Advanced Electrochemical Synthesis of Pyrazole Compounds for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking more sustainable and efficient pathways for constructing heterocyclic scaffolds, which serve as the backbone of countless bioactive molecules. Patent CN114000167A introduces a groundbreaking electrochemical methodology for the synthesis of pyrazole compounds, addressing critical pain points associated with traditional transition metal catalysis. This innovation utilizes an undivided electrolytic cell to drive the cyclization of benzaldehyde derivatives and p-toluenesulfonylhydrazine, effectively replacing hazardous oxidants and expensive metal catalysts with clean electricity. For R&D directors and procurement managers alike, this represents a significant shift towards greener chemistry that does not compromise on yield or purity. The method operates under mild conditions, typically between 40 to 70°C, and demonstrates robust performance across various substituted substrates, ensuring high versatility for diverse drug discovery programs. By leveraging this technology, manufacturers can achieve substantial cost savings and streamline their supply chains while adhering to increasingly stringent environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
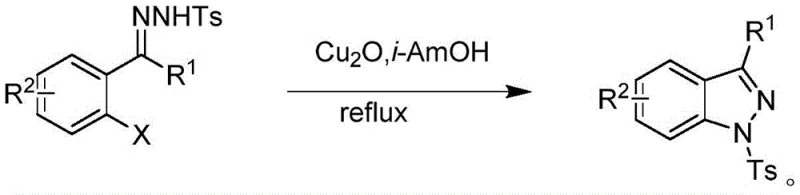
Historically, the construction of the pyrazole core has heavily relied on transition metal catalysis, often utilizing copper or palladium complexes to facilitate the cyclization process. These conventional routes, as illustrated in prior art, frequently necessitate harsh reaction conditions, including high temperatures and the use of stoichiometric amounts of toxic oxidants to drive the reaction to completion. The reliance on heavy metals introduces significant downstream processing challenges, requiring rigorous purification steps to ensure that residual metal levels meet the strict ppm limits mandated by regulatory bodies for pharmaceutical ingredients. Furthermore, the disposal of metal-containing waste streams poses a substantial environmental burden and increases the overall cost of goods sold due to complex waste treatment protocols. These factors collectively create bottlenecks in production scalability and can lead to supply chain disruptions if specific catalyst precursors become scarce or regulated.
The Novel Approach
In stark contrast, the electrochemical method disclosed in the patent offers a metal-free alternative that fundamentally simplifies the synthetic landscape for pyrazole derivatives. By employing anodic oxidation to generate the necessary reactive intermediates in situ, this approach eliminates the need for external chemical oxidants and transition metal catalysts entirely. The reaction proceeds smoothly in a mixed solvent system of acetonitrile and tert-amyl alcohol, utilizing simple electrolytes such as tetrabutylammonium hexafluorophosphate to maintain conductivity. This shift not only reduces the chemical footprint of the synthesis but also drastically simplifies the work-up procedure, as there is no need for specialized metal scavenging resins or complex extraction protocols. The result is a cleaner reaction profile with high atom economy, making it an ideal candidate for reliable agrochemical intermediate supplier networks seeking to optimize their manufacturing portfolios.
Mechanistic Insights into Electrochemical Cyclization
The core of this innovation lies in the electrochemical generation of reactive species that facilitate the coupling of the hydrazine and aldehyde components without external redox agents. In this mechanism, the anode serves as the electron acceptor, oxidizing the hydrazine derivative to form a radical or cationic intermediate that is highly susceptible to nucleophilic attack by the carbonyl group of the benzaldehyde. This electro-driven activation bypasses the high energy barriers typically associated with thermal activation, allowing the reaction to proceed at significantly lower temperatures, often around 50 to 55°C. The use of a carbon cloth anode and a platinum cathode provides a stable and durable surface for electron transfer, ensuring consistent reaction kinetics over extended periods. This precise control over the oxidation potential minimizes side reactions and over-oxidation, which are common pitfalls in chemical oxidant-based methods, thereby enhancing the overall selectivity for the desired pyrazole scaffold.
From an impurity control perspective, the absence of metal catalysts inherently reduces the complexity of the impurity profile, eliminating metal-organic complexes that are difficult to separate. The electrochemical environment promotes a clean conversion where the primary byproducts are typically benign salts or solvent-derived species that are easily removed during aqueous work-up. The patent data indicates yields ranging from 77% to 86% across various substrates, demonstrating that the method maintains high efficiency even with electron-withdrawing or electron-donating groups on the aromatic ring. This robustness is crucial for commercial scale-up of complex pharmaceutical intermediates, where batch-to-batch consistency is paramount. The simplified purification process not only saves time but also reduces the consumption of chromatography materials, contributing to a more sustainable and cost-effective manufacturing lifecycle.
