Advanced Synthesis of Guanine Intermediates: A Cost-Effective and Green Manufacturing Strategy
Advanced Synthesis of Guanine Intermediates: A Cost-Effective and Green Manufacturing Strategy
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally sustainable pathways for the production of critical antiviral intermediates. Patent CN111646994A introduces a transformative approach to synthesizing 2,4-diamino-6-hydroxy-5-formamidopyrimidine, a pivotal precursor for guanine and subsequently for lovir-class antiviral drugs such as acyclovir and ganciclovir. This technology departs from the conventional reliance on nucleoside degradation or hazardous catalytic hydrogenation, instead employing a direct reductive acylation strategy using readily available sulfite catalysts. By shifting the synthetic paradigm, this method addresses long-standing pain points regarding atomic economy, waste generation, and operational safety, positioning it as a superior choice for modern reliable pharmaceutical intermediates supplier networks aiming to optimize their supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of guanine and its derivatives has been plagued by inefficient and hazardous processes. Traditional routes often involve the catalytic hydrogenation of nitrosopyrimidines using precious metals like palladium, platinum, or Raney nickel, which not only incur high capital and operational expenditures but also introduce significant safety risks associated with high-pressure hydrogen gas. Furthermore, alternative pathways starting from guanosine suffer from low atom utilization and difficult separation profiles, often yielding complex mixtures that require extensive purification. These legacy methods generate substantial amounts of acidic wastewater and hazardous solid waste, creating a heavy burden on environmental compliance teams and inflating the overall cost of goods sold (COGS) for cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN111646994A utilizes a clever reductive acylation of 2,4-diamino-5-nitroso-6-hydroxypyrimidine directly in a formamide-water system. This innovative route eliminates the need for high-pressure equipment and expensive noble metal catalysts, replacing them with inexpensive and safe sulfite salts like sodium metabisulfite. The process operates under atmospheric pressure with a controlled two-stage heating profile, ensuring high conversion rates while preventing thermal runaway. The result is a streamlined workflow where the target intermediate precipitates directly from the reaction mixture upon cooling, simplifying isolation and dramatically improving the purity profile compared to older techniques.
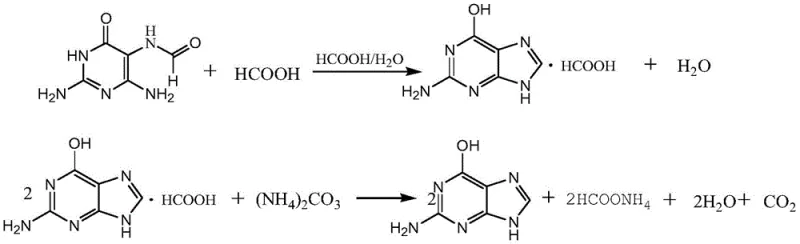
Mechanistic Insights into Sulfite-Catalyzed Reductive Acylation
The core chemical innovation lies in the dual role of the reaction medium and the catalyst. In this system, formamide acts not only as the solvent but also as the source of the formyl group, while the sulfite catalyst facilitates the reduction of the nitroso group to an amine concurrently with the acylation. This tandem transformation avoids the formation of unstable triaminopyrimidine intermediates that are prone to oxidation in air, a common failure point in previous art. The reaction mechanism proceeds through a nucleophilic attack and subsequent rearrangement that is highly selective for the 5-position of the pyrimidine ring, ensuring that regio-isomeric impurities are kept to a minimum. This high selectivity is crucial for maintaining the quality standards required for high-purity pharmaceutical intermediates destined for antiviral API synthesis.
Furthermore, the process design incorporates an elegant waste management strategy where gaseous by-products like ammonia and carbon dioxide are captured in ice water to form ammonium carbonate solution. This by-product stream is not merely waste; it can be directly utilized in the subsequent neutralization step during the conversion of guanine formate to free guanine. This closed-loop logic exemplifies green chemistry principles, turning potential pollutants into valuable reagents. By controlling the reaction temperature in two distinct phases—initially at 70-80°C and then ramping to 105-115°C—the process maximizes the stability of formamide and prevents decomposition, thereby ensuring consistent batch-to-batch reproducibility essential for commercial scale-up of complex pharmaceutical intermediates.
