Advanced L-Carnosine Manufacturing: A Novel Aminolysis Strategy for Commercial Scale-Up
The pharmaceutical and nutraceutical industries are constantly seeking robust synthetic pathways for bioactive dipeptides, and the recent disclosure in patent CN112480206A presents a transformative approach to the production of L-carnosine. This intellectual property details a highly efficient synthesis method that fundamentally rethinks the classical peptide coupling strategies by leveraging the inherent chemoselectivity of ester aminolysis. Unlike traditional methodologies that rely on harsh activation agents and multiple protection-deprotection cycles, this novel route utilizes beta-alanine methyl ester as a key starting material, reacting it directly with histidine under mild basic conditions. The strategic innovation lies in the selective protection of only the alanine amino group, thereby circumventing the cumbersome and yield-limiting steps associated with protecting the histidine imidazole ring. For R&D directors and process chemists, this represents a significant leap forward in atom economy and operational simplicity, offering a pathway to high-purity intermediates with reduced environmental impact. As a reliable peptide intermediate supplier, understanding these mechanistic nuances is critical for evaluating the long-term viability and cost-effectiveness of sourcing such critical building blocks for antioxidant and anti-aging formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of carnosine has been plagued by significant chemical inefficiencies stemming from the reliance on acyl chloride activation. In the conventional workflow, the carboxyl group of beta-alanine must first be activated, typically using thionyl chloride or oxalyl chloride, to form a highly reactive acyl chloride species. This step is not only hazardous due to the evolution of corrosive gases but also necessitates rigorous moisture control, adding complexity to the engineering requirements. Furthermore, to achieve acceptable regioselectivity during the coupling with histidine, the imidazole nitrogen and the alpha-amino group of histidine often require orthogonal protection strategies. These additional protection and deprotection sequences drastically increase the number of unit operations, consume excessive amounts of reagents, and inevitably lead to cumulative yield losses. The harsh conditions associated with acyl chloride formation also pose a risk of racemization, threatening the optical purity of the final L-carnosine product, which is paramount for its biological activity in pharmaceutical applications. Consequently, the traditional route suffers from high production costs, significant waste generation, and extended lead times, creating bottlenecks for manufacturers aiming for cost reduction in pharmaceutical manufacturing.
The Novel Approach
In stark contrast, the methodology outlined in the patent introduces a streamlined condensation strategy that capitalizes on the differential nucleophilicity of amino groups. By employing beta-alanine methyl ester protected at the amino terminus (e.g., with a BOC group), the process enables a direct aminolysis reaction with unprotected L-histidine. The use of a catalytic amount of sodium methoxide in a polar protic solvent like methanol facilitates the nucleophilic attack of the histidine alpha-amino group onto the ester carbonyl. This mechanism is inherently selective; the primary amine of histidine is significantly more nucleophilic than the imidazole nitrogen under these specific conditions, effectively rendering histidine side-chain protection unnecessary. This breakthrough simplifies the synthetic tree, reducing the total number of steps and eliminating the need for hazardous chlorinating agents. The reaction proceeds at a moderate temperature of 50°C, ensuring thermal stability and minimizing energy consumption. The result is a process that delivers high yields, as evidenced by the patent examples showing conversion rates exceeding 90% in the coupling step, while maintaining excellent stereochemical integrity. This approach exemplifies how modern process chemistry can achieve cost reduction in pharmaceutical manufacturing through intelligent molecular design rather than brute-force optimization.
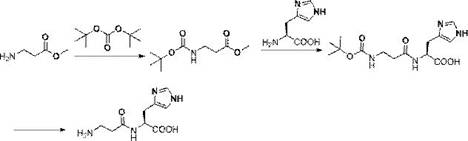
Mechanistic Insights into Sodium Methoxide-Catalyzed Aminolysis
To fully appreciate the technical superiority of this route for high-purity L-carnosine production, one must delve into the mechanistic details of the sodium methoxide-catalyzed aminolysis. The reaction operates on the principle of nucleophilic acyl substitution, where the methoxide ion acts as a base to deprotonate the alpha-amino group of histidine, generating a highly reactive free amine nucleophile in situ. This activated amine then attacks the electrophilic carbonyl carbon of the protected beta-alanine methyl ester. The tetrahedral intermediate subsequently collapses, expelling the methoxide leaving group to form the stable peptide bond. Crucially, the pKa difference between the alpha-amino group and the imidazole nitrogen of histidine ensures that the base selectively activates the desired nucleophile, preventing N-alkylation or side reactions on the heterocyclic ring. This intrinsic chemoselectivity is the cornerstone of the process, allowing for the omission of histidine protecting groups which are traditionally difficult to install and remove without affecting the peptide backbone. Furthermore, the mild basic conditions and the absence of strong activating agents like carbodiimides or acid chlorides significantly reduce the risk of epimerization at the chiral center of the histidine residue. For quality control teams, this means a cleaner impurity profile with fewer diastereomeric byproducts, simplifying downstream purification and ensuring the final product meets stringent pharmacopeial standards for optical purity.
Impurity control in this synthesis is further enhanced by the choice of solvents and the specific deprotection protocols. The use of methanol or ethanol as the reaction medium not only solubilizes the reactants but also suppresses potential hydrolysis of the ester prior to aminolysis. Following the coupling, the removal of the BOC protecting group is achieved under controlled acidic conditions, typically using hydrochloric acid or trifluoroacetic acid. The patent specifies that careful pH adjustment during the workup allows for the selective precipitation of the zwitterionic L-carnosine product, leaving soluble impurities in the mother liquor. This crystallization-driven purification is highly effective for removing trace organic byproducts and residual salts. Additionally, the avoidance of transition metal catalysts in the coupling step (unlike some cross-coupling approaches) eliminates the risk of heavy metal contamination, a critical parameter for API intermediates intended for human consumption. The combination of selective reactivity, mild conditions, and crystallization-based purification creates a robust process window that is forgiving of minor variations in raw material quality, thereby enhancing the overall reliability of the supply chain for complex peptide intermediates.
