Advanced Purification Technology for High-Purity Difluoro Pharmaceutical Intermediates
Advanced Purification Technology for High-Purity Difluoro Pharmaceutical Intermediates
The introduction of fluorine atoms into organic molecules is a cornerstone strategy in modern medicinal chemistry, significantly enhancing metabolic stability, membrane permeability, and bioavailability of drug candidates. However, the synthesis of gem-difluoro compounds often faces a critical bottleneck: the formation of elimination by-products, specifically cyclic olefins, which possess physical properties nearly identical to the target difluoro species. Patent CN115850156A addresses this pervasive challenge by disclosing a novel purification methodology that transforms a difficult separation problem into a simple chemical workup. By leveraging an oxidative post-treatment step, this technology enables the efficient removal of olefinic impurities, delivering high-purity difluoro intermediates essential for the development of next-generation therapeutics. This report analyzes the technical merits and commercial implications of this breakthrough for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of gem-difluoro compounds involves the direct fluorination of ketones using reagents like DAST (Diethylaminosulfur trifluoride). While effective for bond formation, this reaction frequently competes with an elimination pathway, generating cyclic alkene by-products. The fundamental issue lies in the separation; these olefinic impurities exhibit polarity and solubility profiles that are strikingly similar to the desired difluoro products. Consequently, standard purification techniques such as recrystallization often fail to achieve the requisite purity levels for pharmaceutical applications. Manufacturers are frequently forced to rely on preparative chromatography or repeated distillation, processes that are not only cost-prohibitive but also technically challenging to scale. The reliance on such resource-intensive purification steps creates significant bottlenecks in the supply chain, inflating the cost of goods and extending lead times for critical API intermediates.
The Novel Approach
The methodology described in CN115850156A offers a paradigm shift by introducing a chemical differentiation step rather than relying solely on physical separation. After the initial fluorination generates a crude mixture containing both the target difluoro compound and olefin by-products, the process employs a selective oxidation step using agents like potassium permanganate. This oxidant specifically targets the carbon-carbon double bonds of the olefinic impurities, converting them into carboxylic acids or other polar oxygenated species. Unlike the neutral difluoro product, these oxidized by-products become highly soluble in aqueous basic media. This allows for their complete removal through a simple acid-base extraction workup. This approach effectively bypasses the need for complex chromatography, streamlining the workflow and ensuring consistent high purity regardless of the initial elimination ratio.
Mechanistic Insights into Oxidative Purification of Difluoro Compounds
The core innovation of this technology lies in the orthogonal reactivity between the stable C-F bonds of the product and the reactive C=C bonds of the impurities. The process begins with the nucleophilic substitution of the carbonyl oxygen by the fluorinating agent, typically DAST, under controlled low-temperature conditions (0-5°C) to minimize thermal decomposition. However, the acidic nature of the alpha-protons in the intermediate can lead to E2 elimination, forming the problematic alkene. The subsequent purification step exploits the susceptibility of these alkenes to oxidative cleavage. When treated with potassium permanganate in a solvent like acetonitrile under reflux, the alkene undergoes oxidation to form carboxylic acid derivatives. This chemical transformation drastically alters the partition coefficient of the impurity.
Following the oxidation, the reaction mixture is treated with a base, such as sodium hydroxide. The newly formed carboxylic acid impurities are deprotonated to form water-soluble carboxylate salts, while the neutral gem-difluoro compound remains in the organic phase. This distinct difference in solubility facilitates a clean separation during the extraction phase. The robustness of this mechanism is evidenced by its applicability across a wide range of substrates, including those with sensitive functional groups like esters, amines, and boronates, demonstrating that the oxidative conditions are sufficiently mild to preserve the integrity of the target molecule while aggressively removing the specific olefinic contaminants.
How to Synthesize 4,4-Difluoropiperidine Derivatives Efficiently
The practical implementation of this purification strategy is best illustrated through the synthesis of 4,4-difluoropiperidine derivatives, which are valuable scaffolds in medicinal chemistry. The protocol initiates by dissolving the ketone precursor, such as tert-butyl 4-oxo-1-piperidinecarboxylate, in anhydrous dichloromethane. Under strict temperature control in an ice bath, DAST is added dropwise to maintain the reaction temperature between 0°C and 5°C, preventing exothermic runaway and minimizing side reactions. After stirring for approximately 1.5 hours, the reaction is quenched carefully with saturated sodium bicarbonate solution. The organic layer is separated, washed, dried over anhydrous magnesium sulfate, and concentrated to yield the crude fluorinated material, which still contains the olefinic impurities.
- React the carbonyl-containing precursor with DAST (1: 2 to 1:3 molar ratio) in DCM at 0-5°C for 1-2 hours to generate the crude difluorinated product.
- Quench the reaction with saturated sodium bicarbonate, extract with DCM, dry over magnesium sulfate, and concentrate to isolate the crude material containing olefin by-products.
