Revolutionizing Rifampicin Production: A Deep Dive into One-Pot Synthetic Efficiency
Introduction to Next-Generation Rifampicin Manufacturing
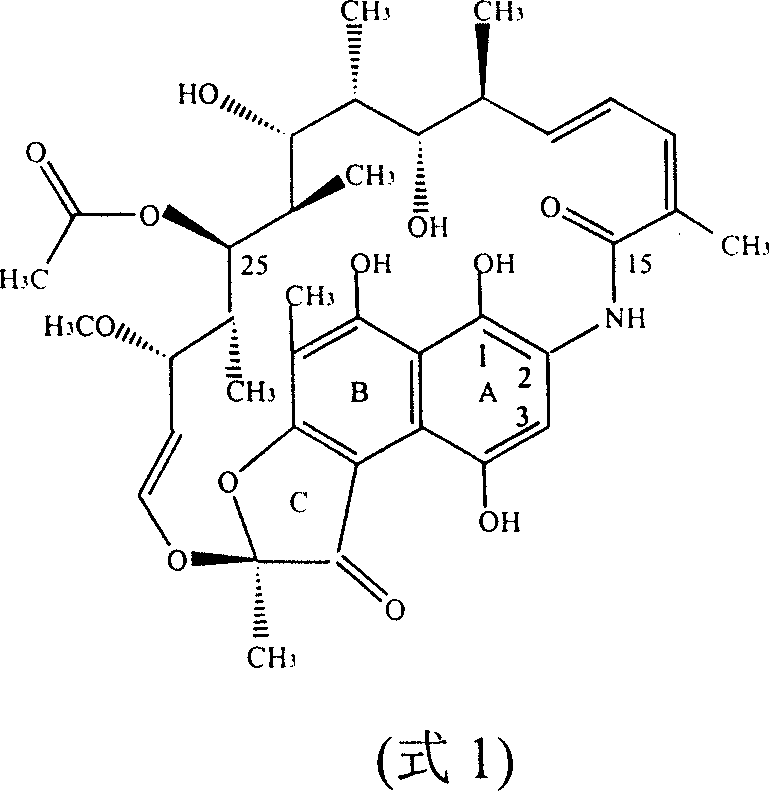
The pharmaceutical landscape for semi-synthetic antibiotics is constantly evolving, driven by the need for higher purity and more sustainable manufacturing processes. Patent CN100383143C introduces a groundbreaking one-pot processing method for synthesizing Rifampicin, a critical antitubercular agent, directly from purified Rifamycin SV. This technology fundamentally addresses the limitations of traditional multi-step isolation procedures by utilizing sodium hypochlorite as a selective oxidizing agent to convert Rifamycin SV into Rifamycin S in situ. Unlike conventional routes that require the crystallization and drying of intermediate sodium salts, this novel approach proceeds directly through cyclization, hydrolysis, and condensation reactions within a unified workflow. The structural complexity of the starting material, Rifamycin Sodium, characterized by its macrocyclic lactam ansa-bridge, demands precise control to prevent degradation, which this patent successfully achieves.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional Rifampicin synthesis is plagued by inefficiencies stemming from the rigorous isolation of intermediates. In standard protocols, Rifamycin SV is oxidized, extracted, and then converted into a sodium salt which must be vacuum-dried at elevated temperatures (80-90°C) for extended periods. This thermal stress, combined with subsequent acidification using concentrated sulfuric acid to regenerate Rifamycin S, inevitably triggers numerous side reactions. Literature indicates that these harsh conditions can lead to impurity levels reaching nearly 20%, primarily due to the hydrolysis of the C25 acetoxy group and oxidative degradation of the sensitive hydroquinone moiety. Furthermore, the reliance on strong inorganic acids and bases necessitates extensive neutralization and waste treatment steps, complicating the supply chain and increasing the environmental footprint of the manufacturing process.
The Novel Approach
The innovative one-pot strategy described in the patent circumvents these pitfalls by maintaining the reaction mixture in a controlled organic phase throughout the critical transformation stages. By employing sodium hypochlorite for oxidation under mild conditions, the process avoids the aggressive acidic environments that typically degrade the ansamycin core. The elimination of the intermediate salt isolation step means there is no need for energy-intensive vacuum drying or the use of corrosive sulfuric acid for acidification. Instead, the Rifamycin S generated in situ undergoes immediate cyclization with dihydroxymethyl tert-butyl amine. This seamless transition not only preserves the integrity of the sensitive molecular scaffold but also drastically reduces the number of unit operations required, thereby streamlining the production timeline and enhancing overall throughput.
