Advanced Asymmetric Synthesis of 2-Azabicyclo[3.1.0]hexane Derivatives for Commercial Pharma Production
The global demand for direct-acting antivirals targeting Hepatitis C Virus (HCV) has driven intense research into efficient synthetic routes for key bicyclic intermediates. Patent CN103450071A introduces a groundbreaking asymmetric synthesis method for 2-azabicyclo[3.1.0]hexane-2,3-dicarbonate tert-butyl ester, a critical scaffold in the production of next-generation protease inhibitors. This technology addresses the urgent need for reliable pharmaceutical intermediate suppliers who can deliver high-purity materials with consistent stereochemical integrity. By shifting away from legacy methodologies that rely on hazardous reagents and convoluted step sequences, this innovation offers a streamlined pathway that aligns perfectly with modern green chemistry principles and cost-efficiency mandates in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 2-azabicyclo[3.1.0]hexane core has been plagued by significant synthetic inefficiencies and safety concerns. Early approaches, such as the method published by Hanessian et al., utilized organotin reagents like Me3SnCH2 in conjunction with strong bases like LiHMDS to generate carbenoids. As illustrated in the reaction scheme below, this pathway often resulted in complex mixtures of diastereomers requiring difficult separations and generating toxic tin waste that is costly to dispose of in compliance with environmental regulations.
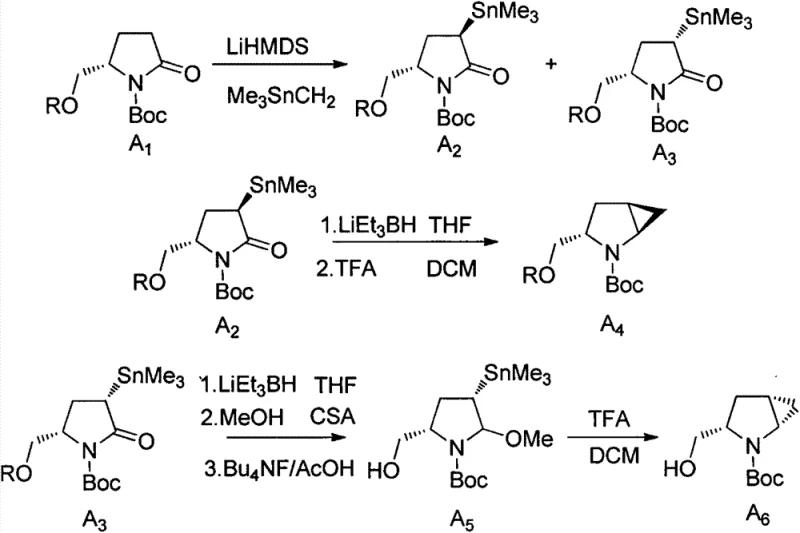
Furthermore, subsequent improvements reported in patents like US20050090539 and WO2011075439A1 attempted to utilize diethylzinc but suffered from poor stereoselectivity or relied on expensive and unstable cyclopropanation agents such as chloroiodomethane (ICH2Cl). These limitations translated directly into higher production costs and lower overall yields, creating bottlenecks for procurement managers seeking cost reduction in API manufacturing. The necessity for cryogenic conditions combined with sensitive reagents often made these processes fragile and difficult to reproduce reliably on a multi-kilogram scale, posing risks to supply chain continuity.
The Novel Approach
The methodology disclosed in CN103450071A represents a paradigm shift by employing a robust diethylzinc-mediated cyclopropanation using stable and cost-effective dihalomethanes such as diiodomethane, dibromomethane, or chlorobromomethane. This approach allows for the direct conversion of chiral pyrroline precursors, specifically (5R)2 or (5S)2, into the desired bicyclic structures with exceptional efficiency. The reaction proceeds smoothly in common solvents like toluene or benzene at temperatures between -25°C and -10°C, avoiding the extreme conditions required by previous methods.
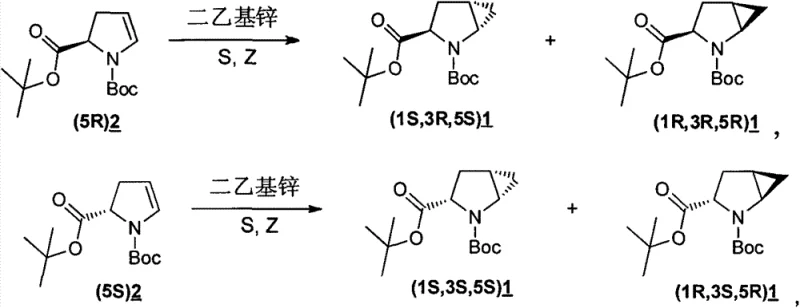
Crucially, this novel route achieves total yields greater than 70%, a substantial improvement over the often mediocre yields of prior art. The process generates a major diastereomer alongside a minor isomer, which can be easily separated via standard column chromatography using a petroleum ether and ethyl acetate system. This simplicity in downstream processing significantly reduces the time and resources required for purification, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates where throughput and purity are paramount.
