Advanced Manufacturing of Bifunctional Phenylindane Photoinitiators for High-Performance UV Curing Systems
Introduction to Next-Generation Photoinitiator Technology
The global demand for high-performance UV curing systems in coatings, printing inks, and electronic circuit manufacturing continues to surge, driving the need for more efficient and environmentally friendly photoinitiators. Patent CN102603509B discloses a groundbreaking preparation method for a bifunctional phenylindane photoinitiator, specifically targeting the limitations of legacy synthesis routes. This technology represents a significant leap forward in fine chemical manufacturing, utilizing industrial by-product alpha-methylstyrene (AMS) as a low-cost feedstock to produce a mixture of isomers: 2-hydroxy-1-{3-[4-(2-hydroxy-2-methyl-propionyl)-phenyl]-1,1,3-trimethyl-inden-5-yl}-2-methylacetone (Ic) and its structural counterpart (IIc). These molecules are critical for ensuring rapid crosslinking and deep curing in complex polymer matrices.
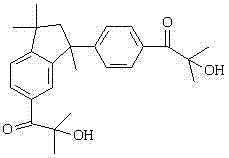
The strategic value of this patent lies in its ability to streamline production while enhancing product purity. Unlike traditional methods that suffer from oligomer contamination and waxy textures, this novel approach yields a cleaner product with superior photoinitiation efficiency. For R&D directors and procurement managers seeking a reliable photoinitiator supplier, understanding the mechanistic advantages of this 3-step route is essential for securing a competitive edge in the UV curing market. The following analysis details how this process optimizes both chemical performance and commercial viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of bifunctional photoinitiators has been plagued by inefficient multi-step protocols, such as those described in U.S. Pat. No. 4,987,159 and US 7,307,192 B2. These conventional routes typically involve the initial synthesis of 1,1,3-trimethyl-3-phenylindane, followed by Friedel-Crafts acylation, and critically, a hazardous halogenation step using chlorine gas or sulfuryl chloride to introduce the alpha-functional group. This halogenation requires subsequent alkaline hydrolysis to convert the alpha-halo ketone into the desired alpha-hydroxy ketone. Such processes are not only operationally cumbersome but also generate substantial amounts of wastewater and corrosive hydrogen chloride gas. Furthermore, the multiple isolation and purification steps inherent in these legacy methods lead to significant product loss and the formation of difficult-to-remove oligomers, resulting in a waxy, viscous final product that complicates downstream formulation.
The Novel Approach
In stark contrast, the methodology outlined in CN102603509B introduces a highly efficient "one-pot two-step" strategy that fundamentally restructures the synthesis workflow. By leveraging cheap and readily available AMS, the process achieves the formation of the indane skeleton and the subsequent acylation in a single reactor setup without intermediate workup. This is immediately followed by a direct hydroxylation reaction using carbon tetrachloride and solid sodium hydroxide, completely bypassing the dangerous chlorination and methoxylation steps found in prior art. The entire synthesis is condensed into just three reaction stages, dramatically simplifying operations and reducing production time.
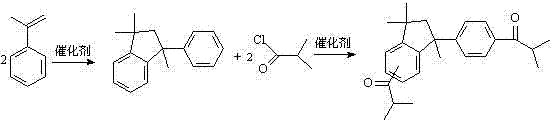
This streamlined approach not only enhances the overall yield to approximately 75.3% but also ensures a higher purity profile by minimizing side reactions associated with intermediate handling. For manufacturers focused on cost reduction in UV curing material manufacturing, this elimination of unit operations translates directly into lower energy consumption and reduced labor costs. The ability to proceed from crude reaction mixtures to the final active ingredient without extensive purification underscores the robustness of this novel chemical engineering solution.
Mechanistic Insights into Lewis Acid-Catalyzed Cyclization and Direct Hydroxylation
The core of this synthesis relies on precise Lewis acid catalysis to drive the cyclization of AMS into the indane framework. Using catalysts such as aluminum trichloride, zinc chloride, or ferric chloride, the reaction proceeds at temperatures between 80°C and 130°C, achieving conversion rates as high as 99.94% with exceptional selectivity. The mechanism involves the generation of a carbocation intermediate from AMS, which undergoes intramolecular electrophilic aromatic substitution to form the stable 1,1,3-trimethyl-3-phenylindane structure. Crucially, the reaction conditions are optimized to prevent polymerization of the styrenic double bond, ensuring that the monomer is efficiently converted into the cyclic precursor required for the subsequent acylation step.
Following the formation of the indane core, the process employs a direct hydroxylation mechanism that avoids the pitfalls of alpha-halogenation. Instead of generating an alpha-chloro ketone which requires harsh hydrolysis, the patented method utilizes a phase transfer catalyst in conjunction with solid sodium hydroxide and carbon tetrachloride. This unique reagent system facilitates the direct introduction of the hydroxyl group at the alpha-position of the ketone. This mechanistic shift is vital for impurity control, as it prevents the formation of chlorinated by-products and reduces the risk of equipment corrosion caused by hydrogen chloride evolution. The result is a cleaner reaction profile that supports the production of high-purity photoinitiators suitable for sensitive applications like electronic circuit manufacturing.