- Reflux the crude product in acetonitrile with potassium permanganate (0.1 to 0.3 eq) for 2.5-4.5 hours to oxidize olefins into water-soluble acids, followed by base neutralization and extraction.
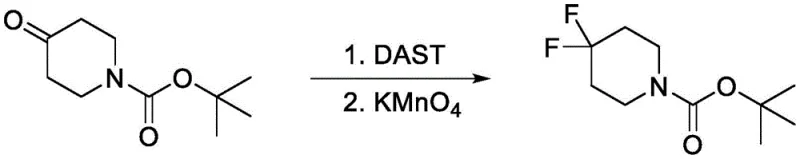
The critical purification phase involves dissolving this crude residue in acetonitrile and adding potassium permanganate (typically 0.2 equivalents relative to the substrate). The mixture is heated to reflux for about 3 hours, allowing the oxidant to fully convert the alkene by-products. Monitoring via LCMS confirms the disappearance of the olefin peaks. Once conversion is complete, the mixture is cooled, and a base like 2M sodium hydroxide is added to neutralize the acidic oxidation products. A final extraction with dichloromethane isolates the pure difluoro compound in the organic layer, which is then dried and concentrated to afford the final product with high purity and yield, ready for downstream coupling reactions.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this purification technology translates directly into enhanced operational efficiency and risk mitigation. The primary advantage is the elimination of preparative chromatography, a step that is notoriously difficult to scale and expensive to operate due to high solvent consumption and silica gel costs. By replacing this with a simple reflux and extraction sequence, the process significantly reduces the consumption of hazardous solvents and solid waste, aligning with green chemistry principles and reducing disposal costs. Furthermore, the use of commodity chemicals like potassium permanganate ensures that reagent costs remain low and supply is stable, avoiding the volatility associated with specialized purification resins or columns.
- Cost Reduction in Manufacturing: The streamlined workflow drastically lowers the cost of goods sold (COGS) by removing the most expensive unit operation in small molecule synthesis. The avoidance of chromatography not only saves on direct material costs but also reduces labor hours and equipment occupancy time. The high yields reported (80-90%) further contribute to cost efficiency by maximizing the throughput of raw materials. Additionally, the mild reaction conditions reduce energy consumption compared to high-temperature or high-pressure alternatives, resulting in substantial overall cost savings for large-scale production campaigns.
- Enhanced Supply Chain Reliability: Reliance on complex purification methods often introduces single points of failure in the supply chain, such as column availability or solvent quality issues. This robust chemical purification method relies on widely available, stable reagents, ensuring consistent production capability. The high purity achieved (≥95%) reduces the risk of batch rejection during QC testing, thereby stabilizing delivery schedules. For partners seeking a reliable difluoro compound supplier, this technology guarantees a steady flow of high-quality intermediates without the delays typically associated with troubleshooting difficult separations.
- Scalability and Environmental Compliance: The process is inherently scalable from gram to ton quantities because it utilizes standard reactor equipment (stirred tanks) rather than specialized chromatography columns. The simplified workup reduces the volume of organic waste generated per kilogram of product, facilitating easier compliance with environmental regulations. The ability to handle diverse substrates, including those with boron and nitrogen functionalities, means this single platform technology can be applied to multiple product lines, simplifying facility validation and operator training while maintaining strict environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this purification technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity for R&D teams evaluating this route for their specific projects. Understanding these nuances is critical for assessing the feasibility of integrating this method into existing manufacturing workflows.
Q: How does this purification method improve upon traditional chromatography?
A: Traditional methods struggle to separate difluoro compounds from olefin by-products due to similar polarities, often requiring expensive preparative HPLC. This patent utilizes a chemical differentiation strategy where KMnO4 selectively oxidizes the olefin impurities into carboxylic acids, which are easily removed via aqueous base wash, eliminating the need for complex chromatography.
Q: What is the typical purity and yield achievable with this process?
A: According to the experimental data in patent CN115850156A, this method consistently achieves product purities of ≥95% (often reaching 98%) with isolated yields ranging from 80% to 90% across various substrates including piperidines, cyclohexanes, and boronate esters.
Q: Is this method scalable for industrial production?
A: Yes, the process is highly scalable. It avoids column chromatography, uses inexpensive reagents like potassium permanganate, and operates under mild conditions (ice bath for fluorination, reflux for oxidation), making it suitable for multi-kilogram to ton-scale manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4-Difluoropiperidine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of fluorinated APIs depends on access to high-quality, cost-effective intermediates. Our technical team has extensively evaluated the purification methodology described in CN115850156A and integrated similar oxidative workup strategies into our CDMO operations. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits observed at the bench level are fully realized in manufacturing. Our rigorous QC labs and stringent purity specifications guarantee that every batch of difluoro intermediate meets the exacting standards required for global pharmaceutical registration.
We invite you to collaborate with us to leverage this advanced technology for your pipeline. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific molecule. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized purification processes can accelerate your development timelines and reduce your overall project costs.
