Advanced Synthesis of 3-Hydroxymethyl-9-Substituted Carbazole for High-Performance Pharmaceutical and Material Applications
Advanced Synthesis of 3-Hydroxymethyl-9-Substituted Carbazole for High-Performance Pharmaceutical and Material Applications
The chemical landscape for functionalized heterocyclic compounds is constantly evolving, driven by the demand for high-purity intermediates in the pharmaceutical and advanced material sectors. A significant breakthrough in this domain is documented in patent CN110003089B, which details a highly efficient preparation method for 3-hydroxymethyl-9-substituted carbazole. This compound class serves as a pivotal building block for developing novel antibacterial agents and fluorescent materials, particularly in the realm of organic light-emitting diodes (OLEDs). The patented methodology addresses longstanding challenges in regioselectivity and operational complexity, offering a streamlined pathway that utilizes readily available reagents such as paraformaldehyde and sodium ethoxide. By leveraging a unique solvent system involving dimethyl sulfoxide (DMSO) and absolute ethanol, this process achieves exceptional yields under remarkably mild conditions. For industry leaders seeking a reliable pharmaceutical intermediate supplier, understanding the nuances of this synthesis is crucial for optimizing supply chains and ensuring the consistent quality of downstream products.
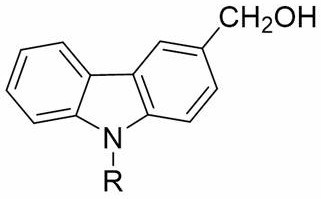
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of a hydroxymethyl group onto the carbazole scaffold has been fraught with technical difficulties that hinder large-scale production. Traditional aldehyde-alcohol reduction methods often necessitate harsh experimental conditions, including extreme temperatures and the use of aggressive reducing agents that pose significant safety risks in a manufacturing environment. Furthermore, these conventional routes frequently suffer from poor regioselectivity, leading to complex mixtures of isomers that are difficult and costly to separate. The reliance on expensive catalysts or multi-step protection-deprotection sequences further inflates the production cost, making the final API or material intermediate economically unviable for many applications. Additionally, the generation of hazardous waste streams from these older processes creates substantial environmental compliance burdens, complicating the disposal protocols required for modern chemical facilities. Consequently, there has been a persistent industry need for a more robust, selective, and environmentally benign synthetic strategy.
The Novel Approach
The methodology outlined in patent CN110003089B represents a paradigm shift by utilizing a direct hydroxymethylation strategy that bypasses the need for complex reduction steps. This novel approach employs paraformaldehyde as a stable and inexpensive source of formaldehyde, reacting directly with N-alkylcarbazole in the presence of sodium ethoxide. The reaction is conducted in a polar aprotic solvent system (DMSO) mixed with absolute ethanol, which facilitates the dissolution of reactants and stabilizes the transition states involved in the electrophilic substitution. One of the most striking features of this process is its incredible speed; the reaction reaches completion in merely 3 minutes under ice-salt bath cooling, drastically reducing energy consumption compared to refluxing methods. The workup procedure is equally elegant, involving a simple acid quench followed by water precipitation and recrystallization, which eliminates the need for column chromatography or extensive solvent exchanges. This simplicity translates directly into cost reduction in pharmaceutical intermediate manufacturing, as it minimizes labor hours and solvent usage while maximizing throughput.
Mechanistic Insights into Base-Catalyzed Hydroxymethylation
To fully appreciate the technical superiority of this route, one must delve into the mechanistic underpinnings of the base-catalyzed electrophilic aromatic substitution. The carbazole nucleus possesses a rigid conjugated planar structure, and the presence of the nitrogen atom at the 9-position significantly enhances the electron density of the aromatic rings, particularly at the 3 and 6 positions. In this specific transformation, sodium ethoxide acts as a strong base that likely activates the paraformaldehyde or generates a reactive methylene species in situ. The electron-rich nature of the N-alkylcarbazole ring makes it highly susceptible to attack by this electrophilic species. The steric environment created by the N-alkyl group directs the substitution preferentially to the 3-position, avoiding the sterically hindered 1, 4, 5, and 8 positions. This inherent regioselectivity is a key factor in achieving the high product purity reported in the patent data, as it suppresses the formation of unwanted positional isomers that typically plague Friedel-Crafts type alkylations.
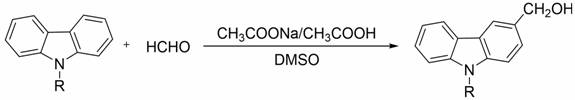
Furthermore, the choice of DMSO as the primary solvent plays a critical role in stabilizing the ionic intermediates formed during the reaction cycle. DMSO is known for its ability to solvate cations effectively while leaving anions relatively naked and reactive, which accelerates the nucleophilic or electrophilic interactions depending on the specific mechanism variant. The rapid reaction time of 3 minutes suggests a low activation energy barrier, likely due to the high reactivity of the in situ generated electrophile and the activated carbazole substrate. Impurity control is inherently managed by the mild conditions; since the reaction is stopped quickly with concentrated hydrochloric acid once the conversion is complete, there is minimal opportunity for over-reaction or polymerization of the formaldehyde. This precise control over the reaction endpoint ensures that the impurity profile remains clean, simplifying the subsequent recrystallization step and ensuring that the final high-purity OLED material or pharmaceutical precursor meets stringent quality specifications without extensive downstream processing.
