Advanced Synthesis of 4-Fluorobenzoylacetonitrile for Commercial Scale-Up of Complex Pharmaceutical Intermediates
Advanced Synthesis of 4-Fluorobenzoylacetonitrile for Commercial Scale-Up of Complex Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks robust synthetic routes for critical intermediates that balance efficiency, safety, and cost-effectiveness. Patent CN111233707B introduces a groundbreaking methodology for the synthesis and refining of 4-fluorobenzoylacetonitrile, a pivotal building block in the production of Blonanserin, a potent antipsychotic agent. This technical disclosure represents a significant departure from traditional base-catalyzed condensation or oxidative pathways, utilizing a superacid-mediated coupling strategy that dramatically improves conversion rates and operational safety. By leveraging trifluoromethanesulfonic acid (TfOH) as a catalyst, the process achieves exceptional yields exceeding 92% with purity levels surpassing 99.8%, addressing long-standing challenges in impurity profiles and downstream processing. For R&D directors and procurement specialists, this innovation offers a compelling alternative to legacy methods, promising enhanced supply chain reliability and reduced manufacturing costs through simplified unit operations.
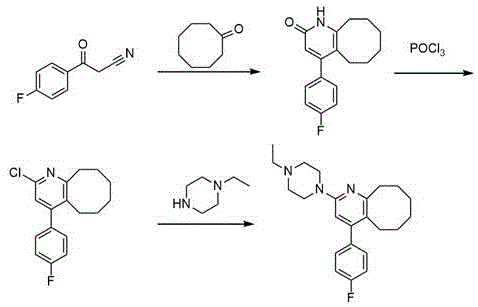
The strategic importance of 4-fluorobenzoylacetonitrile extends beyond its immediate application; it serves as a versatile scaffold for constructing complex heterocyclic systems found in modern CNS drugs. As illustrated in the reaction scheme leading to Blonanserin, the integrity of this intermediate directly impacts the quality of the final API. Consequently, securing a reliable pharma intermediate supplier capable of delivering this material with consistent stereochemical and chemical purity is paramount for drug developers. The patented method not only optimizes the synthesis of this specific nitrile but also sets a new benchmark for electrophilic aromatic substitutions involving electron-deficient arenes, providing a template for cost reduction in pharmaceutical intermediates manufacturing across similar chemical classes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-fluorobenzoylacetonitrile has been plagued by inefficiencies inherent to base-catalyzed condensations and oxidative protocols. One prevalent prior art method involves the condensation of p-fluorobenzonitrile with acetonitrile under strong basic conditions, a process notorious for its low yield of approximately 63% and extreme sensitivity to temperature fluctuations. Furthermore, alternative routes utilizing methyl p-fluorobenzoate often suffer from the formation of stubborn ether impurities due to nucleophilic substitution on the fluorine atom under harsh alkaline environments, making it exceptionally difficult to achieve purity levels above 99% without extensive and costly purification steps. Another oxidative approach using copper chloride catalysts requires prolonged reaction times exceeding three days and still fails to deliver competitive yields, creating bottlenecks in production schedules and inflating inventory holding costs for supply chain managers.
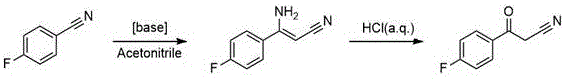
The Novel Approach
In stark contrast, the novel approach detailed in CN111233707B employs a direct acylation strategy using fluorobenzene and malononitrile mediated by trifluoromethanesulfonic acid. This electrophilic substitution bypasses the need for pre-functionalized starting materials like esters or nitriles that require activation, thereby shortening the synthetic sequence and reducing raw material costs. The reaction proceeds smoothly at moderate temperatures between 70°C and 80°C, completing within 8 to 12 hours, which represents a drastic improvement in throughput compared to multi-day oxidative methods. Moreover, the use of a superacid catalyst minimizes side reactions such as ether formation, resulting in a cleaner crude profile that is amenable to simple recrystallization rather than complex chromatographic separation, fundamentally altering the economic landscape of producing high-purity OLED material or pharmaceutical precursors.
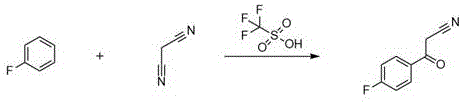
Mechanistic Insights into TfOH-Catalyzed Electrophilic Acylation
The core of this technological breakthrough lies in the unique activation mechanism facilitated by trifluoromethanesulfonic acid, one of the strongest known Brønsted acids. In this system, TfOH likely protonates the malononitrile or facilitates the formation of a highly reactive nitrilium ion species, which then acts as a potent electrophile attacking the electron-rich positions of the fluorobenzene ring. Despite the deactivating nature of the fluorine substituent, the superacidic environment enhances the electrophilicity of the acylating agent sufficiently to drive the reaction forward with high regioselectivity for the para-position. This mechanistic pathway avoids the generation of carbanions typical in base-catalyzed routes, thereby eliminating the risk of self-condensation of the nitrile or hydrolysis of sensitive functional groups, ensuring a robust and reproducible reaction profile suitable for commercial scale-up of complex polymer additives or fine chemicals.
