Revolutionizing Statin Production: Green One-Pot Synthesis for Commercial Scale-Up
The pharmaceutical industry is currently undergoing a paradigm shift towards sustainable manufacturing, driven by stringent environmental regulations and the economic necessity of process intensification. Patent CN110963951A represents a significant technological breakthrough in this domain, specifically addressing the synthesis of the simvastatin side chain, methyl 3-[(2,2-dimethyl-1-oxobutyl)thio]propionate. This intermediate is critical for the production of simvastatin, a widely prescribed HMG-CoA reductase inhibitor used to manage cholesterol levels and prevent cardiovascular disease. The disclosed method departs radically from conventional multi-step batch processes by employing a novel one-pot, solvent-free strategy. By integrating the acyl chlorination of 2,2-dimethylbutyric acid directly with the subsequent thioesterification using methyl 3-mercaptopropionate, the invention eliminates the need for volatile organic compounds (VOCs) and stoichiometric acid-binding agents. This approach not only streamlines the operational workflow but also fundamentally alters the impurity profile of the final product, offering a compelling value proposition for reliable simvastatin side chain supplier partnerships focused on long-term sustainability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this key statin intermediate has been plagued by inefficiencies inherent to traditional solution-phase chemistry. Prior art, such as the methods described in literature by Liu Yusheng or patents like CN105837482 and US9399785, typically relies heavily on the use of organic solvents such as dichloromethane, tetrahydrofuran, or isopropyl acetate. These solvents necessitate energy-intensive recovery and concentration steps, significantly inflating the utility costs associated with cost reduction in pharmaceutical intermediates manufacturing. Furthermore, these conventional routes invariably require the addition of acid-binding agents like triethylamine, diisopropylethylamine, or pyridine to neutralize the hydrogen chloride generated during the reaction. The reliance on these amines introduces severe downstream processing challenges; for instance, triethylamine is difficult to recover completely, leading to elevated ammonia nitrogen levels and Chemical Oxygen Demand (COD) in wastewater streams. Additionally, the formation of solid waste, such as ammonium chloride salts when using ammonia gas, creates disposal bottlenecks that complicate regulatory compliance and increase the overall environmental footprint of the production facility.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN110963951A leverages a neat, solvent-free reaction environment that maximizes atomic economy and process safety. The core innovation lies in the direct thermal conversion of 2,2-dimethylbutyric acid to its acid chloride using thionyl chloride, followed immediately by the addition of the thiol component without isolating the reactive intermediate. This one-pot methodology effectively removes the unit operations associated with solvent exchange, filtration of salt byproducts, and extensive washing procedures. By operating without an acid-binding agent, the process avoids the generation of amine salts entirely, resulting in a much cleaner reaction matrix. The elimination of solvent handling also mitigates the risks associated with flammable vapor accumulation and reduces the potential for operator exposure to malodorous sulfur-containing compounds during material transfer. This streamlined approach not only enhances the commercial scale-up of complex statin side chains but also ensures a higher degree of consistency in batch-to-batch quality, which is paramount for GMP-compliant API production.
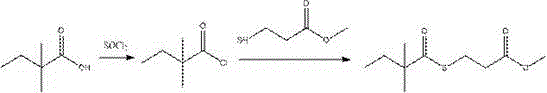
Mechanistic Insights into Solvent-Free Thioesterification
The mechanistic elegance of this process stems from the precise thermal management of the acyl chlorination and subsequent nucleophilic attack. In the first stage, thionyl chloride reacts with 2,2-dimethylbutyric acid to form the corresponding acid chloride, releasing sulfur dioxide and hydrogen chloride gases which are vented or scrubbed, driving the equilibrium forward without the need for chemical scavengers. The absence of solvent increases the effective concentration of the reactants, thereby accelerating the reaction kinetics according to the law of mass action. When methyl 3-mercaptopropionate is introduced in the second stage, the highly electrophilic acid chloride undergoes rapid thioesterification. The lack of a competing base means that the reaction relies solely on the nucleophilicity of the thiol group and the thermal energy provided (50-115°C). This specific condition prevents the formation of quaternary ammonium impurities that are notoriously difficult to remove via standard distillation. From a kinetic perspective, the elevated temperatures permissible in a solvent-free system (up to 115°C) ensure that the activation energy barrier is overcome efficiently, allowing for shorter reaction times compared to ambient temperature solution reactions.