How to Synthesize L-Carnosine Efficiently
The practical implementation of this synthesis route offers a clear roadmap for process engineers looking to adopt this technology. The procedure begins with the straightforward protection of beta-alanine methyl ester, followed by the key condensation step with histidine, and concludes with a simple deprotection and isolation sequence. The operational simplicity of these steps makes the technology highly transferable from laboratory scale to multi-ton commercial production. Detailed standardized synthesis steps are provided below to guide technical teams in replicating these results.
- Protect the amino group of beta-alanine methyl ester using BOC anhydride in a solvent like THF or acetonitrile to form the protected intermediate.
- Perform a condensation reaction between the protected alanine ester and L-histidine using a catalytic amount of sodium methoxide in methanol at 50°C.
- Remove the protecting group via acid hydrolysis (e.g., HCl) or hydrogenation to yield the final pure L-carnosine product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthesis route offers profound strategic advantages that extend beyond mere technical elegance. The elimination of histidine protection steps translates directly into a reduction in raw material complexity and cost. Traditional routes require the purchase of specialized protected histidine derivatives or the reagents to synthesize them in-house, both of which carry premium price tags and longer lead times. By utilizing bulk commodity histidine and beta-alanine methyl ester, manufacturers can leverage existing supply chains and negotiate better pricing due to the high availability of these starting materials. Furthermore, the reduction in the number of synthetic steps inherently lowers the labor and utility costs associated with production. Fewer reactors are tied up for shorter durations, increasing the overall throughput of the manufacturing facility. This efficiency gain is crucial for meeting the growing global demand for carnosine in the nutraceutical and cosmetic sectors without requiring massive capital expenditure on new infrastructure. The process also aligns with green chemistry principles by reducing solvent usage and waste generation, which helps companies meet increasingly strict environmental regulations and sustainability goals.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the drastic simplification of the synthetic pathway. By removing the need for histidine protection and deprotection, the process eliminates the cost of protecting group reagents, the solvents required for those steps, and the disposal costs for the resulting waste streams. Additionally, the replacement of hazardous acyl chloride activators with benign sodium methoxide reduces the need for specialized corrosion-resistant equipment and safety monitoring systems. The high yields reported in the patent examples suggest that less starting material is wasted, improving the overall mass balance and reducing the cost per kilogram of the final active ingredient. These factors combine to create a leaner, more cost-effective manufacturing model that can withstand market fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available, commodity-grade chemicals. Beta-alanine, histidine, and sodium methoxide are produced globally in large volumes, reducing the risk of supply disruptions caused by the scarcity of niche reagents. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent grades, further ensures consistent production output even when facing variations in raw material quality. The simplified workup, involving filtration and crystallization rather than complex chromatographic separations, allows for faster batch turnover times. This agility enables suppliers to respond more quickly to urgent customer orders and maintain healthier inventory levels, ensuring continuity of supply for downstream pharmaceutical formulators who depend on timely delivery of high-purity intermediates.
- Scalability and Environmental Compliance: The scalability of this process is evidenced by its use of standard unit operations such as stirred tank reactions, filtration, and drying, which are easily replicated in large-scale GMP facilities. The mild reaction temperatures minimize energy consumption for heating and cooling, contributing to a lower carbon footprint. Moreover, the avoidance of chlorinated solvents and heavy metal catalysts simplifies wastewater treatment and reduces the regulatory burden associated with hazardous waste disposal. This environmental compatibility is increasingly becoming a deciding factor for multinational corporations selecting suppliers, as they strive to meet their own Scope 3 emissions targets. The ability to produce high-quality L-carnosine with a smaller environmental footprint positions this technology as a future-proof solution for sustainable chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of the new route. Understanding these details is essential for stakeholders evaluating the potential for technology transfer or procurement partnerships.
Q: How does the new aminolysis method improve selectivity over traditional acyl chloride routes?
A: The novel method utilizes the nucleophilic attack of the primary amine of histidine on the activated ester carbonyl. This mechanism inherently favors the alpha-amino group over the imidazole nitrogen, eliminating the need for complex histidine side-chain protection required in acyl chloride methods.
Q: What are the supply chain advantages of avoiding histidine protection?
A: By bypassing the protection and deprotection steps for histidine, the process significantly reduces the consumption of expensive protecting reagents and solvents. This simplification shortens the overall production cycle and minimizes waste generation, leading to substantial cost savings and enhanced supply reliability.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the reaction conditions are mild (50°C) and utilize common solvents like methanol and THF. The workup involves simple filtration and crystallization steps, which are highly amenable to large-scale reactor operations, ensuring consistent quality from pilot to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Carnosine Supplier
The technological advancements detailed in patent CN112480206A underscore the dynamic nature of peptide synthesis and the continuous drive for more efficient manufacturing solutions. At NINGBO INNO PHARMCHEM, we recognize the value of such innovations and possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring these laboratory breakthroughs to the global market. Our state-of-the-art facilities are equipped to handle the specific requirements of aminolysis reactions and subsequent purification steps, ensuring that every batch meets stringent purity specifications. With our rigorous QC labs and commitment to process excellence, we guarantee the delivery of high-purity L-carnosine that adheres to the highest international standards, providing our partners with a competitive edge in the formulation of advanced health and wellness products.
We invite procurement leaders and R&D directors to explore the potential of this optimized synthesis route for their supply chains. By collaborating with us, you gain access to not just a product, but a comprehensive partnership focused on value creation. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals in the pharmaceutical and nutraceutical industries.