How to Synthesize 3-Hydroxymethyl-9-Substituted Carbazole Efficiently
Implementing this synthesis in a pilot or production plant requires adherence to specific operational parameters to replicate the high yields observed in the patent examples. The process begins with the preparation of the base solution, where precise molar ratios of sodium ethoxide to paraformaldehyde (1:1.5) are maintained to ensure complete consumption of the carbazole substrate. The use of an ice-salt bath is non-negotiable, as it controls the exotherm associated with the rapid addition of the N-alkylcarbazole DMSO solution. Following the brief reaction period, the quenching step with concentrated hydrochloric acid must be performed carefully to neutralize the base and protonate any alkoxide intermediates, facilitating the precipitation of the product upon the addition of distilled water. The detailed standardized synthesis steps, including specific solvent volumes and stirring rates for scale-up, are provided in the guide below.
- Prepare the reaction mixture by dissolving paraformaldehyde and sodium ethoxide in a DMSO and absolute ethanol solvent system under ice-salt bath cooling.
- Rapidly add the N-alkylcarbazole substrate dissolved in DMSO to the reaction vessel and stir for approximately 3 minutes.
- Quench the reaction with concentrated hydrochloric acid, precipitate the product with distilled water, and purify via recrystallization with absolute ethanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond mere technical feasibility. The shift from complex, multi-step reductions to a single-pot hydroxymethylation significantly streamlines the manufacturing workflow, reducing the number of unit operations required. This simplification directly correlates to a reduction in capital expenditure for equipment and a decrease in the overall footprint of the production facility. Moreover, the use of commodity chemicals like paraformaldehyde and sodium ethoxide ensures a stable and predictable raw material supply, mitigating the risks associated with sourcing exotic or regulated reagents. The robustness of the process also implies a lower rate of batch failures, enhancing the reliability of supply for downstream customers who depend on just-in-time delivery models for their own production schedules.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the elimination of expensive transition metal catalysts and the reduction in solvent consumption. By avoiding the need for chromatographic purification and relying instead on simple filtration and recrystallization, the operational costs associated with solvent recovery and waste disposal are drastically lowered. The short reaction time of 3 minutes allows for higher throughput in existing reactor vessels, effectively increasing the annual production capacity without the need for new infrastructure investments. Additionally, the high yields reported across various alkyl substituents mean that less raw material is wasted, optimizing the atom economy of the process and further driving down the cost per kilogram of the final active ingredient.
- Enhanced Supply Chain Reliability: The simplicity of the raw material list contributes significantly to supply chain resilience. Paraformaldehyde and sodium ethoxide are widely produced bulk chemicals with established global supply networks, reducing the likelihood of shortages that can occur with specialized reagents. The mild reaction conditions also reduce the wear and tear on processing equipment, leading to less unplanned downtime for maintenance. Furthermore, the ability to produce a wide range of derivatives (from methyl to hexadecyl substituents) using the same core protocol allows manufacturers to respond flexibly to changing market demands without retooling entire production lines, ensuring a continuous flow of diverse intermediates to clients.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method offers a greener alternative to traditional synthesis routes. The absence of heavy metals and harsh reducing agents simplifies the treatment of effluent streams, making it easier to meet increasingly stringent environmental regulations. The use of DMSO, while requiring careful handling, is manageable within standard industrial hygiene frameworks, and the aqueous workup minimizes the volume of organic waste generated. The process is inherently scalable, as demonstrated by the straightforward transition from laboratory glassware to larger reactors, ensuring that the commercial scale-up of complex pharmaceutical intermediates can be achieved with minimal technical risk and maximum regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable reference for technical teams evaluating the process for adoption. Understanding these specifics is essential for ensuring that the transfer of technology from the lab to the plant floor is executed smoothly and efficiently.
Q: What are the critical reaction conditions for this hydroxymethylation?
A: The process requires strict temperature control using an ice-salt bath and a specific molar ratio of sodium ethoxide to paraformaldehyde (1:1.5) to ensure high selectivity at the 3-position.
Q: How is the product purified to achieve high purity?
A: After quenching with acid and precipitation with water, the crude solid is recrystallized using absolute ethanol, which effectively removes impurities and yields a high-purity crystalline product.
Q: Can this method be applied to long-chain alkyl substituents?
A: Yes, the patent demonstrates successful application across a wide range of N-alkyl groups, including methyl, ethyl, propyl, and long-chain variants like dodecyl and hexadecyl, maintaining high yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hydroxymethyl-9-Substituted Carbazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final products. Our team of expert chemists has thoroughly analyzed the pathway described in CN110003089B and is fully equipped to execute this synthesis with precision and consistency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the clinical trial phase or full-scale manufacturing. Our state-of-the-art facilities are designed to handle sensitive reactions under strict temperature control, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of 3-hydroxymethyl-9-substituted carbazole meets your exacting standards.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. By leveraging our expertise in process chemistry, we can provide a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our commitment to quality and efficiency can support your long-term business goals and accelerate your time to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