Impurity control is intrinsically built into this mechanism by avoiding the harsh nucleophilic conditions that lead to defluorination. In traditional base-mediated reactions, the fluoride ion is a good leaving group when attacked by alkoxides or strong bases, leading to the formation of ethoxy- or methoxy-substituted byproducts that share similar physical properties with the target molecule. By shifting to an acidic regime, the nucleophilicity of potential contaminants is suppressed, and the fluorine atom remains intact on the aromatic ring. The subsequent refining step utilizes a binary solvent system of alcohol and small molecular alkanes, exploiting subtle solubility differences to exclude trace organic impurities. This dual strategy of mechanistic selectivity and physical purification ensures that the final product meets stringent purity specifications required for regulatory submission and clinical trials.
How to Synthesize 4-Fluorobenzoylacetonitrile Efficiently
The operational protocol for this synthesis is designed for scalability and safety, utilizing standard reactor equipment and readily available solvents. The process begins with the dissolution of malononitrile in a chlorinated solvent such as chloroform, followed by the controlled addition of fluorobenzene and the acid catalyst. Maintaining precise thermal control during the exothermic addition is critical to maximizing yield and minimizing degradation. Following the reaction, a straightforward aqueous workup isolates the crude material, which is then subjected to a specialized recrystallization procedure. Detailed standardized synthesis steps see the guide below.
- Dissolve malononitrile in a chlorinated solvent such as chloroform, then add fluorobenzene and trifluoromethanesulfonic acid while maintaining a temperature between 70°C and 80°C for 8 to 12 hours.
- Quench the reaction mixture with ice water, separate the organic phase, extract the aqueous layer with dichloromethane, and concentrate under reduced pressure to obtain the crude product.
- Purify the crude material by dissolving in ethanol at 55°C, dropwise adding n-hexane to induce turbidity, cooling to 0-10°C, and filtering to isolate white flaky crystals with >99.8% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route translates into tangible operational efficiencies and risk mitigation. The shift from expensive, pre-functionalized starting materials to commodity chemicals like fluorobenzene and malononitrile significantly reduces the raw material cost base. Additionally, the high conversion rate minimizes waste generation and solvent consumption per kilogram of product, aligning with green chemistry principles and reducing disposal costs. The simplified purification process eliminates the need for resource-intensive column chromatography or multiple distillation passes, allowing for faster batch turnover and improved asset utilization in manufacturing facilities.
- Cost Reduction in Manufacturing: The elimination of expensive catalysts like copper salts and the avoidance of low-yielding steps drastically lowers the cost of goods sold. By achieving yields over 92% compared to the historical average of 63%, the process effectively increases capacity without requiring additional capital investment in reactor volume. Furthermore, the use of common solvents like chloroform and ethanol, which are easily recovered and recycled, contributes to substantial cost savings in solvent procurement and waste management budgets.
- Enhanced Supply Chain Reliability: Relying on commodity feedstocks such as fluorobenzene ensures a stable supply base that is less susceptible to market volatility compared to specialized esters or nitriles. The robustness of the reaction conditions, which tolerate minor variations in temperature and stoichiometry without catastrophic failure, reduces the risk of batch failures and production delays. This reliability is crucial for maintaining continuous supply lines to downstream API manufacturers, preventing costly stockouts and ensuring timely delivery of critical medicinal intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in multi-hundred-gram batches with consistent results, indicating readiness for ton-scale production. The absence of heavy metal catalysts simplifies the environmental compliance profile, removing the need for complex heavy metal scavenging steps and reducing the burden on wastewater treatment systems. This ease of scale-up facilitates the rapid transition from pilot plant to commercial production, enabling faster time-to-market for new drug formulations dependent on this key intermediate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on yield expectations, purification strategies, and safety considerations for potential licensees or manufacturing partners.
Q: What are the primary advantages of the triflic acid catalyzed method over traditional condensation routes?
A: The triflic acid method eliminates the need for strong bases like NaH which cause side reactions, resulting in significantly higher yields (over 92%) compared to the 63% typical of older base-catalyzed methods, while simplifying purification.
Q: How is high purity achieved in the refining process of 4-fluorobenzoylacetonitrile?
A: High purity (>99.8%) is achieved through a specific recrystallization technique using a binary solvent system of ethanol and small molecular alkanes like n-hexane, which effectively removes unreacted starting materials and byproducts.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process utilizes commercially available raw materials like fluorobenzene and malononitrile, operates at moderate temperatures (70-80°C), and avoids hazardous reagents, making it highly scalable and safe for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Fluorobenzoylacetonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of pharmaceutical development pipelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering 4-fluorobenzoylacetonitrile with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific applications. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain resilience and drive down overall production costs.
