Advanced One-Step Synthesis of Pentoxifylline for Commercial API Manufacturing
Introduction to Next-Generation Pentoxifylline Manufacturing
The pharmaceutical landscape for cerebrovascular therapeutics demands increasingly efficient and pure synthetic routes for active ingredients. Patent CN113214259A introduces a transformative approach to the production of pentoxifylline, a critical hemorheologic agent used to treat peripheral vascular diseases and improve cerebral circulation. Unlike legacy processes that rely on complex multi-step sequences and arduous purification protocols, this disclosed methodology achieves the construction of the xanthine core through a highly efficient one-step condensation cyclization. By reacting 4-(N-methylcarbamoyl chloride)-1-methylimidazole-5-carbonyl chloride with 6-amino-2-hexanone, the process directly assembles the target molecule with exceptional precision. This technological leap not only simplifies the operational workflow but also fundamentally alters the impurity profile, offering a robust solution for manufacturers seeking to enhance supply chain reliability and product quality.
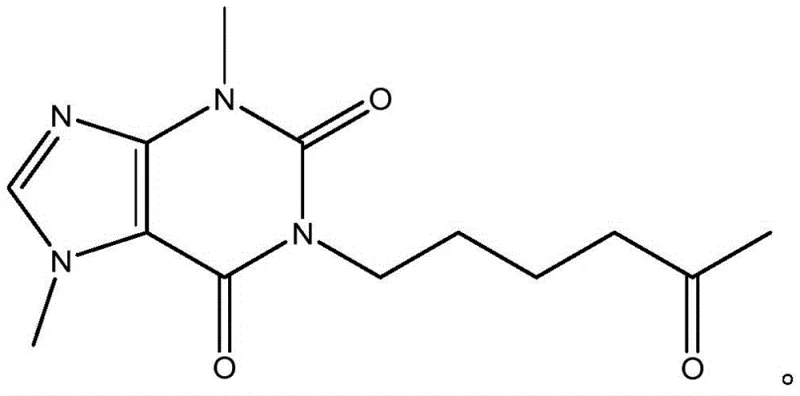
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of pentoxifylline has been plagued by significant downstream processing challenges. Prior art, such as the methods described in CN101896484B and CN108164530A, focuses heavily on the refining of crude theobromine or recovered pentoxifylline rather than optimizing the initial synthesis. These traditional pathways often involve dissolving crude products in alkaline solutions, decolorizing with activated carbon, and performing multiple acidification and crystallization cycles. Despite these intensive efforts, achieving a single impurity level below 0.1% remains difficult, often requiring repeated recrystallization that drastically reduces overall yield. Furthermore, these refining-centric approaches fail to address the root cause of impurity generation during the bond-forming reactions, leading to inconsistent batch quality and increased waste generation. The reliance on post-synthesis purification creates a bottleneck that limits scalability and inflates the cost of goods sold (COGS) due to excessive solvent consumption and extended processing times.
The Novel Approach
In stark contrast, the method disclosed in CN113214259A bypasses these inefficiencies by engineering a direct, one-pot synthesis that constructs the purine skeleton in a single operation. The core innovation lies in the strategic selection of a bifunctional imidazole precursor, 4-(N-methylcarbamoyl chloride)-1-methylimidazole-5-carbonyl chloride, which reacts seamlessly with the amino-ketone side chain precursor. This condensation cyclization eliminates the need for intermediate isolation and the associated handling losses. The reaction is designed such that the by-products generated can be recycled back into the synthesis loop under modified conditions, thereby maximizing atom economy. By shifting the focus from purification to precise reaction control, this novel approach delivers pentoxifylline with purity levels exceeding 99% directly from the reactor, significantly reducing the burden on downstream refining units and enabling a more streamlined, cost-effective manufacturing campaign.