Mechanistic Insights into Diethylzinc-Mediated Asymmetric Cyclopropanation
The core of this synthesis lies in the formation of a zinc carbenoid species in situ, which acts as the active cyclopropanating agent. When diethylzinc reacts with dihalomethanes (Z), it generates an iodomethylzinc or bromomethylzinc intermediate that is electrophilic enough to attack the electron-rich double bond of the pyrroline ring. The presence of the Boc-protected nitrogen and the existing chiral center at the 2-position of the pyrroline ring exerts a powerful directing effect, guiding the approach of the carbenoid to one face of the alkene preferentially. This substrate-controlled diastereoselection is the key to obtaining the specific (1R,3R,5R) or (1S,3S,5S) configurations required for biological activity in HCV drugs.
From an impurity control perspective, the reaction is remarkably clean, primarily producing only two diastereomeric products rather than a complex soup of regioisomers. The minor isomer, while structurally similar, possesses distinct physical properties such as melting point and polarity, allowing for effective removal during the purification stage. For R&D directors focused on purity specifications, understanding that the reaction mechanism inherently limits the formation of unrelated byproducts provides confidence in the robustness of the analytical profile. The use of mild quenching agents like citric acid further ensures that sensitive functional groups remain intact, preserving the integrity of the final high-purity OLED material or pharmaceutical intermediate.
How to Synthesize 2-Azabicyclo[3.1.0]hexane Derivatives Efficiently
Implementing this synthesis requires careful attention to reagent quality and temperature control to maximize the diastereomeric ratio. The process begins with the dissolution of the chiral starting material in an inert solvent under a nitrogen atmosphere to prevent moisture degradation of the organozinc species. Following the addition of the dihalomethane, the mixture is cooled to the optimal range before the slow addition of diethylzinc, which initiates the cyclopropanation. Detailed standardized synthetic steps see the guide below.
- Dissolve the chiral pyrroline starting material ((5R)2 or (5S)2) in a suitable solvent such as toluene or benzene under nitrogen protection.
- Add the cyclopropanation reagent (e.g., diiodomethane or dibromomethane) and cool the mixture to a temperature range between -25°C and -10°C.
- Slowly add diethylzinc solution dropwise, maintain reaction for 19-24 hours, then quench with citric acid and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals and supply chain heads, the transition to this synthetic route offers tangible strategic benefits beyond mere chemical elegance. The elimination of toxic tin reagents removes a significant regulatory burden and waste disposal cost, directly contributing to cost reduction in pharmaceutical intermediate manufacturing. Additionally, the reagents employed—diethylzinc and dihalomethanes—are commodity chemicals with stable global supply chains, reducing the risk of raw material shortages that can plague specialty reagent-dependent processes.
- Cost Reduction in Manufacturing: The simplified workflow eliminates multiple isolation and purification steps associated with older tin-based methods, leading to substantial cost savings in labor and solvent consumption. By achieving higher yields in a single pot, the overall material throughput is increased, meaning less starting material is wasted per kilogram of final product. The avoidance of expensive and unstable reagents like chloroiodomethane further lowers the bill of materials, allowing for more competitive pricing in long-term supply contracts without sacrificing margin.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted production schedules for downstream API manufacturers. Since the process does not rely on exotic catalysts or hard-to-source chiral auxiliaries, the risk of supply disruption is minimized. This reliability allows supply chain planners to forecast inventory needs with greater accuracy and reduce safety stock levels, optimizing working capital while ensuring timely delivery to clients.
- Scalability and Environmental Compliance: The use of standard industrial solvents and manageable cryogenic temperatures makes this process highly amenable to scale-up from pilot plant to full commercial production. The reduced generation of hazardous heavy metal waste aligns with increasingly strict environmental regulations, facilitating easier permitting and operation in regulated jurisdictions. This environmental compatibility enhances the long-term sustainability of the supply source, protecting partners from future regulatory shocks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on performance metrics and operational feasibility.
Q: What are the key advantages of this diethylzinc-mediated cyclopropanation over traditional methods?
A: This method eliminates the need for toxic organotin reagents and complex multi-step sequences found in older protocols like the Hanessian route. It utilizes readily available dihalomethanes and diethylzinc to achieve direct cyclopropanation with yields exceeding 70%, significantly simplifying the purification process and reducing environmental hazards associated with heavy metal waste.
Q: How does the process control stereoselectivity for the four possible isomers?
A: The stereoselectivity is inherently controlled by the chirality of the starting pyrroline substrate, either (5R)2 or (5S)2. The reaction conditions allow for the preferential formation of specific diastereomers such as (1R,3R,5R)1 or (1S,3S,5S)1 as the major products, while minor isomers can be effectively separated through standard silica gel column chromatography, ensuring high optical purity for downstream drug synthesis.
Q: Is this synthesis method suitable for large-scale industrial manufacturing?
A: Yes, the process is highly scalable due to its operational simplicity and the use of common industrial solvents like toluene. The reaction operates at manageable cryogenic temperatures (-25°C to -10°C) which are achievable with standard industrial cooling systems, and the workup involves simple aqueous washes and crystallization or chromatography, making it robust for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Azabicyclo[3.1.0]hexane Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful development of antiviral therapies depends on the availability of high-quality building blocks. Our technical team has extensively evaluated the pathway described in CN103450071A and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-azabicyclo[3.1.0]hexane intermediate meets the exacting standards required for clinical and commercial applications.
We invite you to collaborate with us to leverage this advanced synthetic technology for your pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing capabilities can accelerate your time to market while optimizing your overall cost structure.
