Scalable Synthesis of Chiral Amide Imidazolium Bromide Ionic Liquids for Advanced Applications
Scalable Synthesis of Chiral Amide Imidazolium Bromide Ionic Liquids for Advanced Applications
The landscape of green chemistry and asymmetric synthesis is undergoing a significant transformation, driven by the demand for sustainable, high-performance chiral media. A pivotal advancement in this domain is detailed in patent CN102643236A, which outlines a robust methodology for preparing chiral amide imidazolium bromide ionic liquids directly from natural amino acids. This technology represents a paradigm shift away from petrochemical-derived chiral sources, leveraging the inherent chirality of abundant biological feedstocks to create functionalized ionic liquids. For R&D directors and procurement specialists alike, this approach offers a compelling value proposition: it combines the unique physicochemical properties of ionic liquids—such as negligible vapor pressure and high thermal stability—with the precise stereochemical control offered by amino acid scaffolds. The resulting materials are not merely solvents but active participants in asymmetric induction, opening new avenues for catalytic applications and chiral separations in the pharmaceutical and fine chemical sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of chiral ionic liquids has been fraught with challenges related to cost, complexity, and environmental impact. Conventional routes often rely on the modification of expensive chiral auxiliaries or the resolution of racemic mixtures, processes that are inherently inefficient and generate substantial waste. Furthermore, many existing methods require harsh reaction conditions, including extreme temperatures or the use of toxic heavy metal catalysts, which complicate downstream purification and raise safety concerns for industrial scale-up. The reliance on non-renewable starting materials also conflicts with the growing corporate mandates for sustainability and carbon footprint reduction. Consequently, the supply chain for high-purity chiral ionic liquids has historically been constrained by high production costs and limited availability, creating bottlenecks for companies seeking to implement green asymmetric synthesis protocols in their manufacturing pipelines.
The Novel Approach
The methodology disclosed in the patent data introduces a streamlined, three-step synthetic route that effectively circumvents these historical barriers. By utilizing natural amino acids as the foundational chiral building blocks, the process taps into a readily available and cost-effective resource pool. The strategy involves an initial protection step followed by amidation and a final quaternization reaction, all of which proceed under mild and controllable conditions. 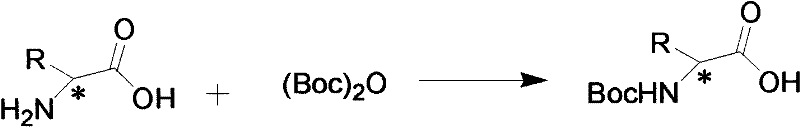 As illustrated in the reaction pathway, the preservation of the chiral center throughout the synthesis ensures that the optical activity of the starting amino acid is faithfully transferred to the final ionic liquid product. This approach not only simplifies the operational workflow but also significantly enhances the atom economy of the process. For a reliable pharmaceutical intermediates supplier, adopting such a route translates to a more resilient supply chain capable of delivering high-quality chiral materials with reduced lead times and lower environmental liability.
As illustrated in the reaction pathway, the preservation of the chiral center throughout the synthesis ensures that the optical activity of the starting amino acid is faithfully transferred to the final ionic liquid product. This approach not only simplifies the operational workflow but also significantly enhances the atom economy of the process. For a reliable pharmaceutical intermediates supplier, adopting such a route translates to a more resilient supply chain capable of delivering high-quality chiral materials with reduced lead times and lower environmental liability.
Mechanistic Insights into Amino Acid-Derived Ionic Liquid Synthesis
The core of this technology lies in the strategic functionalization of the amino acid backbone to create a cationic imidazolium species paired with a bromide anion. The mechanism begins with the protection of the amino group using di-tert-butyl dicarbonate ((Boc)2O), forming a stable N-Boc-amino acid intermediate. This step is crucial as it prevents unwanted side reactions at the nitrogen center during subsequent coupling. Following protection, the carboxylic acid moiety is activated and coupled with 3-bromopropylamine hydrobromide. This amidation reaction creates a flexible linker that connects the chiral center to the future cationic headgroup. The final and most critical step is the nucleophilic substitution where the terminal bromine atom on the propyl chain is displaced by the nitrogen of methylimidazole. This quaternization reaction generates the imidazolium ring, locking the structure into its ionic liquid state. The entire sequence is designed to maintain the integrity of the stereocenter, ensuring that the resulting liquid exhibits distinct optical rotation properties essential for chiral applications.
From a purity and quality control perspective, the choice of reagents and solvents plays a vital role in minimizing impurity profiles. The use of common organic solvents such as dichloromethane, acetonitrile, and ethyl acetate allows for efficient extraction and crystallization techniques to remove unreacted starting materials and by-products. The patent data indicates that the final products are light yellow viscous liquids with specific optical rotations, confirming the successful retention of chirality. 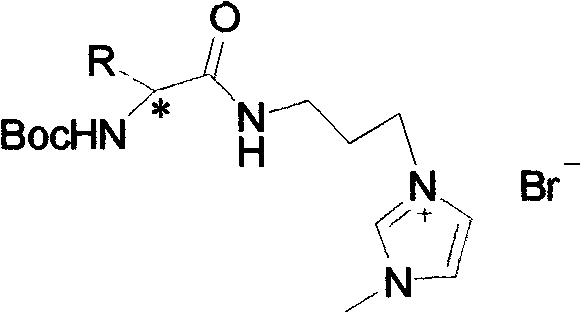 Understanding this mechanistic pathway is essential for high-purity pharmaceutical intermediates manufacturers, as it highlights the critical control points where process parameters such as temperature and stoichiometry must be tightly regulated to prevent racemization or degradation. The robustness of the Boc protecting group under the reaction conditions further ensures that the amino acid side chains remain intact, allowing for the diversification of the ionic liquid library by simply swapping the input amino acid.