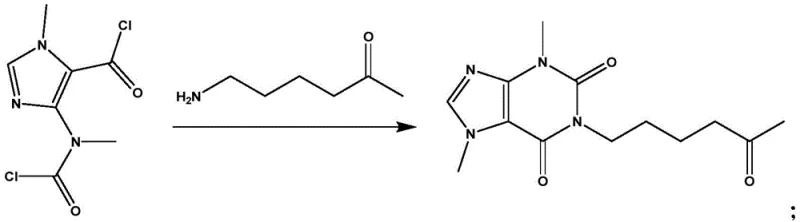
Mechanistic Insights into Temperature-Controlled Cyclization
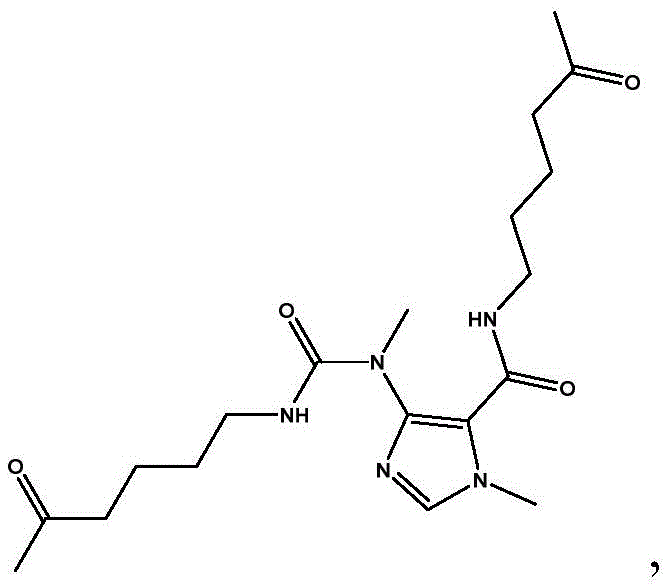
The success of this synthesis hinges on a delicate balance of kinetic and thermodynamic factors, primarily governed by reaction temperature. The condensation involves the nucleophilic attack of the amine on the acid chloride functionalities, followed by an intramolecular cyclization to close the pyrimidine ring of the xanthine system. Experimental data indicates that the reaction window is exceptionally narrow, optimized between -15°C and 5°C. Deviations outside this range trigger distinct mechanistic pathways that lead to stable, non-productive intermediates. For instance, conducting the reaction at 10°C promotes the formation of a specific by-product with a molecular weight of 393.4, likely resulting from incomplete cyclization or alternative acylation patterns where the amine reacts preferentially with the carbamoyl chloride moiety rather than facilitating ring closure. Conversely, lowering the temperature to -30°C kinetically traps a different intermediate (MW 314.1), preventing the necessary activation energy for the final cyclization step from being reached. This sensitivity underscores the importance of precise thermal management in reactor design to ensure the reaction proceeds exclusively through the desired transition state.
Furthermore, the stoichiometry of the acid-binding agent plays a pivotal role in driving the equilibrium towards the product while neutralizing the hydrochloric acid by-product. The patent specifies a molar ratio of the acid-binding agent (such as TEA or DIPEA) to the total reactants of between 1.8:1 and 3.3:1. This excess base ensures that the protonated amine species are rapidly deprotonated, maintaining a high concentration of the free nucleophile required for the acylation steps. Insufficient base leads to incomplete conversion and the accumulation of mono-acylated intermediates, while excessive base may promote hydrolysis of the sensitive acid chloride groups if moisture is present. The ability to recycle by-products suggests that the reaction mechanism allows for the reversion or reprocessing of these off-pathway species, providing a safety net that enhances the overall robustness of the process against minor fluctuations in feedstock quality or mixing efficiency.
How to Synthesize Pentoxifylline Efficiently
The implementation of this synthesis route requires strict adherence to the defined process parameters to replicate the high yields and purity reported in the patent. The procedure begins with the dissolution of the reactants in a suitable anhydrous organic solvent, followed by the controlled addition of the acid-binding agent at sub-ambient temperatures.
- Dissolve 4-(N-methylcarbamoyl chloride)-1-methylimidazole-5-carbonyl chloride and 6-amino-2-hexanone in an organic solvent such as THF or DMF.
- Add an acid-binding agent like Triethylamine (TEA) or DIPEA while maintaining the reaction temperature strictly between -15°C and 5°C.
