistent API availability.
- Scalability and Environmental Compliance: The inherent safety of the reagents and the simplicity of the workup procedures make this route exceptionally well-suited for commercial scale-up from pilot plant to multi-ton production. The absence of heavy metal contaminants like tin, which are difficult to remove and strictly regulated in pharmaceuticals, simplifies the purification process and ensures compliance with ICH Q3D guidelines for elemental impurities. This ease of purification not only accelerates the release of batches but also reduces the environmental burden associated with heavy metal waste treatment, supporting a sustainable and compliant manufacturing lifecycle.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, providing clarity on its practical application and benefits. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, offering a transparent view of the process capabilities. Understanding these aspects is essential for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this new synthesis route improve safety compared to traditional methods?
A: The patented process eliminates the need for highly toxic reagents like trimethylsilyldiazomethane and cuprous cyanide, as well as pyrophoric substances such as n-butyllithium and tri-n-butyltin hydride, significantly reducing operational hazards and waste disposal costs.
Q: What is the overall yield efficiency of the Luteoride A synthesis described in CN114890932A?
A: The process demonstrates robust efficiency with individual step yields of 87% for oxime formation, 82% for the heteroatom Diels-Alder cyclization, and up to 92-94% for the final Heck coupling, ensuring high material throughput.
Q: Is this synthetic route suitable for large-scale commercial manufacturing?
A: Yes, the route utilizes readily available starting materials like methyl bromopyruvate and 7-bromoindole, avoids complex protection-deprotection sequences, and employs standard catalytic systems, making it highly amenable to kilogram-to-ton scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Luteoride A Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented synthesis route for the production of high-value indole alkaloids and are fully equipped to bring this technology to commercial fruition. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our state-of-the-art facilities are designed to handle complex organic syntheses with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards. We are committed to delivering not just a product, but a comprehensive manufacturing solution that optimizes cost, quality, and speed for our global partners.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can accelerate your development timeline and enhance your competitive edge in the pharmaceutical marketplace.
