Advanced Manufacturing of Epiandrosterone: A Cost-Effective Androstenedione Route
Introduction to Patent CN108752409B
The pharmaceutical industry continuously seeks robust and scalable pathways for critical steroid intermediates, and the recent disclosure in Chinese patent CN108752409B presents a transformative approach to synthesizing Epiandrosterone. This document details a novel four-step methodology that utilizes Androstenedione as the primary starting material, effectively bypassing the logistical and chemical bottlenecks of traditional biosynthetic routes. By leveraging a strategic sequence of enol esterification, ketal protection, palladium-carbon catalytic reduction, and final acidic hydrolysis, this technology delivers a streamlined process that is exceptionally well-suited for large-scale industrial manufacturing. The significance of this innovation lies not only in its chemical elegance but also in its ability to utilize abundant, fermentation-derived raw materials, thereby decoupling production from the volatile agricultural supply chains that have historically constrained steroid hormone synthesis.
For R&D directors and process chemists, this patent represents a pivotal shift towards safer, more controllable chemistry. The method explicitly addresses the challenges of selectivity in poly-functionalized steroid skeletons, ensuring that the reduction occurs precisely where needed without compromising the integrity of other sensitive functional groups. As a reliable pharmaceutical intermediates supplier, understanding these mechanistic nuances is crucial for evaluating the long-term viability of supply contracts. The transition from complex, hazard-prone reagents to catalytic hydrogenation underscores a broader industry trend towards green chemistry principles, offering a pathway that aligns with increasingly stringent environmental regulations while maintaining high throughput and product quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
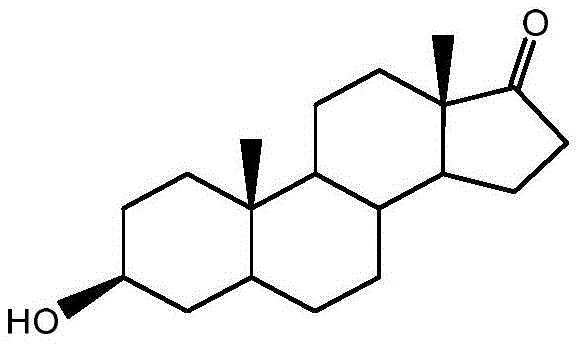
Historically, the industrial production of Epiandrosterone has been heavily reliant on Diosgenin, a sapogenin extracted from wild yams or turmeric. As illustrated in the traditional synthetic route, this pathway involves a cumbersome series of transformations including esterification, cracking, oxidation, and hydrolysis to first obtain Dehydroepiandrosterone (DHEA), followed by oximation, Beckmann rearrangement, and finally hydrogenation. This multi-step lineage is inherently inefficient, suffering from low overall yields due to the accumulation of losses at each stage. Moreover, the reliance on plant-based raw materials introduces significant supply chain volatility; the availability of Diosgenin is strictly bound by seasonality, geographical location, and the ecological impact of over-harvesting, making it an unreliable foundation for consistent commercial scale-up of complex pharmaceutical intermediates.
Furthermore, alternative synthetic attempts using mono-enol ketone acetates have introduced severe safety hazards into the manufacturing environment. Specific prior art methods necessitate the use of phosphorus oxychloride (POCl3) during the rearrangement steps, a reagent known for its high toxicity and corrosive nature, which demands specialized handling equipment and rigorous waste treatment protocols. Other routes employing lithium-ammonia reduction or borohydride reductions add layers of operational complexity and cost, requiring cryogenic conditions or generating substantial amounts of boron-containing waste. These conventional methodologies are not only economically burdensome due to high reagent costs and low atom economy but also pose significant occupational health and safety risks that modern facilities strive to eliminate.
The Novel Approach
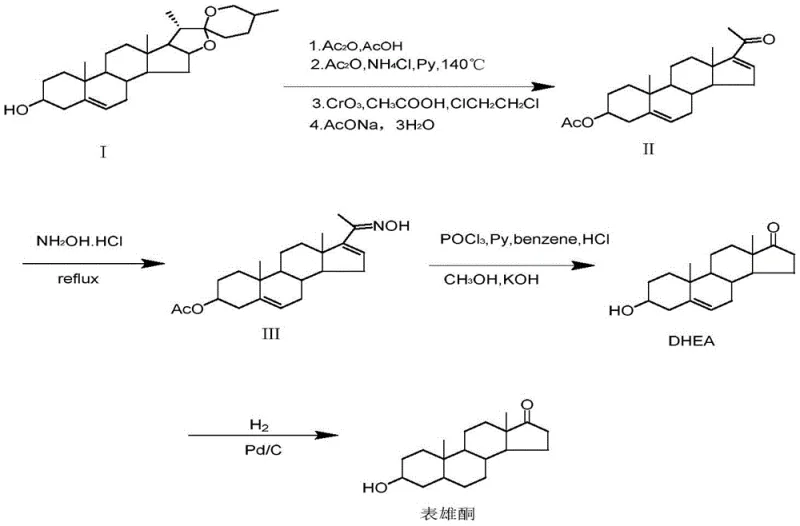
In stark contrast to these legacy processes, the novel approach detailed in CN108752409B leverages Androstenedione, a readily available fermentation product, to construct the target molecule with remarkable efficiency. This strategy fundamentally reimagines the synthetic logic by prioritizing protective group chemistry to manage reactivity. The process initiates with a selective enol esterification at the C3 position, followed by the formation of a ketal at the C17 position. This dual-protection strategy is the key intellectual breakthrough, as it temporarily masks the reactive carbonyls, allowing for a highly selective catalytic hydrogenation of the C4-C5 double bond in subsequent steps without affecting the ketone functionality. This level of control is difficult to achieve in the unprotected substrate and eliminates the need for harsh reducing agents.