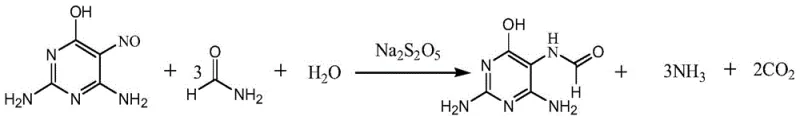
How to Synthesize 2,4-Diamino-6-Hydroxy-5-Formamidopyrimidine Efficiently
Implementing this synthesis requires precise control over stoichiometry and thermal gradients to achieve the reported high yields. The protocol dictates a specific molar ratio of nitrosopyrimidine to formamide and water, typically around 1:3-6:3-15, to balance solubility and reaction kinetics. Operators must adhere to the staged heating regimen to manage the exothermic nature of the reduction while avoiding the evaporation losses of formamide. For a detailed breakdown of the operational parameters, including catalyst loading percentages and crystallization temperatures, please refer to the standardized guide below which outlines the critical process controls necessary for successful execution.
- Dissolve 2,4-diamino-5-nitroso-6-hydroxypyrimidine and a sulfite-based catalyst (e.g., sodium metabisulfite) in a mixture of formamide and water.
- Heat the reaction mixture in two stages: first to 70-80°C to initiate reaction safely, then to 105-115°C to drive completion.
- Cool the solution to crystallize the product, filter, and dry to obtain the target intermediate with over 97% molar yield.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this technology offers compelling advantages that directly impact the bottom line and supply resilience. By removing the dependency on volatile noble metal markets and high-pressure hydrogen infrastructure, manufacturers can stabilize their production costs and reduce exposure to supply chain disruptions. The elimination of hazardous hydrogenation steps also lowers insurance premiums and safety compliance overheads, contributing to a leaner operational model. Additionally, the ability to recycle formic acid in the downstream cyclization step significantly reduces solvent procurement volumes, aligning with both economic and sustainability goals for forward-thinking organizations.
- Cost Reduction in Manufacturing: The substitution of expensive noble metal catalysts (Pd, Pt, Rh) with commodity chemicals like sodium metabisulfite results in a drastic reduction in raw material costs. Furthermore, the simplified work-up procedure, which avoids complex extraction and decolorization steps involving large quantities of activated carbon, reduces labor hours and consumable usage. The high atomic economy of the reaction means less raw material is wasted as by-products, directly enhancing the overall process efficiency and margin potential for bulk producers.
- Enhanced Supply Chain Reliability: The reliance on stable, non-hazardous reagents ensures that production schedules are not subject to the strict regulatory controls and logistics challenges associated with high-pressure hydrogen gas or pyrophoric catalysts. The robustness of the reaction conditions allows for flexible manufacturing across different facilities without requiring specialized high-pressure reactors. This flexibility enables suppliers to respond more agilely to market demand fluctuations, effectively reducing lead time for high-purity pharmaceutical intermediates and ensuring continuity of supply for downstream API manufacturers.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to traditional acid-heavy routes, simplifying wastewater treatment and lowering disposal fees. The capture and utilization of ammonia and carbon dioxide by-products demonstrate a commitment to circular chemistry, which is increasingly valued by global regulators and partners. The straightforward crystallization isolation method scales linearly from pilot to commercial plants, minimizing the technical risks typically associated with process transfer and validation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on yield expectations, purity profiles, and operational safety. Understanding these details is vital for technical teams evaluating the feasibility of adopting this greener alternative for their existing production lines.
Q: How does this new method improve upon traditional guanine synthesis?
A: Unlike traditional methods requiring expensive noble metal catalysts (Pd, Pt, Ni) and high-pressure hydrogenation, this patent utilizes inexpensive sulfite catalysts under atmospheric pressure, significantly reducing safety risks and raw material costs.
Q: What is the purity profile of the intermediate produced?
A: The process yields 2,4-diamino-6-hydroxy-5-formamidopyrimidine with a purity exceeding 99.0% and a molar yield greater than 97%, minimizing the need for complex downstream purification.
Q: Is the formic acid solvent recoverable in the downstream cyclization step?
A: Yes, the subsequent cyclization to guanine is performed in formic acid which can be distilled and recycled directly back into the process, drastically lowering solvent consumption and waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4-Diamino-6-Hydroxy-5-Formamidopyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient intermediate synthesis in the global antiviral supply chain. Our technical team has extensively analyzed the pathway described in CN111646994A and possesses the expertise to implement this green chemistry route at scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the exacting standards required for GMP API synthesis.
We invite procurement leaders and R&D directors to collaborate with us to leverage this cost-effective technology for your projects. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and discuss route feasibility assessments that can drive tangible value and efficiency into your supply chain operations.
