Advanced Synthesis of CTPA Fluorescent Probe for Precision Lipid Droplet Imaging
The pharmaceutical and bio-imaging sectors are witnessing a paradigm shift in cellular diagnostics, driven by the urgent need to distinguish metabolic states in cancer research. Patent CN108440475B introduces a groundbreaking ratiometric fluorescent probe, chemically defined as (E)-7-(N,N-diethylamino)-3-(3-(4-(diphenylamine)phenyl)acryloyl)-2H-benzopyrone, commonly abbreviated as CTPA. This innovative molecule addresses the critical limitation of existing probes by offering specific sensitivity to lipid droplet polarity, a key biomarker for differentiating cancer cells from normal tissue. The probe leverages a unique combination of a coumarin fluorophore and a triphenylamine polarity-sensitive group, enabling two-photon excitation and aggregation-induced emission properties that significantly enhance imaging depth and resolution. As illustrated in the chemical structure below, the molecular architecture is meticulously designed to ensure electrical neutrality, facilitating superior localization within lipid-rich organelles.
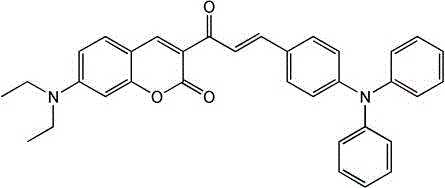
For R&D directors and procurement specialists seeking reliable pharmaceutical intermediates supplier partnerships, this technology represents a significant leap forward in diagnostic reagent manufacturing. The ability to detect lipid droplet polarity in vivo and in organs opens new avenues for drug discovery and pathological analysis, positioning CTPA as a high-value specialty chemical. The synthesis pathway described in the patent is not only robust but also aligns with green chemistry principles by utilizing common solvents and avoiding toxic heavy metal catalysts. This report delves into the technical nuances of this synthesis, offering a comprehensive analysis of its commercial viability and supply chain advantages for global stakeholders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for detecting lipid droplets, such as immunochemical staining or standard NMR imaging, have long plagued researchers with significant operational drawbacks and limited specificity. Conventional fluorescent probes often suffer from aggregation-caused quenching (ACQ), where fluorescence intensity diminishes at high concentrations, leading to inaccurate quantification in lipid-rich environments. Furthermore, many existing probes rely on single-photon excitation, which limits tissue penetration depth and increases background autofluorescence, thereby reducing the signal-to-noise ratio in complex biological samples. Most critically, current commercial probes fail to distinguish between lipid droplets of varying polarities, a crucial parameter for identifying metabolic shifts in cancer cells versus healthy cells. This lack of polarity sensitivity renders them ineffective for advanced oncological research where microenvironmental changes are subtle yet diagnostically vital.
The Novel Approach
The novel approach detailed in patent CN108440475B overcomes these hurdles through a sophisticated molecular design that integrates a ratiometric fluorescence mechanism with two-photon absorption capabilities. By employing a Twisted Intramolecular Charge Transfer (TICT) effect, the CTPA probe exhibits a distinct spectral shift in response to environmental polarity, allowing for precise differentiation of cellular states without the need for complex calibration. The synthesis strategy utilizes a straightforward condensation reaction that avoids the use of expensive transition metal catalysts, thereby simplifying the purification process and reducing the risk of metal contamination in the final product. This method ensures high optical stability and low cytotoxicity, making it safe for live-cell imaging and in vivo applications. The result is a robust, high-performance reagent that provides clear, ratio-based readouts of lipid droplet polarity, significantly enhancing the reliability of diagnostic data.
Mechanistic Insights into Piperidine-Catalyzed Knoevenagel Condensation
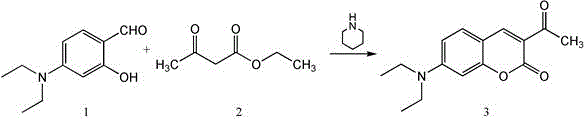
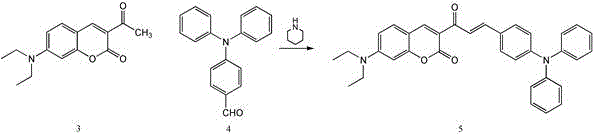
The core of the CTPA synthesis lies in a classic yet highly optimized Knoevenagel condensation, catalyzed by piperidine, which efficiently links the electron-rich coumarin scaffold with the polarity-sensitive triphenylamine aldehyde. In the first step, N,N-diethylaminosalicylaldehyde reacts with ethyl acetoacetate to form the 3-acetyl-7-(diethylamino)coumarin intermediate, a reaction that proceeds smoothly at room temperature in ethanol. The second step involves the condensation of this intermediate with 4-diphenylamine benzaldehyde under reflux conditions, where the piperidine catalyst facilitates the dehydration process to form the conjugated double bond essential for the probe's optical properties. This conjugation extends the pi-electron system, red-shifting the absorption and emission wavelengths into a range ideal for biological imaging while minimizing photodamage to living tissues. The reaction mechanism ensures high regioselectivity, preventing the formation of unwanted isomers that could compromise the probe's ratiometric accuracy.
