Advanced Copper-Catalyzed Synthesis of Beta-Mercapto Azaphospho Heterocyclic Derivatives for Pharma
Introduction to Patent CN110218229B Technology
The pharmaceutical industry constantly seeks robust synthetic pathways for organophosphorus compounds due to their profound biological activities, ranging from anticancer properties to anti-inflammatory effects. Patent CN110218229B, published in August 2021, introduces a groundbreaking preparation method for beta-mercapto azaphospho heterocyclic derivatives that fundamentally shifts the paradigm from hazardous traditional chemistry to a safer, copper-catalyzed radical addition strategy. This technology leverages readily available alkyne starting materials, phosphorus reagents, and trimethylsilyl isothiocyanate to construct complex phosphorus-sulfur heterocycles under remarkably mild conditions of 30-70°C. For R&D directors and procurement specialists, this patent represents a critical opportunity to access high-purity pharmaceutical intermediates through a route that eliminates the generation of toxic byproducts like carbon disulfide, thereby aligning modern drug discovery with stringent environmental and safety standards while maintaining high yields and operational simplicity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of S,N-bisphosphonates and related heterocyclic structures has relied heavily on the use of benzothiazine-2,4-dithione as a key precursor, a pathway notably disclosed by researchers such as W.M. Abdou. While effective in generating biologically active compounds, this conventional approach suffers from severe drawbacks that hinder its industrial viability and safety profile. The process necessitates harsh reaction conditions and involves the release of carbon disulfide (CS2), a highly volatile and foul-smelling liquid that poses significant health risks by invading the human body through the respiratory tract and skin, potentially causing damage to the nervous system and blood vessels. Furthermore, the raw materials required for these older methods are often difficult to obtain commercially, leading to supply chain bottlenecks, and the reaction routes are typically long with poor selectivity, resulting in lower overall yields and complicated purification processes that increase manufacturing costs and waste generation.
The Novel Approach
In stark contrast, the novel approach detailed in CN110218229B utilizes a modular, multicomponent reaction strategy that begins with the radical addition of phosphorus reagents and trimethylsilyl isothiocyanate to alkynes. This method completely bypasses the need for toxic CS2 generation, replacing it with stable and commercially abundant reagents like phenylacetylene derivatives and dialkyl phosphites. The reaction proceeds efficiently in common solvents such as ethanol, acetonitrile, or toluene at temperatures as low as 30°C, driven by a copper catalyst and peroxide oxidant. This shift not only simplifies the operational workflow but also drastically improves the atom economy and safety of the process. By avoiding the handling of hazardous gases and utilizing a shorter synthetic sequence, this new methodology offers a scalable solution that is ideally suited for the commercial production of complex organophosphorus intermediates required for next-generation therapeutics.
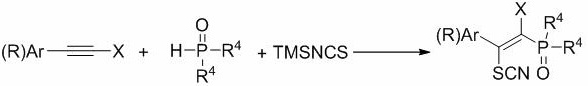
Mechanistic Insights into Copper-Catalyzed Radical Phosphorylation
The core of this technological breakthrough lies in the copper-catalyzed radical mechanism that facilitates the simultaneous introduction of phosphorus and thiocyanato groups across the alkyne triple bond. The reaction initiates with the activation of the peroxide oxidant by the copper catalyst, generating radical species that abstract hydrogen or interact with the phosphorus reagent to form phosphorus-centered radicals. These reactive intermediates then add regioselectively to the alkyne substrate, forming a vinyl radical that is subsequently trapped by the trimethylsilyl isothiocyanate. This cascade ensures high stereocontrol, predominantly yielding the E-isomer of the beta-thiocyanato alkenyl phosphono derivative, as evidenced by the large coupling constants observed in NMR analysis (e.g., J = 19.1 Hz). The versatility of this mechanism allows for tolerance of various functional groups on the alkyne, including electron-withdrawing nitro and ester groups as well as electron-donating methoxy and alkyl substituents, without compromising the integrity of the phosphorus-sulfur bond formation.
Following the initial addition, the subsequent transformation into the final heterocyclic structure involves a carefully orchestrated sequence of reduction and cyclization steps that preserve the sensitive phosphonate moieties. The reduction of the thiocyanato group to a thiocarbonyl functionality using zinc and acetic acid proceeds with exceptional efficiency, often achieving yields near quantitative levels (e.g., 98% in specific examples), which minimizes material loss during intermediate processing. The final cyclization step, mediated by sodium ethoxide or oxidative conditions with iodosobenzene, closes the ring to form the stable azaphospho heterocyclic core. This mechanistic pathway is designed to suppress side reactions such as polymerization or over-oxidation, ensuring that the impurity profile of the final product remains clean and manageable, a critical factor for meeting the rigorous purity specifications demanded by regulatory bodies for pharmaceutical ingredients.