- Monitor the reaction until the raw material peak disappears, then proceed with aqueous workup, extraction, and crystallization using ethanol and petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis technology offers tangible strategic benefits beyond mere technical elegance. The consolidation of multiple synthetic steps into a single unit operation drastically reduces the manufacturing footprint and the number of required reactor turnovers. This simplification translates directly into lower operational expenditures, as there is less demand for intermediate storage, reduced solvent recovery loads, and minimized labor hours per kilogram of output. Moreover, the capability to recycle reaction by-products back into the process stream enhances the overall material efficiency, effectively lowering the raw material consumption rate. This circular approach within the batch process mitigates the impact of raw material price volatility and reduces the volume of hazardous waste requiring disposal, aligning with increasingly stringent environmental regulations and sustainability goals.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation and purification steps significantly lowers the cost of goods sold. By avoiding the energy-intensive distillation and crystallization cycles associated with multi-step syntheses, manufacturers can achieve substantial utility savings. Additionally, the high selectivity of the reaction minimizes the loss of valuable starting materials to side products, ensuring that a greater proportion of the input mass is converted into saleable API. The use of common, commercially available solvents like THF and DMF further ensures that solvent procurement remains cost-stable and logistically simple, avoiding the need for exotic or highly regulated reagents that often carry premium pricing and long lead times.
- Enhanced Supply Chain Reliability: The robustness of this one-step process enhances supply continuity by reducing the number of potential failure points in the production line. Traditional multi-step syntheses are vulnerable to delays at each stage; a bottleneck in purification can halt the entire campaign. In contrast, this streamlined route shortens the overall cycle time from raw material intake to finished goods, allowing for faster response to market demand spikes. The tolerance for recycling by-products also acts as a buffer against yield fluctuations, ensuring that even batches with minor deviations can be recovered rather than scrapped. This resilience is critical for maintaining consistent inventory levels and meeting the rigorous delivery schedules demanded by global pharmaceutical partners.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by its straightforward reaction profile. The absence of complex catalytic systems or sensitive organometallic reagents simplifies the engineering requirements for large-scale reactors. The reaction operates effectively in standard glass-lined or stainless steel vessels without the need for specialized high-pressure or cryogenic equipment beyond standard chilling capabilities. From an environmental perspective, the reduced solvent usage and the ability to recycle internal by-products significantly lower the E-factor (mass of waste per mass of product). This aligns with green chemistry principles, making the facility more compliant with environmental permits and reducing the carbon footprint associated with the manufacturing of this essential vascular medication.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pentoxifylline synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific manufacturing needs. Understanding these nuances is essential for R&D teams planning technology transfer and for quality assurance teams establishing control strategies.
Q: Why is temperature control critical in this pentoxifylline synthesis?
A: Temperature control between -15°C and 5°C is vital because deviations lead to specific impurities. At 10°C, a by-product with molecular weight 393.4 forms, while at -30°C, a different impurity (MW 314.1) dominates, both preventing the formation of the target purine ring.
Q: What are the advantages of this one-step method over traditional refining?
A: Traditional methods often require multiple refining steps to reduce impurities below 0.1%. This novel one-step condensation cyclization controls impurities at the source, achieving purities above 99% directly and allowing for the recycling of reaction by-products.
Q: Which solvents are compatible with this synthesis route?
A: The process demonstrates flexibility with polar aprotic solvents, specifically Tetrahydrofuran (THF), 1,4-dioxane, and N,N-Dimethylformamide (DMF), allowing manufacturers to select based on availability and downstream processing requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pentoxifylline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving medications like pentoxifylline. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ advanced analytical techniques to verify identity and assay every batch. Our infrastructure is designed to handle complex condensation reactions with the precise temperature control and safety measures required by this specific patent technology, guaranteeing consistency and reliability for our global partners.
We invite you to collaborate with us to leverage this advanced synthesis route for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your pentoxifylline sourcing strategy, reduce lead times, and secure a stable supply of this vital pharmaceutical intermediate.