Mechanistic Insights into Oxidative Cyclization and Condensation
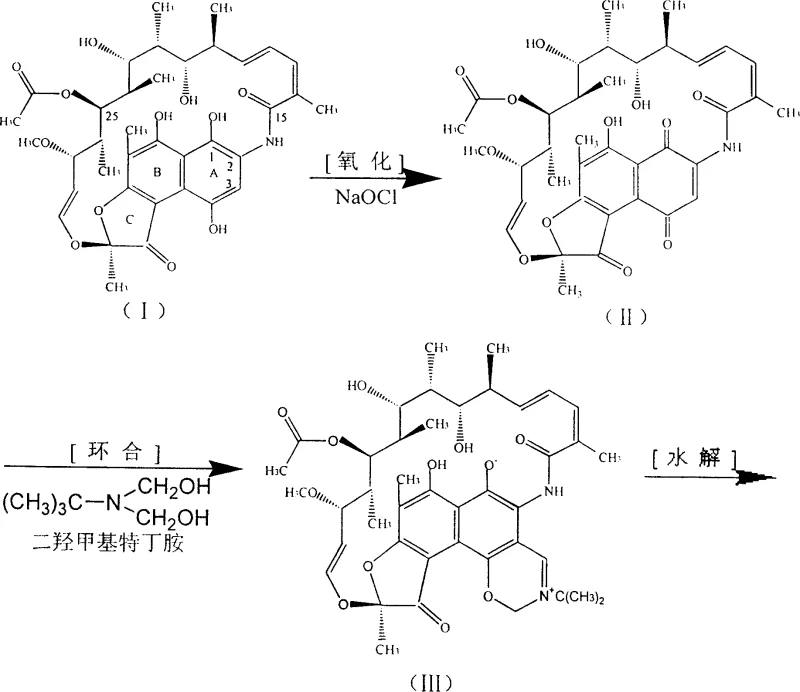
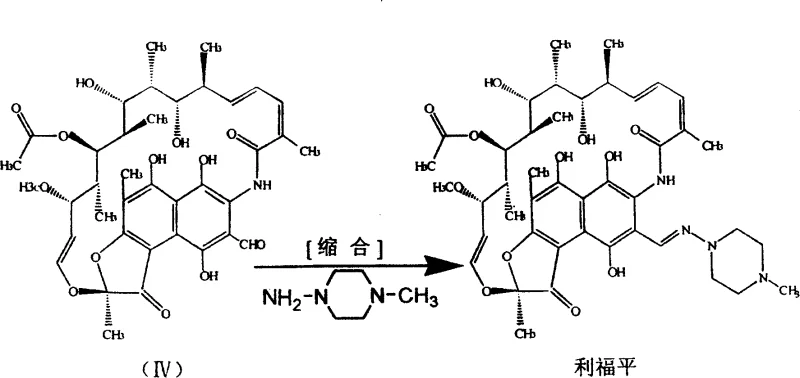
The core of this technological advancement lies in the precise orchestration of the oxidation and cyclization mechanisms. The use of sodium hypochlorite allows for a clean conversion of the hydroquinone system in Rifamycin SV to the quinone form in Rifamycin S without over-oxidation. Following this, the addition of dihydroxymethyl tert-butyl amine facilitates a rapid cyclization to form the N-tert-butyl-1,3-oxazine derivative. Crucially, the subsequent hydrolysis and condensation steps are engineered to occur in a single pot. The introduction of morpholine prior to the addition of 1-methyl-4-aminopiperazine creates a weakly alkaline environment that is optimal for the condensation reaction. This specific chemical environment promotes the formation of the Schiff base linkage at the C3 position while minimizing the hydrolysis of the imine bond, a common failure mode in less controlled systems.
Furthermore, the mechanistic pathway leverages the nucleophilic properties of morpholine to partially substitute the more expensive 1-methyl-4-aminopiperazine during the initial phases of the reaction. This dual-amine strategy ensures that the reaction kinetics favor the desired product formation while suppressing the formation of polymeric byproducts often seen in highly alkaline conditions. The final crystallization is triggered by adjusting the pH to a narrow range of 5.0 to 5.5 using dilute acetic acid, which ensures the precipitation of high-purity Rifampicin crystals. This level of control over the reaction microenvironment is what allows the process to achieve superior quality metrics compared to the erratic outcomes of batch-wise isolation methods.