The culmination of this route is the use of palladium on carbon (Pd/C) for the reduction step, a heterogeneous catalytic process that is vastly superior to homogeneous metal reductions in terms of workup and scalability. By replacing lithium and borohydride reagents with hydrogen gas and a recoverable solid catalyst, the process drastically simplifies the downstream purification. The final acidic hydrolysis step cleanly removes the protecting groups to reveal the desired 3-beta-hydroxy-17-keto stereochemistry of Epiandrosterone. This streamlined four-step sequence not only shortens the production timeline but also significantly enhances the safety profile of the manufacturing plant, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing where margin compression is a constant pressure.
Mechanistic Insights into Pd/C Catalytic Hydrogenation and Protection Strategy
The core of this synthetic innovation lies in the precise orchestration of protection and deprotection cycles coupled with heterogeneous catalysis. The initial esterification of Androstenedione with acetic anhydride creates an enol acetate, which serves a dual purpose: it protects the C3 oxygen functionality and modifies the electronic environment of the A-ring. Subsequently, the reaction with ethylene glycol under acidic conditions forms a cyclic ketal at the C17 position. This step is critical because the C17 ketone is susceptible to reduction under hydrogenation conditions; by converting it into a sterically bulky and electronically distinct ketal, the chemist ensures that the subsequent hydrogenation catalyst interacts exclusively with the C4-C5 olefinic bond. This chemoselectivity is the hallmark of a mature process design, preventing the formation of over-reduced byproducts that would otherwise contaminate the final API intermediate.
The reduction step utilizing Pd/C is mechanistically straightforward yet operationally powerful. Under pressurized hydrogen conditions (0.5-2 MPa) and moderate temperatures (20-100°C), the palladium surface adsorbs hydrogen and facilitates its syn-addition across the double bond. The presence of an alkaline reagent during this phase aids in the simultaneous hydrolysis of the C3 enol ester, regenerating the hydroxyl group in situ or preparing it for the final step. The beauty of using a heterogeneous catalyst like Pd/C is the ease of separation; post-reaction, the catalyst can be removed via simple filtration, washed, and potentially regenerated for reuse. This contrasts sharply with soluble metal catalysts that require complex extraction or chromatography to remove trace metal impurities, a critical consideration for meeting the stringent purity specifications required by regulatory bodies for hormonal drug substances.
How to Synthesize Epiandrosterone Efficiently
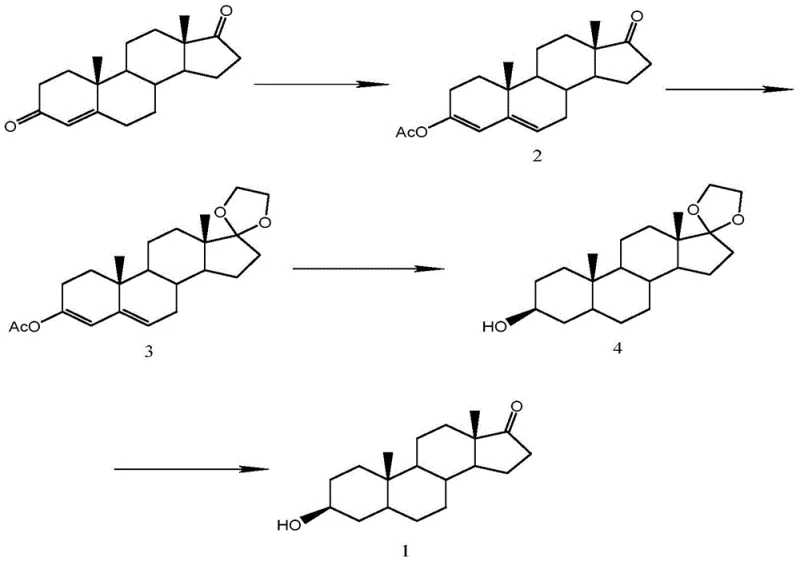
The execution of this synthesis requires careful attention to reaction parameters, particularly temperature control and stoichiometry, to maximize the yield of each intermediate. The process begins with the dissolution of Androstenedione in a suitable organic solvent such as dichloromethane, followed by the addition of acetic anhydride and an acid catalyst like p-toluenesulfonic acid. Maintaining the reaction temperature between 20-80°C for a duration of 5-30 hours ensures complete conversion to the enol ester intermediate. Following isolation, the second step involves ketalization using ethylene glycol and a water scavenger like trimethyl orthoformate, which drives the equilibrium towards the protected product. The detailed standardized operating procedures for these transformations, including specific quenching and crystallization techniques, are essential for reproducibility.