Understanding this mechanistic pathway is essential for high-purity pharmaceutical intermediates manufacturers, as it highlights the critical control points where process parameters such as temperature and stoichiometry must be tightly regulated to prevent racemization or degradation. The robustness of the Boc protecting group under the reaction conditions further ensures that the amino acid side chains remain intact, allowing for the diversification of the ionic liquid library by simply swapping the input amino acid.
How to Synthesize Chiral Amide Imidazolium Bromide Efficiently
Implementing this synthesis requires a disciplined approach to reaction monitoring and workup procedures to ensure consistent quality. The process is divided into three distinct stages: Boc-protection, amidation, and quaternization. Each stage has specific temperature windows and molar ratios that optimize yield and purity. For instance, the initial protection is best conducted at 0-30°C to balance reaction rate with selectivity, while the final quaternization benefits from reflux conditions in acetonitrile to drive the reaction to completion. Detailed standard operating procedures regarding reagent addition rates, stirring speeds, and purification methods are critical for reproducibility.
- Protect natural amino acids with di-tert-butyl dicarbonate to form N-Boc-amino acids.
- React the protected amino acid with 3-bromopropylamine hydrobromide to generate the bromo-propionamide intermediate.
- Perform quaternization with methylimidazole in acetonitrile to yield the final chiral ionic liquid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of this patented technology are profound. The shift from synthetic chiral precursors to natural amino acids fundamentally alters the cost structure of producing chiral ionic liquids. Amino acids are commodity chemicals produced on a massive global scale, meaning their prices are stable and generally low compared to specialized chiral ligands. This raw material advantage translates directly into cost reduction in pharmaceutical intermediates manufacturing. Furthermore, the high yields reported in the patent examples—often exceeding 90% in the final step—mean that less raw material is wasted, and the throughput per batch is maximized. This efficiency reduces the overall cost of goods sold (COGS) and improves the margin profile for downstream applications.
- Cost Reduction in Manufacturing: The elimination of expensive chiral catalysts and the use of inexpensive, renewable feedstocks drastically lower the entry barrier for producing these advanced materials. By avoiding complex resolution steps or precious metal catalysts, the process removes significant cost drivers associated with traditional asymmetric synthesis. Additionally, the ability to recycle the ionic liquid due to its low volatility further enhances the long-term economic viability for end-users, creating a closed-loop system that minimizes solvent purchase costs over the lifecycle of the process.
- Enhanced Supply Chain Reliability: Sourcing natural amino acids is far less risky than sourcing specialized fine chemicals, as the former benefits from a diversified global supply base. This diversity insulates the supply chain from disruptions caused by single-source dependencies or geopolitical instability. Moreover, the synthetic route is straightforward and does not require exotic equipment, allowing for flexible manufacturing across multiple sites. This flexibility ensures reducing lead time for high-purity chiral ionic liquids, enabling faster response to market demands and more agile inventory management strategies for downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The process operates under mild conditions and utilizes solvents that are well-understood in terms of waste management and regulatory compliance. The absence of heavy metals simplifies the wastewater treatment process and reduces the burden of hazardous waste disposal. From a commercial scale-up of complex fine chemicals perspective, the linear nature of the synthesis allows for easy transition from laboratory gram-scale to multi-ton production without significant re-engineering of the process flow. This scalability ensures that supply can grow in tandem with demand, supporting long-term strategic partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these chiral ionic liquids. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing production workflows.
Q: What are the primary advantages of using natural amino acids for ionic liquid synthesis?
A: Using natural amino acids provides a renewable, inexpensive chiral pool, eliminating the need for complex asymmetric catalysis while ensuring high optical purity in the final ionic liquid product.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process utilizes mild reaction conditions (0-30°C for initial steps) and common solvents like dichloromethane and acetonitrile, making it highly scalable and safe for commercial manufacturing.
Q: What are the typical yields for the final quaternization step?
A: According to patent data, the final quaternization step consistently achieves high yields ranging from 91% to 98%, depending on the specific amino acid side chain used.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Ionic Liquid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green chiral technologies in modern drug discovery and manufacturing. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of chiral ionic liquid meets the exacting standards required for asymmetric synthesis and chiral separation applications. Our infrastructure is designed to handle the specific solvent systems and temperature controls necessary for this amino acid-based synthesis, providing a secure and compliant manufacturing environment.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific chemical challenges. Whether you require custom synthesis or bulk supply, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your R&D outcomes and your bottom line. Together, we can advance the frontiers of green chemistry while securing a sustainable and cost-effective supply chain for your critical intermediates.
