Optimizing Alpha,Alpha-Difluoroacetaldehyde Hemiacetal Production via Ruthenium Catalysis for Commercial Scale-up
The pharmaceutical and agrochemical industries continuously seek robust synthetic routes for fluorinated intermediates, driven by the unique physicochemical properties imparted by the difluoromethyl group. Patent CN110520403B introduces a transformative methodology for the production of alpha,alpha-difluoroacetaldehyde hemiacetal (DFAL-ROH), a critical building block for advanced materials and bioactive molecules. This invention addresses the longstanding challenge of dimerization and instability associated with alpha,alpha-difluoroacetaldehyde derivatives. By leveraging a specialized ruthenium-catalyzed hydrogenation protocol coupled with stringent environmental controls during purification, the process achieves high purity while suppressing the formation of stable dimeric impurities. For R&D directors and procurement specialists, this technology represents a significant leap forward in process safety and yield optimization, moving away from hazardous hydride reductions toward catalytic hydrogenation.
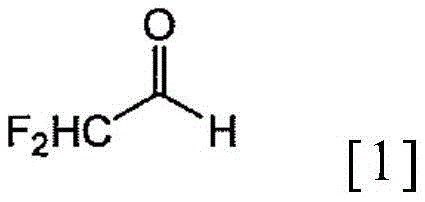
The structural integrity of the difluoromethyl moiety is paramount for the biological activity of the final API, making the stability of the intermediate crucial for supply chain reliability. The patent elucidates how the direct bonding of the difluoromethyl group to the aldehyde carbon creates a highly electrophilic center prone to self-polymerization. Traditional methods often struggled to isolate the monomeric hemiacetal without significant contamination from these polymeric species. The novel approach detailed in CN110520403B not only synthesizes the target molecule efficiently but also incorporates a unique post-synthesis treatment that actively reverses dimer formation. This capability ensures that the final product delivered to downstream synthesis lines meets the rigorous purity specifications required for GMP manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha,alpha-difluoroacetaldehyde derivatives relied heavily on stoichiometric reduction using metal hydrides such as lithium aluminum hydride (LiAlH4). While effective in small-scale laboratory settings, these reagents pose severe safety hazards and waste disposal challenges when scaled to industrial production levels. Furthermore, earlier catalytic methods disclosed in prior art, including previous patents by the same applicants, revealed a critical flaw: the inevitable formation of stable dimers during the purification distillation step. As illustrated in the chemical structures below, these dimers are chemically robust species that do not easily revert to the desired hemiacetal under standard conditions.
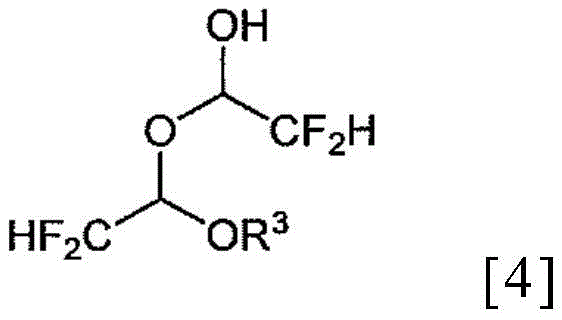
The persistence of these dimeric impurities necessitated additional processing steps, often involving strong acids and intense heating, which ironically led to the decomposition of the product into difluoroacetic acid. This degradation not only reduced overall yield but also introduced corrosive by-products that could damage reactor vessels and compromise equipment longevity. For supply chain heads, the variability in yield and the generation of hazardous waste streams represented a significant bottleneck. The inability to effectively manage the equilibrium between the hemiacetal and its dimer meant that batch-to-batch consistency was difficult to maintain, leading to potential delays in API production schedules and increased costs for raw material consumption.