Impurity control is inherently managed by the thermodynamic stability of the trans-alkene product formed during the condensation, which is the predominant species under the specified reflux conditions. The use of ethanol and acetonitrile as solvents not only provides excellent solubility for the reactants but also aids in the crystallization of the final product, effectively excluding side products and unreacted starting materials. The electrical neutrality of the final molecule is preserved throughout the synthesis, ensuring that no charged impurities interfere with the probe's ability to passively diffuse into lipid droplets. This mechanistic precision translates directly to a cleaner crude product, reducing the burden on downstream purification steps and ensuring a consistent quality profile suitable for sensitive biological assays. The high yield reported in the examples underscores the efficiency of this catalytic system in driving the reaction to completion.
How to Synthesize CTPA Efficiently
The synthesis of CTPA is designed for operational simplicity, requiring standard laboratory equipment and readily available reagents that facilitate easy technology transfer to manufacturing scales. The process begins with the formation of the coumarin core, followed by a condensation step that can be monitored via TLC or HPLC to ensure complete conversion. Detailed standardized operating procedures for temperature control, solvent ratios, and work-up protocols are critical to maintaining the high purity required for bio-imaging applications. For a comprehensive guide on the specific molar ratios, reaction times, and purification techniques validated in the patent, please refer to the structured synthesis protocol below.
- React N,N-diethylaminosalicylaldehyde with ethyl acetoacetate in ethanol using piperidine catalyst at room temperature to form the coumarin intermediate.
- Condense the coumarin intermediate with 4-diphenylamine benzaldehyde in an ethanol/acetonitrile mixture under reflux conditions.
- Purify the final orange solid product via filtration and recrystallization or column chromatography to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the CTPA synthesis route offers substantial cost reduction in pharmaceutical intermediates manufacturing by eliminating the need for precious metal catalysts and exotic reagents. The reliance on bulk commodities like ethanol, acetonitrile, and piperidine ensures a stable and predictable supply chain, insulating production from the volatility associated with specialized catalytic systems. The high yield achieved in the final step minimizes raw material waste, directly contributing to a lower cost of goods sold (COGS) and improved margin potential for large-scale producers. Furthermore, the purification process relies on standard techniques such as recrystallization and column chromatography, which are easily scalable and do not require capital-intensive specialized equipment. This operational simplicity allows for rapid scale-up from gram to kilogram quantities without significant process re-engineering.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the costly and time-consuming step of heavy metal scavenging, which is often a regulatory bottleneck in pharmaceutical production. By using organic base catalysis, the process simplifies the workflow and reduces the consumption of expensive purification resins. The high atom economy of the condensation reaction ensures that the majority of the starting mass is incorporated into the final product, drastically reducing waste disposal costs. Additionally, the use of common solvents allows for efficient recovery and recycling, further lowering the environmental and financial overhead of the manufacturing process.
- Enhanced Supply Chain Reliability: The starting materials, including salicylaldehyde derivatives and benzaldehyde analogs, are widely produced commodity chemicals with multiple global suppliers, ensuring robust supply continuity. This diversity in sourcing mitigates the risk of single-supplier dependency and allows procurement teams to negotiate favorable terms based on market availability. The stability of the intermediates and the final product under standard storage conditions simplifies logistics and warehousing, reducing the need for cold-chain transportation. Consequently, lead times for high-purity fluorescent probes can be significantly shortened, enabling faster response to research demands.
- Scalability and Environmental Compliance: The synthesis operates at moderate temperatures and atmospheric pressure, presenting minimal safety hazards and allowing for implementation in standard multipurpose reactors. The solvent system is compatible with existing waste treatment infrastructure, and the absence of halogenated solvents in the primary reaction steps aligns with increasingly stringent environmental regulations. The high yield and selectivity of the process reduce the volume of chemical waste generated per unit of product, supporting sustainability goals. This makes the technology attractive for contract manufacturing organizations (CMOs) looking to expand their portfolio of eco-friendly fine chemical processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of the CTPA fluorescent probe. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation. Understanding these details is essential for evaluating the feasibility of integrating this probe into your research or product development pipeline.
Q: What is the primary advantage of the CTPA probe over traditional lipid droplet stains?
A: Unlike traditional stains that suffer from aggregation quenching or single-photon limitations, CTPA offers ratiometric detection with two-photon properties, allowing for precise differentiation of lipid droplet polarity in cancer versus normal cells without background interference.
Q: Is the synthesis of CTPA scalable for commercial production?
A: Yes, the synthesis utilizes readily available starting materials like salicylaldehyde derivatives and employs standard organic solvents such as ethanol. The process achieves high yields (up to 90%) and uses simple purification methods like recrystallization, making it highly suitable for scale-up.
Q: How does the probe achieve specificity for lipid droplets?
A: The probe molecule is electrically neutral, which facilitates its passive diffusion and specific localization within the hydrophobic core of lipid droplets. Additionally, its Twisted Intramolecular Charge Transfer (TICT) mechanism ensures a specific fluorescence response to the polarity of the microenvironment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable CTPA Fluorescent Probe Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, leveraging deep expertise in organic chemistry to deliver complex molecules like CTPA with uncompromising quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your demand whether for preclinical research or full-scale manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee the optical performance of every batch. Our commitment to process optimization means we can offer competitive pricing without sacrificing the high standards required for bio-imaging reagents.
We invite you to collaborate with us to explore the full potential of this advanced fluorescent probe technology for your specific applications. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to support your project timelines and help you accelerate your path to market.
