Advanced Catalytic Synthesis of Dicoumarin Derivatives for Commercial Scale-Up
Advanced Catalytic Synthesis of Dicoumarin Derivatives for Commercial Scale-Up
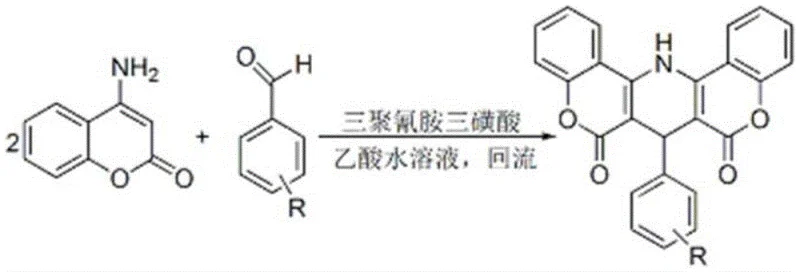
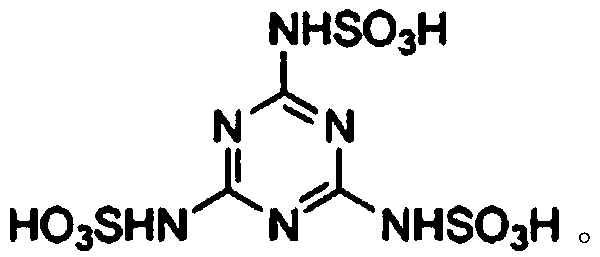
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways for synthesizing complex heterocyclic scaffolds. Patent CN110016042B introduces a groundbreaking methodology for the preparation of 1,4-dihydropyrido[3,2-c][5,6-c]dicoumarin derivatives, utilizing a novel catalytic system based on melamine trisulfonic acid. This technology represents a significant leap forward from traditional acid-catalyzed condensations, addressing critical pain points such as long reaction times, excessive catalyst loading, and cumbersome purification protocols. By leveraging the unique Brønsted acidity of melamine trisulfonic acid in an aqueous acetic acid medium, this process achieves high conversion rates under mild reflux conditions, making it an ideal candidate for reliable pharmaceutical intermediate supplier operations aiming for green chemistry compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyridocoumarin derivatives has relied heavily on protonic acids like potassium bisulfate (KHSO4) or expensive acidic ionic liquids. While KHSO4 provides a viable pathway, it suffers from relatively low acidity, necessitating high molar equivalents of the catalyst and extended reaction durations to drive the condensation to completion. Furthermore, the purification of products synthesized via these traditional routes often involves multiple washing steps and recrystallization from glacial acetic acid, which generates substantial solvent waste and increases processing time. On the other hand, while acidic ionic liquids offer improved catalytic performance, their synthesis is complex and costly, and separating the product from the viscous ionic medium often requires intricate workup procedures that hinder industrial scalability and increase the overall cost of goods sold.
The Novel Approach
The innovative method disclosed in the patent utilizes melamine trisulfonic acid as a highly active, reusable solid acid catalyst dispersed in an aqueous acetic acid solution. This system effectively overcomes the kinetic barriers associated with weaker acids, enabling the reaction to proceed rapidly at atmospheric pressure with significantly reduced catalyst loading. The use of an aqueous solvent system not only lowers the environmental footprint by reducing volatile organic compound emissions but also facilitates a unique precipitation mechanism where the product crystallizes directly upon cooling. This eliminates the need for complex extraction or chromatographic purification, allowing for a simple suction filtration and drying process that yields high-purity materials suitable for downstream pharmaceutical applications without further refinement.
Mechanistic Insights into Melamine Trisulfonic Acid Catalysis
The exceptional performance of this catalytic system stems from the molecular structure of melamine trisulfonic acid, which features three strongly electron-withdrawing sulfonic acid groups attached to a melamine core. This configuration creates a potent Brønsted acid environment that effectively activates the carbonyl group of the aromatic aldehyde towards nucleophilic attack by the active methylene group of the 4-aminocoumarin. The synergy between the strong acidity and the solubility characteristics in the aqueous acetic acid medium ensures a homogeneous reaction phase during the reflux period, maximizing molecular collisions and reaction efficiency. This robust activation allows the reaction to tolerate a wide range of substituents on the aromatic aldehyde, including electron-donating and electron-withdrawing groups, without compromising yield or selectivity.
Impurity control is another critical aspect where this mechanistic approach excels. The mild yet effective acidity prevents the formation of polymeric by-products or degradation of the sensitive coumarin ring, which can occur under harsher acidic conditions or prolonged heating. The specific interaction between the catalyst and the reactants promotes a clean Knoevenagel-Michael cascade sequence, leading to the formation of the pyrido-dicoumarin scaffold with high regioselectivity. Consequently, the crude product obtained after filtration exhibits purity levels exceeding 98%, as confirmed by HPLC analysis in the patent examples. This inherent selectivity reduces the burden on quality control laboratories and minimizes the risk of genotoxic impurities carrying over into the final active pharmaceutical ingredient.