The Novel Approach
The methodology described in CN110520403B fundamentally alters the purification landscape by introducing a controlled atmosphere and pH management strategy. Instead of accepting dimer formation as an unavoidable loss, the new process treats the distillation residue as a convertible intermediate. By maintaining the reaction environment under strict light-shielding conditions and regulating the gas-phase oxygen concentration to below 5000 ppm, the process inhibits oxidative degradation pathways. Crucially, the patent demonstrates that by adjusting the pH of the mixture to a neutral or weakly alkaline range (pH 3.5 to 10.0) prior to alcohol addition, the stable dimers can be effectively cracked back into the target alpha,alpha-difluoroacetaldehyde hemiacetal. This "self-healing" aspect of the synthesis significantly enhances the atom economy of the process.
Moreover, the shift from stoichiometric hydrides to catalytic hydrogenation using molecular hydrogen eliminates the generation of massive amounts of aluminum or boron waste. This transition aligns with modern green chemistry principles, reducing the environmental footprint of the manufacturing site. For procurement managers, this means a drastic simplification of the waste treatment workflow and a reduction in the cost of goods sold (COGS) associated with reagent procurement and disposal. The ability to produce high-purity intermediates with minimal by-product accumulation ensures a more reliable supply of key starting materials for complex drug synthesis, thereby reducing lead times for high-purity pharmaceutical intermediates.
Mechanistic Insights into Ruthenium-Catalyzed Hydrogenation and Dimer Control
The core of this technological advancement lies in the selection of highly active ruthenium catalysts, specifically pincer complexes such as the Ru-MACHO type depicted below. These catalysts facilitate the selective reduction of the ester functionality to the aldehyde level without over-reducing to the alcohol, a common side reaction in fluorinated systems. The electron-withdrawing nature of the fluorine atoms makes the carbonyl carbon highly susceptible to nucleophilic attack, requiring a catalyst that can modulate the hydride transfer with precision. The ruthenium center, coordinated with specific phosphine and amine ligands, creates a steric and electronic environment that favors the formation of the hemiacetal in the presence of the alcohol solvent.
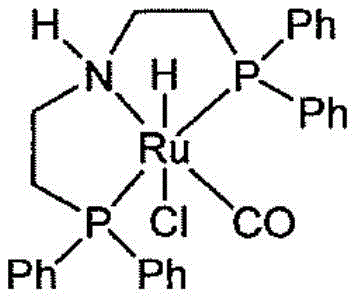
Beyond the initial reduction, the mechanism for impurity control is equally sophisticated. The patent reveals that the stability of the dimer is pH-dependent; in highly acidic conditions (pH < 3.5), often caused by trace difluoroacetic acid, the dimer becomes kinetically trapped. The innovation here is the realization that maintaining a pH between 3.5 and 10.0 keeps the system in a state where the dimer-alcohol equilibrium can be manipulated. By introducing fresh alcohol under low-oxygen conditions, the Le Chatelier's principle is leveraged to drive the equilibrium back toward the monomeric hemiacetal. This mechanistic understanding allows for the design of a continuous or semi-continuous process where impurities are managed in real-time rather than requiring costly batch rework.
Furthermore, the strict control of oxygen levels is mechanistically justified by the susceptibility of the alpha-carbon to radical oxidation. Even trace amounts of oxygen can initiate radical chains that lead to polymerization or the formation of peroxides, which are safety hazards in themselves. By sparging the reaction vessel with inert gases like nitrogen or argon to achieve oxygen levels below 5000 ppm, the process effectively shuts down these radical pathways. This level of control ensures that the impurity profile remains consistent, providing R&D teams with a predictable material for downstream coupling reactions. The result is a process that is not only chemically elegant but also robust enough for commercial scale-up of complex fluorinated intermediates.
How to Synthesize Alpha,Alpha-Difluoroacetaldehyde Hemiacetal Efficiently
Implementing this synthesis requires careful attention to the three distinct stages outlined in the patent: the catalytic hydrogenation, the controlled neutralization and distillation, and the final dimer conversion step. The process begins with the reaction of alpha,alpha-difluoroacetates with hydrogen gas in the presence of a base and the selected ruthenium catalyst. Following the reaction, the mixture must be neutralized to the specific pH window and distilled under inert atmosphere. Finally, the addition of alcohol converts any remaining dimers. The detailed standardized synthesis steps see the guide below.