- Perform 3-carbonyl enol esterification of Androstenedione using acetic anhydride and an acid catalyst at 20-80°C.
- Protect the 17-carbonyl group by reacting the intermediate with ethylene glycol and a water removing agent to form a ketal.
- Conduct alkaline hydrolysis followed by Pd/C catalytic hydrogenation at 20-100°C and 0.5-2MPa pressure to reduce the double bond.
- Finalize the synthesis by acidic hydrolysis at 40-60°C to remove the protecting groups and isolate pure Epiandrosterone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Androstenedione-based route offers compelling strategic advantages that extend beyond simple unit cost calculations. The primary benefit is the stabilization of the raw material supply base. By shifting away from agricultural extracts like Diosgenin, which are subject to crop failures and price speculation, manufacturers can secure a steady flow of fermentation-derived Androstenedione. This transition mitigates the risk of production stoppages due to raw material shortages, ensuring enhanced supply chain reliability for downstream customers who depend on just-in-time delivery models for their own drug formulation schedules. The consistency of the starting material quality also reduces the variability in the final product, leading to fewer batch rejections and less waste.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the elimination of expensive and hazardous reagents. By removing the need for lithium metal, liquid ammonia, and potassium borohydride, the facility saves significantly on both material costs and the specialized infrastructure required to handle them safely. Furthermore, the use of Pd/C, which can be recovered and reused, lowers the recurring cost of catalysts compared to stoichiometric reducing agents that are consumed in every batch. The simplified workup procedures, involving filtration rather than complex extractions or chromatography, reduce labor hours and solvent consumption, contributing to substantial cost savings in the overall manufacturing budget.
- Enhanced Supply Chain Reliability: The robustness of the four-step sequence translates directly into shorter lead times and higher predictability. Because the reaction conditions are mild and the intermediates are stable, the process is less prone to deviations that cause delays. The ability to run the hydrogenation step in standard high-pressure reactors, which are common assets in most fine chemical plants, means that production does not require niche equipment that might create bottlenecks. This flexibility allows for easier scaling from pilot batches to multi-ton commercial production, ensuring that suppliers can respond rapidly to fluctuations in market demand without compromising on delivery schedules.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this route is vastly superior. The avoidance of phosphorus oxychloride and heavy metal waste streams simplifies the effluent treatment process, reducing the burden on wastewater treatment facilities and lowering disposal costs. The atom economy of the hydrogenation step is excellent, producing minimal byproducts. This alignment with green chemistry principles not only future-proofs the manufacturing site against tightening environmental regulations but also enhances the corporate sustainability profile, a factor that is increasingly weighted in the vendor selection criteria of major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on the practical aspects of adopting this technology. Understanding these details helps stakeholders assess the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is Androstenedione preferred over Diosgenin for Epiandrosterone production?
A: Androstenedione is preferred because it avoids the seasonal and geographical limitations associated with Diosgenin extraction from turmeric. Furthermore, the Androstenedione route eliminates the need for hazardous reagents like POCl3 and complex multi-step sequences involving oximation and Beckmann rearrangement, resulting in a shorter, safer, and more industrially scalable process.
Q: What are the advantages of using Palladium-Carbon (Pd/C) in this synthesis?
A: The use of Pd/C replaces traditional lithium-ammonia and borohydride reductions. Pd/C offers superior selectivity for the specific double bond reduction required, operates under milder conditions, and allows for easy filtration and catalyst recovery. This significantly simplifies the workup procedure and reduces the generation of toxic waste compared to metal hydride methods.
Q: What purity levels can be achieved with this novel method?
A: According to the patent data, this method achieves high purity levels, with HPLC analysis showing product purity reaching up to 99% after the reduction step and maintaining approximately 98% purity in the final isolated Epiandrosterone. The simplified purification steps contribute to this high quality, making it suitable for high-end pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Epiandrosterone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthesis routes for high-value steroid intermediates like Epiandrosterone. Our technical team has thoroughly analyzed the potential of the Androstenedione pathway and is prepared to support partners in scaling this technology from laboratory benchtop to full commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot to plant is seamless. Our facilities are equipped with state-of-the-art high-pressure hydrogenation reactors and rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required for hormonal pharmaceutical applications.
We invite you to collaborate with us to leverage these process improvements for your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can drive value and security for your global operations.
