Revolutionizing Strigolactone GR24 Production: A Scalable Route for Global Agrochemical Supply Chains
Revolutionizing Strigolactone GR24 Production: A Scalable Route for Global Agrochemical Supply Chains
The global demand for advanced plant growth regulators has surged, driven by the need for sustainable agricultural practices and enhanced crop yields. At the forefront of this sector is Strigolactone GR24, a potent synthetic analog of natural strigolactones that plays a critical role in inhibiting shoot branching and stimulating seed germination. However, the widespread adoption of GR24 has historically been hindered by exorbitant costs and complex synthesis protocols. Patent CN103396390A introduces a transformative total synthesis method that addresses these bottlenecks by utilizing inexpensive starting materials such as 1-indanone and 2-methylbutyrolactone. This innovative approach eliminates the need for strict anhydrous and oxygen-free operations in most steps, marking a significant departure from conventional methodologies. For R&D directors and procurement managers seeking a reliable agrochemical intermediate supplier, this technology represents a paradigm shift towards more accessible and economically viable production of high-purity plant hormones.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of strigolactones like GR24 has been plagued by significant technical and economic barriers. The pioneering work by Johnson et al. in 1981 and subsequent modifications by Zwanenburg in 1992 established the foundational chemistry but suffered from severe drawbacks. These traditional routes are notoriously tedious, involving multi-step sequences that require highly toxic reagents and extremely harsh operating conditions. Specifically, the necessity for strict anhydrous environments throughout the synthesis imposes a heavy burden on manufacturing infrastructure, requiring specialized drying equipment and inert gas systems that drive up capital expenditure. Furthermore, the use of hazardous chemicals poses safety risks and complicates waste disposal, creating substantial environmental compliance challenges for modern chemical plants. These factors collectively result in low overall yields and prohibitively high production costs, limiting GR24 to research laboratories rather than broad agricultural application.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN103396390A offers a streamlined and robust alternative designed specifically for industrial feasibility. This novel approach leverages a convergent synthesis strategy where two distinct fragments are prepared independently and then coupled in the final stage. The use of 1-indanone and 2-methylbutyrolactone as primary building blocks ensures a stable and cost-effective supply chain, as these commodities are widely available in the global chemical market. Crucially, the reaction conditions are remarkably mild; apart from the final purification step, the process does not demand rigorous exclusion of moisture or oxygen, drastically simplifying the operational protocol. This simplicity translates directly into cost reduction in agrochemical intermediate manufacturing, as it reduces energy consumption and equipment maintenance while enhancing operator safety. The route is characterized by high atom economy and straightforward workup procedures, making it an ideal candidate for commercial scale-up of complex plant growth regulators.
Mechanistic Insights into the Convergent Synthesis Strategy
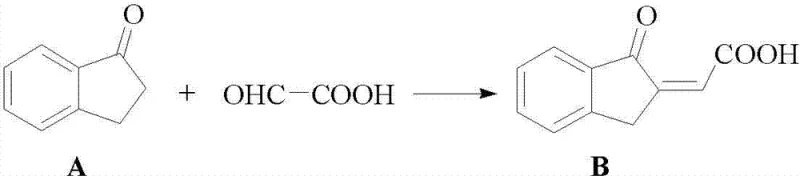
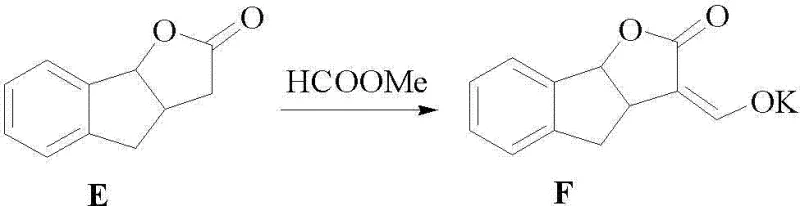
The core of this technological breakthrough lies in the efficient construction of the ABC and D rings of the strigolactone structure through a series of well-controlled transformations. The synthesis begins with the condensation of 1-indanone (Compound A) with glyoxylic acid under strongly acidic conditions to yield Compound B. This step is critical as it establishes the carbon framework necessary for the subsequent ring closures. Following this, Compound B undergoes a reduction using metallic zinc powder in an acidic medium to form Compound C, effectively modifying the oxidation state of the side chain. The process continues with a further reduction using sodium borohydride in an alkaline environment to produce Compound D, which is then subjected to acid-catalyzed lactonization to form the bicyclic lactone structure of Compound E. The final transformation of this fragment involves a condensation reaction with methyl formate in the presence of a potassium alkoxide base to generate the enolate salt, Compound F. This sequence demonstrates a high degree of chemoselectivity, ensuring that functional groups are modified precisely without affecting the integrity of the aromatic system.
Parallel to the preparation of the first fragment, the second fragment is derived from 2-methylbutyrolactone through a dehydrogenation reaction mediated by liquid bromine and red phosphorus at low temperatures ranging from -20°C to -40°C. This yields the unsaturated lactone Compound G, which is subsequently brominated using N-bromosuccinimide (NBS) to afford Compound H. The convergence of these two pathways occurs in the final step, where the nucleophilic enolate salt (Compound F) attacks the electrophilic brominated lactone (Compound H) in a polar aprotic solvent such as N,N-dimethylformamide. This substitution reaction constructs the characteristic enol ether linkage that defines the biological activity of strigolactones. The mechanistic elegance of this route lies in its modularity; by separating the synthesis into two manageable streams, potential side reactions are minimized, and purification is facilitated at intermediate stages. This strategic design is essential for maintaining the high-purity agrochemical intermediates required for consistent biological performance in the field.