How to Synthesize Rifampicin Efficiently
Implementing this one-pot synthesis requires careful attention to reagent ratios and temperature control to maximize yield and purity. The process begins with the oxidation of purified Rifamycin SV using a specific weight ratio of sodium hypochlorite solution, followed by the direct addition of the cyclization agent without intermediate workup. The reaction temperature is maintained between 30°C and 60°C to balance reaction rate with stability. For the final condensation step, the sequential addition of morpholine and 1-methyl-4-aminopiperazine is critical to establishing the correct pH profile for imine formation. Detailed standardized operating procedures regarding specific solvent volumes, stirring rates, and crystallization cooling profiles are essential for reproducible commercial success.
- Oxidize purified Rifamycin SV using sodium hypochlorite to form Rifamycin S directly in the organic phase.
- Perform cyclization with dihydroxymethyl tert-butyl amine followed by hydrolysis to generate the 3-formyl intermediate.
- Execute condensation using a mixture of morpholine and 1-methyl-4-aminopiperazine to finalize the Rifampicin structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this one-pot technology offers transformative benefits in terms of cost structure and operational reliability. The most significant advantage is the drastic reduction in energy consumption achieved by eliminating the vacuum drying step for the Rifamycin S sodium salt intermediate. In traditional manufacturing, drying large batches of wet cake at high temperatures represents a substantial portion of the utility budget; removing this step translates directly into lower variable production costs. Additionally, the reduction in the usage of high-cost raw materials, specifically through the partial substitution of 1-methyl-4-aminopiperazine with the more economical morpholine, further enhances the gross margin potential of the final API. These savings are compounded by the reduced need for corrosive chemicals like concentrated sulfuric acid, which lowers maintenance costs for reactor vessels and piping.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation and drying steps removes a major bottleneck in the production schedule, allowing for faster batch turnover and reduced labor hours per kilogram of product. By avoiding the use of large quantities of inorganic acids and bases, the facility also sees a reduction in waste treatment costs and the consumption of neutralization agents. The optimized stoichiometry of the condensation reagents ensures that expensive amines are not wasted in side reactions, leading to a more efficient utilization of raw materials and a lower cost of goods sold.
- Enhanced Supply Chain Reliability: Simplifying the process flow from multiple isolation steps to a continuous one-pot operation significantly reduces the risk of batch failures due to handling errors during transfer and filtration. The robustness of the sodium hypochlorite oxidation system ensures a consistent supply of high-quality intermediate, reducing the variability that often plagues fermentation-derived starting materials. This consistency allows for more accurate demand forecasting and inventory planning, ensuring that downstream formulation teams receive their API shipments on time without unexpected delays caused by reprocessing off-spec intermediates.
- Scalability and Environmental Compliance: The reduced equipment footprint required for this process makes it highly scalable, as fewer reactors are needed to achieve the same output volume compared to the traditional multi-vessel isolation method. The minimization of acidic and alkaline waste streams aligns with increasingly stringent environmental regulations, reducing the liability associated with effluent discharge. Furthermore, the milder reaction conditions decrease the safety risks associated with handling hot, corrosive acids, creating a safer working environment and reducing the potential for operational shutdowns due to safety incidents.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this one-pot Rifampicin synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the operational parameters and quality outcomes. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into existing manufacturing lines.
Q: How does the one-pot method improve Rifampicin purity compared to traditional methods?
A: By eliminating the isolation and vacuum drying of the Rifamycin S sodium salt intermediate, the process avoids thermal degradation and side reactions caused by strong acid acidification, significantly reducing impurity profiles.
Q: What are the cost advantages of using morpholine in the condensation step?
A: Morpholine acts as a cheaper auxiliary base that partially replaces the expensive 1-methyl-4-aminopiperazine, optimizing the reaction environment while lowering raw material costs without compromising yield.
Q: Does this process require specialized equipment for scale-up?
A: No, the process simplifies equipment requirements by removing the need for separate crystallization and drying units for intermediates, allowing for easier commercial scale-up in standard reactor setups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rifampicin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global antibiotic market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the one-pot Rifampicin process are fully realized in a GMP-compliant environment. We are committed to delivering high-purity Rifampicin that meets stringent purity specifications, leveraging our rigorous QC labs to monitor every stage of the synthesis from the initial oxidation to the final crystallization. Our infrastructure is designed to handle complex ansamycin chemistry with the precision required to minimize impurities and maximize yield.
We invite pharmaceutical partners to engage with our technical procurement team to discuss how this innovative manufacturing route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed projection of the potential economic benefits specific to your volume requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions about securing a stable and cost-effective source of this vital antitubercular agent.