How to Synthesize Pyrazole Compounds Efficiently
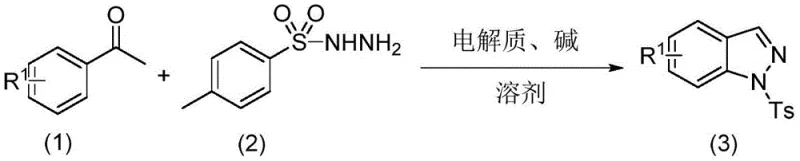
To implement this synthesis effectively, one must carefully prepare the reaction mixture by dissolving the benzaldehyde substrate and p-toluenesulfonylhydrazine in the optimized solvent system with the appropriate electrolyte and base. The detailed standardized synthesis steps involve setting up an undivided cell with specific electrode configurations and maintaining a constant current throughout the reaction duration to ensure complete conversion. The process is designed to be operationally simple, requiring minimal specialized equipment beyond a standard power supply and electrolytic cell, making it accessible for both laboratory research and pilot plant operations. For a comprehensive guide on the exact molar ratios, electrode preparation, and work-up procedures, please refer to the structured protocol provided below.
- Dissolve benzaldehyde compound, p-toluenesulfonylhydrazine compound, alkali, and electrolyte in a mixed solvent to obtain a reaction solution.
- Insert a carbon cloth anode and a platinum cathode into the reaction solution within an undivided electrolytic cell.
- Switch on constant current in an open system at 40 to 70°C to stir and react, then isolate the pyrazole product via extraction and chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this electrochemical methodology offers compelling strategic advantages that extend beyond mere technical feasibility. The elimination of precious metal catalysts directly addresses volatility in raw material pricing, as the cost of copper or palladium salts can fluctuate wildly based on global market dynamics. By removing these dependencies, manufacturers can stabilize their cost structures and improve margin predictability, which is essential for long-term contract negotiations with pharmaceutical clients. Furthermore, the simplified post-treatment process reduces the demand for specialized purification media and solvents, leading to significant operational efficiencies and reduced inventory requirements for consumables.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts and external oxidants eliminates the need for expensive metal scavenging steps and complex waste treatment procedures, resulting in substantial cost savings. This streamlined process reduces the consumption of high-purity reagents and minimizes the loss of product during purification, thereby improving the overall material throughput. Additionally, the milder reaction conditions lower energy consumption for heating and cooling, contributing to a more economical production profile that enhances competitiveness in the global market.
- Enhanced Supply Chain Reliability: By relying on electricity as the primary reagent rather than sensitive chemical oxidants, the process reduces the risk of supply disruptions caused by the shortage of specific reagents. The use of durable electrode materials like carbon cloth and platinum ensures long equipment lifespan and reduces the frequency of replacement parts, enhancing operational continuity. This robustness makes the supply chain more resilient to external shocks, ensuring consistent delivery of high-purity pharmaceutical intermediates to downstream customers without unexpected delays.
- Scalability and Environmental Compliance: The electrochemical nature of the reaction allows for easy scaling by increasing electrode surface area or using flow chemistry setups, facilitating the transition from laboratory to commercial production. The absence of heavy metal waste simplifies environmental compliance, reducing the regulatory burden and costs associated with hazardous waste disposal. This aligns with global sustainability goals, making the manufacturing process more attractive to environmentally conscious partners and investors seeking green chemistry solutions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis route. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on reaction parameters and scalability. Understanding these details is crucial for R&D teams evaluating the feasibility of adopting this technology for their specific pipeline candidates.
Q: What are the primary advantages of this electrochemical method over traditional metal catalysis?
A: The primary advantage is the elimination of transition metal catalysts such as copper or palladium, which removes the need for expensive and complex heavy metal removal steps during post-treatment. Additionally, the reaction operates under milder conditions without requiring strong external oxidants, significantly enhancing the environmental profile and safety of the manufacturing process.
Q: What specific reaction conditions are required for optimal yield in this patent?
A: Optimal yields are achieved using a mixed solvent system of acetonitrile and tert-amyl alcohol, with tetrabutylammonium hexafluorophosphate as the electrolyte. The reaction is conducted at a constant current of 5 to 15mA and a temperature range of 40 to 70°C, typically completing within approximately 2 hours.
Q: Is this synthesis method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the method is highly scalable due to its simple post-treatment process and the use of readily available electrode materials like carbon cloth and platinum. The absence of sensitive metal catalysts reduces supply chain risks, making it a robust candidate for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazole Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this electrochemical technology for the production of high-value heterocyclic intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our facilities are equipped with state-of-the-art electrochemical reactors and stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards. We are committed to helping our partners leverage this green chemistry approach to enhance their product portfolios while maintaining cost efficiency.
We invite you to collaborate with us to explore how this metal-free synthesis can optimize your supply chain and reduce your environmental footprint. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your path to market with reliable, high-quality pyrazole intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