How to Synthesize Strigolactone GR24 Efficiently
Implementing this synthesis requires precise control over reaction parameters such as temperature, molar ratios, and pH levels to maximize yield and minimize impurity formation. The patent details specific conditions for each step, such as the use of tetrahydrofuran as a solvent for the initial condensation and the careful adjustment of pH to weak acidity (pH 5-6) during the reduction phases. Operators must adhere to the specified reaction times, which can range from 1 hour to 48 hours depending on the transformation, to ensure complete conversion. While the general procedure is robust, attention to detail in the final coupling step is paramount, as this determines the stereochemistry and overall purity of the final GR24 product. For a comprehensive understanding of the operational parameters, please refer to the standardized synthesis guide below.
- Synthesize Fragment 1 (Compound F) by condensing 1-indanone with glyoxylic acid, followed by reduction, lactonization, and salt formation with methyl formate.
- Prepare Fragment 2 (Compound H) by dehydrogenating 2-methylbutyrolactone to Compound G, followed by bromination with NBS.
- Couple Compound F and Compound H in a non-polar solvent like DMF at room temperature to obtain the final strigolactone GR24 product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers compelling strategic benefits that extend beyond mere technical feasibility. The primary advantage is the drastic simplification of the manufacturing process, which directly correlates with reduced operational expenditures. By eliminating the need for specialized anhydrous reactors and inert atmosphere gloveboxes for the majority of the synthesis, facilities can utilize standard glass-lined or stainless steel reactors, thereby lowering capital investment and maintenance costs. Furthermore, the reliance on commodity chemicals like 1-indanone and 2-methylbutyrolactone mitigates the risk of raw material shortages, ensuring a stable and continuous supply chain even during market fluctuations. This reliability is crucial for long-term planning and contract fulfillment in the volatile agrochemical sector.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the elimination of expensive catalysts and the reduction of energy-intensive drying steps. Traditional methods often require cryogenic conditions and extensive solvent drying, which consume significant amounts of electricity and nitrogen. In contrast, this novel route operates largely at room temperature or moderate reflux conditions, leading to substantial energy savings. Additionally, the high yields reported in the patent examples, such as 99% for the initial condensation and 95% for the salt formation, indicate a highly efficient use of raw materials, minimizing waste generation and disposal costs. The avoidance of toxic reagents also reduces the financial burden associated with hazardous waste treatment and regulatory compliance, further enhancing the overall cost-effectiveness of the production line.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved by the use of widely available starting materials. Unlike specialized precursors that may have single-source suppliers or long lead times, 1-indanone and 2-methylbutyrolactone are produced by multiple manufacturers globally, providing procurement teams with flexibility and bargaining power. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by minor variations in utility quality or environmental conditions. This stability allows for more accurate forecasting and inventory management, reducing lead time for high-purity plant growth regulators and ensuring that customers receive their orders promptly. The ability to scale production without encountering significant technical bottlenecks further strengthens the supply chain, enabling rapid response to increased market demand.
- Scalability and Environmental Compliance: From an environmental perspective, this synthesis aligns well with modern green chemistry principles. The reduction in solvent usage and the avoidance of heavy metal catalysts simplify the effluent treatment process, making it easier to meet stringent environmental regulations. The process generates fewer by-products, and those that are formed are generally easier to separate and dispose of safely. This environmental friendliness not only reduces liability but also enhances the corporate social responsibility profile of the manufacturer. Scalability is inherently built into the design, as the reactions do not exhibit exotherms that are difficult to manage on a large scale. The straightforward workup procedures, such as precipitation and filtration, are easily adapted from laboratory to pilot and finally to commercial scale, ensuring a smooth transition from R&D to full-scale commercial scale-up of complex agrochemical intermediates.
Frequently Asked Questions (FAQ)
Understanding the nuances of this synthesis is vital for stakeholders evaluating its potential for integration into their supply chains. The following questions address common concerns regarding technical implementation, quality assurance, and regulatory compliance. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What are the primary advantages of this new GR24 synthesis method over traditional routes?
A: Unlike the tedious Johnson and Zwanenburg methods which require strict anhydrous conditions and toxic reagents, this novel route utilizes cheap, readily available raw materials like 1-indanone and operates under mild conditions without the need for rigorous oxygen-free environments, significantly simplifying industrial scale-up.
Q: How does this process ensure high product purity for agricultural applications?
A: The process achieves high purity through a series of controlled reactions including specific reduction steps using zinc powder and sodium borohydride, followed by a final purification via column chromatography and recrystallization, ensuring the removal of impurities critical for plant hormone efficacy.
Q: Is this synthetic route suitable for large-scale commercial manufacturing?
A: Yes, the route is explicitly designed for scalability, featuring simple operational steps that do not require specialized anhydrous equipment for most stages, allowing for cost reduction in agrochemical intermediate manufacturing and reliable supply continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Strigolactone GR24 Supplier
As the agricultural sector increasingly relies on sophisticated plant growth regulators to enhance food security, the need for a dependable source of high-quality intermediates has never been greater. NINGBO INNO PHARMCHEM stands ready to support your initiatives with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of the strigolactone synthesis described in CN103396390A, ensuring that every batch meets stringent purity specifications. With our rigorous QC labs and commitment to process optimization, we guarantee the consistency and reliability that global agrochemical companies demand for their critical formulations.
We invite you to explore the potential of this cost-effective synthesis route for your product portfolio. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you secure not just a supplier, but a strategic ally dedicated to driving innovation and efficiency in the global agrochemical market.
