Revolutionizing Anastrozole Intermediate Production with Efficient Copper-Catalyzed Decarboxylation
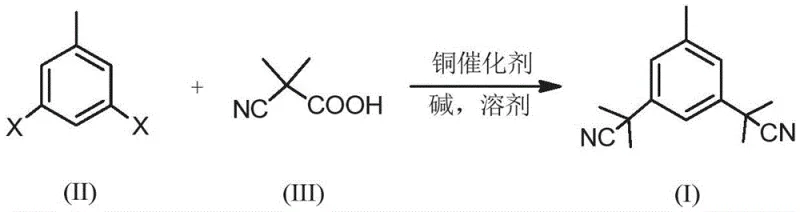
The pharmaceutical industry is constantly seeking robust synthetic pathways that balance efficiency with stringent purity standards, particularly for oncology treatments like breast cancer therapies. Patent CN109096146B introduces a groundbreaking methodology for synthesizing the key intermediate of Anastrozole, a third-generation aromatase inhibitor. This innovation shifts the paradigm from traditional, hazardous multi-step syntheses to a streamlined, one-step copper-catalyzed decarboxylative coupling reaction. By utilizing 3,5-dihalogenated toluene and 2-methyl-2-cyanopropionic acid as primary feedstocks, this process achieves high yields while completely avoiding the use of toxic cyanating agents. For global procurement teams and R&D directors, this represents a significant leap forward in process chemistry, offering a route that is not only economically viable but also aligns perfectly with modern green chemistry principles and regulatory requirements for metal impurities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,5-bis(2,2'-dimethylcyanomethyl)toluene has been plagued by significant operational and safety challenges that hinder efficient commercial production. Traditional routes often rely on Grignard reactions followed by halogenation and cyanide substitution, processes that involve extremely hazardous reagents and generate substantial toxic waste streams. Furthermore, alternative methods utilizing palladium-catalyzed cross-coupling, while shorter in step count, introduce a critical bottleneck: the difficulty of removing trace noble metal residues. In the context of Active Pharmaceutical Ingredients (APIs), residual palladium levels must be reduced to parts per million (ppm) to meet pharmacopeial standards, necessitating expensive and complex purification steps such as specialized scavenging resins or repeated recrystallizations. These legacy methods result in inflated production costs, extended lead times, and increased environmental liability, making them increasingly unsustainable for high-volume manufacturing.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages a copper-catalyzed decarboxylative coupling strategy that elegantly bypasses these historical hurdles. This method employs readily available and inexpensive copper salts, such as CuI or CuOAc, in conjunction with nitrogen-based ligands like 1,10-phenanthroline to facilitate the bond formation. The reaction proceeds in a single pot under relatively mild thermal conditions, directly converting the dihalogenated toluene precursor into the desired dinitrile intermediate. This transformation is remarkable for its atom economy and operational simplicity, as it avoids the generation of stoichiometric metal waste associated with Grignard reagents. Moreover, the use of copper ensures that any residual metal in the final product is far easier to manage and less toxic than palladium, thereby simplifying the downstream purification process and ensuring a cleaner impurity profile for the final drug substance.
Mechanistic Insights into Copper-Catalyzed Decarboxylative Coupling
The core of this technological advancement lies in the mechanistic efficiency of the copper-catalyzed decarboxylative cross-coupling reaction. Unlike palladium cycles which can be prone to catalyst deactivation or difficult oxidative addition steps with certain substrates, the copper system described here operates through a robust catalytic cycle involving the activation of the carboxylic acid derivative. The 2-methyl-2-cyanopropionic acid serves as a safe, solid surrogate for cyanide, undergoing decarboxylation in situ to generate the reactive nucleophilic species necessary for the coupling. The presence of the specific ligand, such as 1,10-phenanthroline or 2,2'-bipyridine, stabilizes the copper center and facilitates the transmetallation and reductive elimination steps required to forge the carbon-carbon bond at the meta-positions of the toluene ring. This precise control over the reaction pathway ensures high regioselectivity and minimizes the formation of mono-substituted byproducts or homocoupling impurities, which are common pitfalls in aromatic substitution chemistry.
From a quality control perspective, the mechanism inherently supports the production of high-purity material suitable for pharmaceutical applications. The choice of base, ranging from cesium carbonate to potassium phosphate, plays a pivotal role in driving the decarboxylation equilibrium forward while neutralizing the acid byproduct. The reaction conditions, typically conducted in polar aprotic solvents like DMSO or DMF at temperatures between 100°C and 150°C, provide sufficient energy to overcome the activation barrier without degrading the sensitive nitrile groups. Crucially, the absence of aggressive cyanide sources means that the impurity profile is devoid of inorganic cyanide salts, simplifying the analytical testing regime. The resulting intermediate exhibits excellent crystallinity, allowing for effective purification via simple ethanol recrystallization, which further strips away any trace organic impurities or catalyst residues, delivering a product that meets the rigorous specifications required for subsequent triazole substitution steps.