How to Synthesize Beta-Mercapto Azaphospho Derivatives Efficiently
The synthesis of these valuable heterocyclic compounds is achieved through a streamlined four-step protocol that balances chemical complexity with operational ease, making it accessible for both laboratory research and pilot plant operations. The process begins with the dissolution of the alkyne, phosphorus reagent, and trimethylsilyl isothiocyanate in a suitable solvent, followed by the addition of the copper catalyst and peroxide to initiate the radical addition at controlled temperatures. Once the beta-thiocyanato intermediate is isolated, it undergoes reduction and subsequent phosphorylation before the final cyclization yields the target molecule. This standardized approach ensures reproducibility and high quality across different batches, providing a reliable foundation for scaling up production.
- React alkyne, phosphorus reagent, and trimethylsilyl isothiocyanate with a copper catalyst and peroxide at 30-70°C to form beta-thiocyanato alkenyl phosphono derivatives.
- Reduce the thiocyanato intermediate using a zinc catalyst and acetic acid to generate beta-thiocarbonylphosphinyl derivatives.
- Perform phosphorylation using phosphite ester, cuprous trifluoromethanesulfonate, and di-tert-butyl peroxide to obtain beta-thiophosphoryl diphosphonate derivatives.
- Execute final cyclization using sodium ethoxide and hydrochloric acid to yield the target beta-mercapto azaphospho heterocyclic derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers substantial strategic benefits that extend beyond mere chemical novelty. The shift away from hazardous reagents like carbon disulfide eliminates the need for specialized containment equipment and expensive scrubbing systems, directly translating to reduced capital expenditure and lower operational overheads. Additionally, the reliance on commodity chemicals such as alkynes and dialkyl phosphites ensures a stable and diversified supply base, mitigating the risks associated with single-source dependencies on exotic or difficult-to-synthesize starting materials. The mild reaction conditions further contribute to energy efficiency, as the process does not require extreme heating or cryogenic cooling, allowing for more flexible scheduling and reduced utility costs in large-scale manufacturing facilities.
- Cost Reduction in Manufacturing: The elimination of toxic gas handling and the simplification of the synthetic route significantly lower the overall cost of goods sold by reducing waste disposal fees and safety compliance burdens. By avoiding the multi-step degradation of unstable precursors found in older methods, this process maximizes material throughput and minimizes the loss of valuable phosphorus-containing intermediates, leading to substantial cost savings in raw material consumption. Furthermore, the high yields reported in the patent examples indicate that less starting material is required to produce the same amount of final product, enhancing the economic viability of producing these complex intermediates for the global market.
- Enhanced Supply Chain Reliability: The use of widely available alkyne derivatives and standard phosphorus reagents ensures that production schedules are not disrupted by the scarcity of niche chemicals. Since the reaction tolerates a broad range of substrates, manufacturers can easily switch between different alkyne sources or suppliers without needing to re-optimize the entire process, providing greater flexibility in sourcing strategies. This robustness against supply chain volatility is crucial for maintaining continuous production lines and meeting the delivery commitments of downstream pharmaceutical clients who rely on timely access to key building blocks for drug development.
- Scalability and Environmental Compliance: The straightforward workup procedures, which often involve simple filtration or extraction rather than complex distillations, make this process highly amenable to scale-up from kilogram to tonne quantities. The absence of volatile sulfur emissions aligns perfectly with increasingly strict environmental regulations, reducing the risk of regulatory fines and community opposition. This green chemistry profile not only safeguards the environment but also enhances the corporate reputation of manufacturers as responsible stewards of sustainable chemical production, a key differentiator in today's eco-conscious marketplace.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, drawing directly from the experimental data and advantages outlined in the patent documentation. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing manufacturing portfolios.
Q: What are the primary safety advantages of this new synthesis route compared to traditional methods?
A: Unlike conventional methods that utilize benzothiazine-2,4-dithione and release toxic carbon disulfide (CS2) gas, this patented process employs stable alkynes and trimethylsilyl isothiocyanate under mild conditions, significantly eliminating respiratory and vascular toxicity risks associated with volatile sulfur compounds.
Q: Is this copper-catalyzed protocol suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights the method's suitability for industrial production due to its simple reaction operations, mild temperature requirements (30-70°C), and straightforward post-treatment processes involving standard column chromatography or extraction, which facilitates easy scale-up from laboratory to commercial manufacturing.
Q: What types of alkyne substrates are compatible with this phosphorylation strategy?
A: The methodology demonstrates broad substrate scope, successfully accommodating aryl alkynes (such as phenylacetylene and substituted variants), heteroaryl alkynes (like 2-ethynylthiophene and 2-ethynylpyridine), and aliphatic alkynes (including n-hept-1-yne), ensuring versatility for diverse pharmaceutical intermediate synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Mercapto Azaphospho Derivative Supplier
As the demand for specialized organophosphorus intermediates grows in the oncology and anti-inflammatory sectors, partnering with an experienced CDMO becomes essential for navigating the complexities of scale-up and quality assurance. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of beta-mercapto azaphospho derivatives meets the exacting standards required for clinical and commercial applications.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced copper-catalyzed technology for their drug discovery programs. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, along with specific COA data and route feasibility assessments. Let us help you secure a stable supply of high-quality intermediates while optimizing your production costs and accelerating your time to market.
