Advanced Benzothiadiazole-TB Derivatives: Scalable Synthesis for OLED and Photodynamic Therapy Applications
The landscape of organic optoelectronic materials and biological probes is constantly evolving, driven by the need for molecules that combine high quantum efficiency with structural rigidity. Patent CN113416196B introduces a groundbreaking class of benzothiadiazole-Tröger's base (TB) compounds that address critical limitations in current fluorescent materials. These novel derivatives, characterized by the general structure A where R can be hydrogen, alkoxy, hydroxyl, or alkyl groups, demonstrate exceptional optical properties including large Stokes shifts and robust solid-state luminescence. The integration of the electron-deficient 2,1,3-benzothiadiazole (BTD) unit onto the rigid TB skeleton creates a push-pull electronic system that significantly enhances charge transfer capabilities. This structural innovation not only positions these compounds as prime candidates for next-generation OLED emitters but also unlocks their potential as highly sensitive fluorescent probes for metal ions like Fe3+ and viscosity changes in physiological environments.
For procurement managers and supply chain directors in the fine chemical sector, the significance of this patent lies in its reliance on commercially available starting materials and standard catalytic processes. The synthesis pathway avoids exotic reagents, utilizing commodity chemicals such as p-bromoaniline and paraformaldehyde, which ensures a stable and cost-effective supply chain. Furthermore, the scalability of the Tröger's base formation and subsequent Suzuki coupling reactions suggests that commercial scale-up of complex electronic chemical manufacturing can be achieved with minimal process re-engineering. This makes the technology highly attractive for industrial adoption where consistency and raw material availability are paramount concerns.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional photosensitizers and fluorescent probes often suffer from aggregation-caused quenching (ACQ), where emission intensity drops drastically in the solid state or high-concentration solutions, severely limiting their utility in OLED devices and biological imaging. Many existing Tröger's base derivatives lack the necessary electronic modulation to achieve large Stokes shifts, leading to self-absorption issues that reduce overall device efficiency. Additionally, conventional synthesis routes for functionalized TB cores often involve harsh conditions or multiple protection-deprotection steps that lower overall yields and increase waste generation. The lack of structural diversity in previous TB-based reports has restricted their application scope, with very few examples demonstrating effective performance in photodynamic therapy (PDT) or specific ion recognition tasks.
The Novel Approach
The methodology outlined in CN113416196B overcomes these hurdles by strategically incorporating the benzothiadiazole moiety, which acts as a strong electron acceptor to facilitate intramolecular charge transfer (ICT). This design effectively suppresses non-radiative decay pathways and enhances the rigidity of the molecular framework, thereby mitigating ACQ effects through the restriction of intramolecular rotation (RIR) mechanism. The synthetic strategy employs a modular approach where the TB core is functionalized via lithiation-borylation followed by palladium-catalyzed cross-coupling. This allows for precise control over the substitution pattern, enabling the fine-tuning of optical properties such as emission wavelength and quantum yield without compromising the structural integrity of the TB scaffold.
Mechanistic Insights into Suzuki-Miyaura Coupling and Lithiation
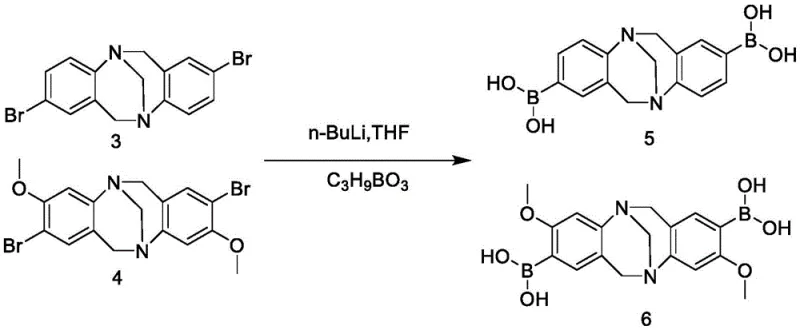
The core of this synthetic achievement relies on a sophisticated sequence of organometallic transformations that ensure high regioselectivity and yield. The process begins with the formation of the dibromo-Tröger's base intermediates (3 and 4), which serve as the foundational scaffold. Subsequent lithiation using n-butyllithium at cryogenic temperatures (-78°C) is critical for generating the reactive aryl lithium species at the specific positions ortho to the nitrogen atoms. This step requires precise temperature control to prevent side reactions such as halogen-metal exchange at the wrong positions or decomposition of the sensitive TB bridge. The resulting organolithium intermediates are then quenched with trimethyl borate to form the corresponding boronic acids (5 and 6), which are stable enough for isolation and purification, ensuring high purity before the final coupling step.