- React alpha,alpha-difluoroacetates with hydrogen gas in the presence of a base and a ruthenium catalyst (such as Ru-MACHO) using an alcohol solvent to generate the crude hemiacetal mixture.
- Neutralize the reaction mixture to a pH between 3.5 and 10.0, then perform distillation under light-shielding conditions while maintaining gas-phase oxygen concentration below 5000 ppm.
- Add additional alcohol to the distilled mixture under continued low-oxygen and light-shielding conditions to convert residual dimers back into the target hemiacetal product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process offers tangible strategic benefits beyond mere chemical yield. The transition from hazardous hydride reagents to catalytic hydrogenation fundamentally changes the risk profile of the manufacturing operation. By eliminating the need for pyrophoric materials, the facility reduces its insurance liabilities and safety compliance burdens. This shift also simplifies the logistics of raw material storage, as hydrogen can be generated on-site or delivered in standard cylinders, unlike the specialized handling required for large quantities of lithium aluminum hydride. Consequently, this leads to substantial cost savings in terms of safety infrastructure and operational overhead.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric reducing agents significantly lowers the direct material costs associated with production. Furthermore, the ability to convert dimer by-products back into the valuable hemiacetal improves the overall mass balance of the process, meaning less raw material is wasted. This efficiency gain translates directly to a lower cost per kilogram of the final intermediate. Additionally, the reduction in hazardous waste generation decreases the expenses related to waste treatment and disposal, further enhancing the economic viability of the process for large-scale operations.
- Enhanced Supply Chain Reliability: The robustness of the ruthenium-catalyzed method ensures consistent batch quality, which is critical for maintaining uninterrupted API production schedules. By minimizing the formation of hard-to-remove impurities, the purification train is simplified, reducing the cycle time for each batch. This efficiency allows manufacturers to respond more agilely to market demand fluctuations. The stability of the intermediate under the prescribed storage conditions also means that inventory can be held with greater confidence, reducing the risk of stockouts due to material degradation during warehousing.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from pilot plant to multi-ton commercial production without the exponential increase in safety risks associated with hydride chemistry. The use of molecular hydrogen and recyclable ruthenium catalysts aligns with increasingly stringent environmental regulations regarding heavy metal discharge and solvent waste. This compliance future-proofs the supply chain against regulatory tightening, ensuring long-term operational continuity. The reduced environmental footprint also supports corporate sustainability goals, a key metric for modern pharmaceutical supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this synthesis route. These answers are derived directly from the experimental data and claims within patent CN110520403B, providing clarity on the operational parameters required for success. Understanding these nuances is essential for technology transfer teams evaluating the feasibility of adopting this method.
Q: Why is oxygen concentration critical during the purification of alpha,alpha-difluoroacetaldehyde hemiacetal?
A: Maintaining oxygen levels below 5000 ppm prevents oxidative degradation and stabilizes the hemiacetal against forming stable dimeric by-products, which are difficult to revert without specific acidic conditions.
Q: How does this patent address the issue of dimer formation compared to previous methods?
A: Unlike prior art where dimers were stable and required harsh acidic heating to decompose, this method utilizes a controlled pH range (3.5-10.0) and alcohol addition to efficiently convert dimers back to the desired hemiacetal without generating difluoroacetic acid.
Q: What specific catalysts are recommended for this hydrogenation process?
A: The patent highlights the efficacy of ruthenium pincer complexes, specifically those resembling the Ru-MACHO structure or supported ruthenium compounds, which offer high activity and selectivity under mild hydrogen pressure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha,Alpha-Difluoroacetaldehyde Hemiacetal Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality fluorinated intermediates play in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate balance of pH, oxygen, and catalysis described in this patent is maintained at every scale. We operate stringent purity specifications and utilize rigorous QC labs to verify that every batch of alpha,alpha-difluoroacetaldehyde hemiacetal meets the exacting standards required for global pharmaceutical applications. Our commitment to quality ensures that your downstream synthesis proceeds without interruption due to impurity-related failures.
We invite you to collaborate with us to leverage this advanced manufacturing technology for your specific project needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to secure a stable, cost-effective, and high-purity supply of this vital intermediate for your commercial success.