How to Synthesize 3,5-bis(2,2'-dimethylcyanomethyl)toluene Efficiently
Implementing this synthesis requires careful attention to the stoichiometry of the copper catalyst and the selection of the appropriate ligand to maximize turnover numbers. The process begins by charging a reactor with the dihalogenated starting material, the cyano-acid coupling partner, and the base under an inert atmosphere to prevent oxidation of the catalyst. Detailed standard operating procedures regarding temperature ramping rates and quenching protocols are essential to maintain safety and consistency across batches. For a comprehensive breakdown of the specific molar ratios, solvent volumes, and isolation techniques validated in the patent examples, please refer to the standardized synthesis guide below.
- Combine 3,5-dihalogenated toluene, 2-methyl-2-cyanopropionic acid, a copper catalyst (e.g., CuI), a ligand (e.g., 1,10-phenanthroline), and a base in a suitable solvent.
- Heat the reaction mixture under nitrogen protection to a temperature between 50°C and 200°C, maintaining stirring for 5 to 100 hours to ensure complete conversion.
- Cool the reaction, precipitate the product with water, and purify via recrystallization from ethanol to obtain the high-purity intermediate free of heavy metal residues.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed route offers transformative benefits that extend well beyond the laboratory bench. The primary advantage is the drastic simplification of the supply chain for raw materials; 3,5-dibromotoluene and 2-methyl-2-cyanopropionic acid are commodity chemicals available from multiple global vendors, reducing the risk of single-source dependency. Furthermore, the elimination of palladium catalysts removes a major cost driver, as noble metals represent a significant portion of the bill of materials in traditional cross-coupling reactions. By switching to base metals like copper, manufacturers can achieve substantial cost savings on catalyst procurement alone, while simultaneously reducing the capital expenditure associated with metal recovery systems. This economic efficiency is compounded by the reduced need for complex purification media, such as expensive scavenger resins, leading to a leaner and more cost-effective manufacturing process overall.
- Cost Reduction in Manufacturing: The transition from noble metal catalysis to earth-abundant copper catalysts fundamentally alters the cost structure of Anastrozole intermediate production. Since copper salts are orders of magnitude cheaper than palladium complexes, the direct material cost is significantly lowered. Additionally, the simplified workup procedure, which relies on water precipitation and recrystallization rather than chromatography or specialized filtration, reduces solvent consumption and labor hours. This holistic reduction in processing complexity translates directly into a lower cost of goods sold (COGS), providing a competitive pricing advantage in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Relying on stable, non-hazardous reagents enhances the resilience of the supply chain against regulatory disruptions. Traditional cyanation methods often face strict transportation and storage regulations due to the toxicity of reagents like sodium cyanide. By utilizing a carboxylic acid precursor, the logistics of raw material handling become safer and less bureaucratic, ensuring uninterrupted production schedules. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further securing the continuity of supply for downstream API synthesis.
- Scalability and Environmental Compliance: The patent data explicitly demonstrates the scalability of this method, with successful runs conducted in 50-liter reactors yielding consistent results. This indicates a clear path to ton-scale production without the need for extensive process re-engineering. From an environmental standpoint, the avoidance of toxic cyanide waste and heavy metal effluents simplifies wastewater treatment and reduces the facility's environmental footprint. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational benefits and quality implications for potential partners.
Q: Why is the copper-catalyzed method superior to palladium catalysis for this intermediate?
A: The copper-catalyzed method eliminates the risk of toxic palladium residues remaining in the final API, which is a critical quality attribute for regulatory compliance. Additionally, copper catalysts are significantly more cost-effective than noble metals, reducing overall production expenses without compromising yield.
Q: What are the safety advantages of this new synthetic route?
A: Unlike traditional methods that require highly toxic cyanation reagents or hazardous Grignard reactions, this process utilizes stable 2-methyl-2-cyanopropionic acid as the cyanide source. This drastically lowers operational risks and simplifies waste treatment protocols.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly demonstrates scalability with examples running in 50L reactors yielding over 80% product. The simple workup procedure involving water precipitation and recrystallization makes it highly adaptable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Anastrozole Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain competitiveness in the global pharmaceutical market. Our technical team has thoroughly analyzed the copper-catalyzed decarboxylation pathway and is fully prepared to implement this state-of-the-art process for the commercial production of 3,5-bis(2,2'-dimethylcyanomethyl)toluene. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and efficient. Our facilities are equipped with rigorous QC labs capable of detecting trace impurities at ppm levels, guaranteeing that every batch meets stringent purity specifications required for oncology drug intermediates.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route for your Anastrozole supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this copper-catalyzed method for your specific volume requirements. Please contact us today to discuss your project needs, and we will be happy to provide specific COA data and route feasibility assessments tailored to your production goals.