How to Synthesize 1,4-Dihydropyrido[3,2-c][5,6-c]dicoumarin Efficiently
The operational simplicity of this synthesis route makes it highly attractive for process chemists looking to transfer laboratory protocols to pilot or production scales. The procedure involves charging a standard three-neck flask with the aromatic aldehyde, 4-aminocoumarin, and a catalytic amount of melamine trisulfonic acid in an aqueous acetic acid solution. The mixture is then heated to reflux, typically requiring between 48 to 115 minutes depending on the electronic nature of the aldehyde substituent. Upon completion, the reaction mass is cooled to ambient temperature, inducing the precipitation of the product as a white crystalline solid which is then isolated via filtration. For detailed standardized operating procedures and safety guidelines regarding this specific catalytic transformation, please refer to the technical guide below.
- Mix aromatic aldehyde, 4-aminocoumarin, and melamine trisulfonic acid catalyst in an aqueous acetic acid solution within a three-neck flask equipped with a condenser.
- Heat the reaction mixture to reflux under atmospheric pressure for 48 to 115 minutes while monitoring progress via TLC.
- Cool the mixture to room temperature, perform suction filtration on the precipitated solid, and vacuum dry the residue to obtain the pure product without recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this melamine trisulfonic acid catalyzed process offers transformative economic benefits. The primary advantage lies in the drastic simplification of the downstream processing unit operations. By eliminating the need for recrystallization and extensive solvent washing, manufacturers can significantly reduce utility consumption, labor hours, and solvent procurement costs. The ability to recycle the catalytic filtrate directly for subsequent batches further amplifies these savings, as it minimizes the consumption of fresh catalyst and reduces the volume of hazardous waste requiring treatment. This streamlined workflow translates directly into a lower cost base for high-purity pharmaceutical intermediates, providing a competitive edge in price-sensitive markets.
- Cost Reduction in Manufacturing: The elimination of expensive and difficult-to-synthesize ionic liquid catalysts in favor of readily available melamine trisulfonic acid results in substantial raw material cost savings. Furthermore, the high atom economy and the avoidance of energy-intensive purification steps like column chromatography or multiple recrystallizations lead to a leaner manufacturing process. The reduced reaction time compared to traditional KHSO4 methods also increases reactor throughput, allowing facilities to produce more batches within the same timeframe, thereby optimizing capital expenditure and operational efficiency without compromising product quality.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, specifically various aromatic aldehydes and 4-aminocoumarin, are commodity chemicals with robust global supply chains, ensuring consistent availability and price stability. The catalyst itself is stable and easy to store, removing the logistical complexities associated with handling moisture-sensitive or air-sensitive reagents. This reliability in raw material sourcing, combined with the robustness of the reaction conditions which tolerate minor variations in stoichiometry, ensures a steady and predictable output of intermediates, mitigating the risk of production delays for downstream drug manufacturers.
- Scalability and Environmental Compliance: The use of an aqueous acetic acid solvent system aligns perfectly with modern green chemistry principles, significantly reducing the environmental impact compared to processes relying on chlorinated solvents or pure organic acids. The simplicity of the workup, involving only filtration and drying, minimizes the generation of liquid waste streams, easing the burden on wastewater treatment facilities. This environmentally friendly profile facilitates regulatory approval and supports corporate sustainability goals, making the commercial scale-up of complex pharmaceutical intermediates smoother and more socially responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic technology in industrial settings. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process robustness and product quality. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into existing production lines.
Q: What are the advantages of using melamine trisulfonic acid over traditional catalysts?
A: Melamine trisulfonic acid offers superior catalytic activity compared to potassium bisulfate, allowing for lower catalyst loading and shorter reaction times. Unlike acidic ionic liquids, it is easier to handle and the catalytic system can be recycled directly from the filtrate, significantly reducing waste and operational costs.
Q: Is the purification process complex for these dicoumarin derivatives?
A: No, the purification process is remarkably simplified. The product precipitates directly from the reaction mixture upon cooling. It requires only suction filtration and vacuum drying to achieve high purity (98-99%), eliminating the need for energy-intensive recrystallization or column chromatography steps often required in conventional methods.
Q: Can the catalytic system be reused for multiple batches?
A: Yes, the patent demonstrates that the filtrate containing the melamine trisulfonic acid and acetic acid aqueous solution can be reused directly for subsequent batches by simply adding fresh reactants. This recyclability maintains high yields over multiple cycles, enhancing the overall economic efficiency of the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Dihydropyrido[3,2-c][5,6-c]dicoumarin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our team of expert process chemists has extensively evaluated the melamine trisulfonic acid catalyzed route and possesses the technical capability to implement this efficient protocol at scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the highest international standards for safety and efficacy.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain. By leveraging our expertise, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our commitment to technological excellence can drive value and reliability in your drug development pipeline.