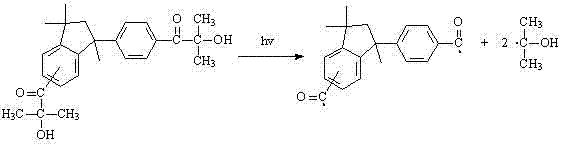
Furthermore, the resulting bifunctional structure exhibits a dual-cleavage mechanism upon UV exposure, known as Norrish Type I cleavage. As illustrated in the photolysis pathway, the molecule can fragment at two distinct sites, generating free radicals that initiate polymerization. This dual functionality not only improves the quantum yield of radical generation but also reduces the migration of photoinitiator fragments from the cured matrix due to the larger molecular weight. For R&D teams, this means enhanced curing depth and reduced odor issues, addressing two of the most persistent challenges in modern UV coating formulations.
How to Synthesize Bifunctional Phenylindane Photoinitiator Efficiently
Implementing this synthesis route requires strict adherence to the specified reaction parameters to maximize yield and safety. The process begins with the careful addition of AMS to a Lewis acid catalyst solution, maintaining temperature control to manage the exothermic cyclization. Once the indane intermediate is formed, isobutyryl chloride is introduced directly into the same vessel for the acylation step, leveraging the residual catalytic activity or supplemented catalyst to drive the reaction to completion. The final hydroxylation step demands precise stoichiometry of solid base and phase transfer agents to ensure complete conversion without degrading the sensitive ketone functionality.
- Perform Lewis acid-catalyzed cyclization of alpha-methylstyrene (AMS) to form 1,1,3-trimethyl-3-phenylindane at 80-130°C.
- Execute a one-pot Friedel-Crafts acylation by adding isobutyryl chloride and aluminum trichloride directly to the crude reaction mixture.
- Conduct direct hydroxylation of the ketone intermediate using carbon tetrachloride and solid sodium hydroxide to yield the final bifunctional photoinitiator.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented manufacturing process offers profound strategic benefits beyond mere technical superiority. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By utilizing alpha-methylstyrene (AMS), which is a widely available industrial by-product, the dependency on expensive, specialized precursors is eliminated. This shift ensures a more stable and cost-effective supply base, insulating the production process from the volatility often associated with niche chemical feedstocks. Additionally, the reduction in reaction steps from a complex multi-stage sequence to a concise three-step protocol significantly lowers the operational overhead required for manufacturing.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of intermediate isolation and purification stages. In traditional synthesis, each isolation step incurs costs related to solvent usage, energy for distillation, and labor for handling. By adopting a "one-pot" strategy for the first two critical steps, the process minimizes solvent consumption and reduces the time operators spend on batch processing. Furthermore, avoiding the use of hazardous chlorine gas removes the need for specialized gas handling infrastructure and scrubbing systems, leading to substantial capital expenditure savings and lower ongoing maintenance costs for safety equipment.
- Enhanced Supply Chain Reliability: The robustness of this synthesis route contributes directly to supply chain continuity. Traditional methods involving gaseous reagents like chlorine are susceptible to logistical disruptions and strict regulatory controls on transportation and storage. By replacing these with solid reagents like sodium hydroxide and liquid acyl chlorides, the manufacturing process becomes more resilient to external supply shocks. The high conversion rates and selectivity reported in the patent data also mean that less raw material is wasted, ensuring that input volumes translate more efficiently into saleable output, thereby stabilizing inventory levels and delivery schedules for downstream customers.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns perfectly with modern green chemistry principles. The avoidance of chlorination steps significantly reduces the generation of saline wastewater and acidic off-gases, simplifying the effluent treatment process and lowering compliance costs. This environmental friendliness facilitates easier permitting for capacity expansion, allowing manufacturers to scale up from pilot batches to commercial tonnage with fewer regulatory hurdles. The simplified workflow also reduces the physical footprint required for production, enabling higher throughput within existing facility constraints.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this bifunctional photoinitiator synthesis. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering clarity on yield optimization, safety protocols, and raw material sourcing. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this technology into their existing production lines.
Q: How does the new one-pot method improve yield compared to traditional chlorination routes?
A: The novel one-pot two-step method eliminates intermediate isolation and purification losses, achieving a total yield of approximately 75.3% compared to roughly 50.9% in conventional multi-step chlorination processes.
Q: What are the safety advantages of avoiding chlorine gas in this synthesis?
A: By replacing hazardous chlorine gas halogenation with a direct hydroxylation using solid sodium hydroxide, the process eliminates the risk of toxic gas leaks, reduces equipment corrosion from hydrogen chloride, and simplifies waste treatment.
Q: Why is alpha-methylstyrene (AMS) considered a cost-effective starting material?
A: AMS is utilized as an industrial by-product, making it significantly cheaper and more readily available than purified precursors, which drastically lowers the raw material costs for large-scale photoinitiator manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bifunctional Phenylindane Photoinitiator Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced UV curing materials depends on a partner who can bridge the gap between laboratory innovation and industrial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN102603509B are fully realized in a manufacturing setting. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of photoinitiator meets the exacting standards required for high-performance coatings and electronic applications.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing process can deliver both superior product quality and significant economic value to your organization.