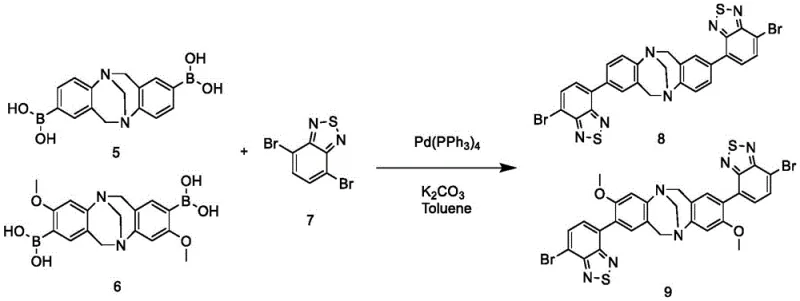
The final assembly of the target molecules involves a Suzuki-Miyaura cross-coupling reaction between the bis-boronic acid intermediates and 4,7-dibromo-benzothiadiazole. This reaction is catalyzed by tetrakis(triphenylphosphine)palladium(0) in the presence of a base like potassium carbonate. The mechanism involves the oxidative addition of the palladium catalyst to the carbon-bromine bond of the benzothiadiazole, followed by transmetallation with the boronic acid species and reductive elimination to form the new carbon-carbon bond. This step is crucial for extending the pi-conjugation system, which directly correlates to the red-shifted emission and enhanced molar absorptivity observed in the final products. The use of standard phosphine ligands and mild bases makes this process highly adaptable for large-scale production while maintaining the stringent purity specifications required for electronic applications.
How to Synthesize Benzothiadiazole-TB Compounds Efficiently
The synthesis protocol described in the patent provides a robust framework for producing these high-value intermediates with consistent quality. The process is divided into four distinct stages: the acid-catalyzed condensation to form the TB core, the low-temperature lithiation-borylation to install boron handles, the palladium-catalyzed coupling to attach the benzothiadiazole units, and finally, optional demethylation to generate hydroxyl-substituted variants. Each step has been optimized to maximize yield and minimize impurities, with detailed workup procedures including recrystallization and column chromatography. For R&D teams looking to replicate or scale this chemistry, adherence to the specified stoichiometry and reaction conditions, particularly the anhydrous environment for lithiation, is essential for success.
- Condensation of bromoaniline derivatives with paraformaldehyde in trifluoroacetic acid to form the Tröger's base core.
- Regioselective lithiation at -78°C followed by quenching with trimethyl borate to generate boronic acid intermediates.
- Palladium-catalyzed Suzuki coupling with 4,7-dibromo-benzothiadiazole to finalize the conjugated system.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the synthesis route presented in this patent offers significant advantages in terms of cost structure and supply chain resilience. The reliance on bulk commodity chemicals such as bromoanilines and paraformaldehyde means that raw material costs are inherently low and subject to less volatility compared to specialized heterocyclic building blocks. Furthermore, the use of well-established catalytic systems like palladium-phosphine complexes allows manufacturers to leverage existing infrastructure and expertise, reducing the capital expenditure required for technology transfer. The ability to produce multiple derivatives (Compounds 8, 9, and 10) from common intermediates enhances production flexibility, allowing suppliers to respond quickly to varying market demands for different optical properties without maintaining separate inventory lines for each product.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for expensive transition metal catalysts beyond standard palladium sources, and the purification steps rely on conventional techniques like recrystallization rather than costly preparative HPLC. This streamlined process significantly reduces the operational expenditure associated with solvent consumption and waste disposal. By avoiding complex multi-step protection strategies, the overall atom economy is improved, leading to substantial cost savings in the final production of high-purity OLED material precursors.
- Enhanced Supply Chain Reliability: Since the key starting materials are widely produced industrial chemicals, the risk of supply disruption is minimized. The robustness of the Tröger's base formation reaction ensures high conversion rates, reducing the dependency on scarce reagents. This stability is crucial for long-term contracts in the electronics and pharmaceutical sectors, where consistent delivery schedules are mandatory. The modular nature of the synthesis also allows for decentralized production of intermediates, further strengthening the supply chain against regional disruptions.
- Scalability and Environmental Compliance: The reaction conditions, primarily involving moderate temperatures and standard organic solvents like toluene and dichloromethane, are easily scalable from gram to kilogram scales without significant safety hazards. The process generates manageable waste streams that can be treated using standard industrial effluent protocols. This alignment with environmental compliance standards facilitates faster regulatory approval for commercial manufacturing, accelerating the time-to-market for new products based on this technology.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new class of compounds is vital for stakeholders evaluating their potential integration into existing product lines. The following questions address common inquiries regarding the stability, application scope, and handling of benzothiadiazole-TB derivatives. These answers are derived directly from the experimental data and characterization results provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What are the primary optical advantages of Benzothiadiazole-TB compounds?
A: These compounds exhibit large Stokes shifts, excellent solid-state luminescence due to restricted intramolecular rotation (RIR), and significant fluorescence enhancement in high-viscosity environments, making them ideal for OLEDs and viscosity sensing.
Q: How does the synthesis ensure high purity for electronic applications?
A: The process utilizes robust purification methods including recrystallization and column chromatography at critical intermediate stages, ensuring the removal of palladium residues and unreacted halides essential for electronic grade materials.
Q: Can these compounds be used for biological imaging?
A: Yes, specifically Compound 9 demonstrates low dark toxicity and high phototoxicity against A549 cancer cells under light irradiation, validating its potential as a photosensitizer for photodynamic therapy and cellular imaging.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzothiadiazole-TB Compound Supplier
As the demand for advanced organic electronic materials and specialized biological probes continues to surge, having a manufacturing partner with deep technical expertise is essential. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped to handle sensitive organometallic reactions under strict inert atmospheres, guaranteeing the stringent purity specifications required for high-performance OLED materials and pharmaceutical intermediates. With rigorous QC labs and a commitment to process optimization, we deliver products that consistently meet the highest industry standards.
We invite you to collaborate with us to explore the full potential of these innovative benzothiadiazole-TB compounds in your applications. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to support your R&D and commercialization efforts, ensuring a seamless transition from laboratory discovery to industrial reality.