Impurity control is another critical aspect where this mechanism offers distinct advantages. In traditional base-mediated reactions, side reactions such as the hydrolysis of the acid chloride or the formation of amide byproducts (if amines are present) can degrade the yield and complicate purification. By excluding water and amines from the reaction matrix, the pathway to the desired thioester becomes highly selective. The patent data indicates that controlling the mass ratio of thionyl chloride to acid (1:1.1 to 1:1.6) is sufficient to drive the conversion to completion without excessive reagent waste. Furthermore, the direct use of the crude acid chloride in the second step avoids the degradation that might occur during isolation and storage of the unstable acid chloride intermediate. This results in high-purity simvastatin intermediates with purity levels consistently above 98.9%, as evidenced by the experimental examples, minimizing the need for costly recrystallization or chromatographic purification steps that would otherwise erode profit margins.
How to Synthesize Methyl 3-[(2,2-dimethyl-1-oxobutyl)thio]propionate Efficiently
The implementation of this green synthesis route requires careful attention to thermal profiles and stoichiometry to maximize the benefits of the one-pot design. The process begins with the charging of 2,2-dimethylbutyric acid and thionyl chloride into a dry reaction vessel equipped with efficient gas scrubbing capabilities. The mixture is heated to a range of 65-100°C for 5-8 hours to ensure complete conversion to the acid chloride, followed by the removal of excess thionyl chloride and low-boiling byproducts under reduced pressure. Once the crude acid chloride is obtained, methyl 3-mercaptopropionate is added directly to the same vessel. The temperature is then adjusted to between 50°C and 115°C depending on the desired reaction rate, maintaining the mixture for 5-18 hours. The final product is isolated via vacuum distillation, collecting the fraction boiling between 137-152°C. For a detailed breakdown of the specific operational parameters and safety protocols required for this synthesis, please refer to the standardized guide below.
- Charge 2,2-dimethylbutyric acid and thionyl chloride into a reaction vessel and heat to 65-100°C for acyl chlorination.
- Distill off low-boiling substances below 120°C to obtain crude dimethylbutyryl chloride without isolation.
- Directly add methyl 3-mercaptopropionate to the crude acid chloride and heat to 50-115°C for thioesterification.
- Perform reduced pressure distillation to collect fractions at 137-152°C, yielding high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this solvent-free technology translates into tangible strategic advantages beyond mere technical novelty. The primary driver of value is the drastic simplification of the Bill of Materials (BOM). By eliminating the need for bulk purchases of organic solvents like dichloromethane and expensive acid scavengers like triethylamine, the direct material costs are significantly lowered. Moreover, the removal of solvent recovery infrastructure reduces the capital expenditure (CAPEX) required for plant equipment and lowers the operational expenditure (OPEX) related to energy consumption for distillation and drying. This lean manufacturing approach allows for reducing lead time for high-purity pharmaceutical intermediates because the overall cycle time per batch is compressed; there are no delays associated with solvent swapping, filtration of salt cakes, or extended drying periods to meet residual solvent limits. Consequently, suppliers utilizing this technology can offer more competitive pricing structures and more flexible delivery schedules, insulating buyers from volatility in the solvent market.
- Cost Reduction in Manufacturing: The economic impact of removing solvents and acid-binding agents is profound. Traditional processes incur hidden costs related to the disposal of hazardous waste streams containing amines and chlorinated solvents, which are subject to increasingly strict environmental levies. By generating a cleaner waste profile primarily consisting of recyclable thionyl chloride derivatives and minimal organic residue, the cost of waste treatment is substantially decreased. Additionally, the higher atom economy of the one-pot method means that less raw material is wasted in side reactions or lost during multiple work-up stages. This efficiency gain allows manufacturers to produce more product per unit of input, directly improving the gross margin on the final API. The avoidance of column chromatography, which is often required in older methods to remove amine impurities, further reduces the consumption of silica gel and eluents, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reduced complexity of the raw material supply base. Sourcing high-purity solvents and managing their logistics can be a bottleneck, especially during global shortages or transport disruptions. Since this process operates neat, the dependency on external solvent suppliers is minimized, reducing the number of critical supply nodes. Furthermore, the one-pot nature of the reaction reduces the physical footprint required for production; fewer reactors and tanks are needed to achieve the same output volume compared to multi-step processes. This intensification allows existing facilities to increase capacity without major construction projects, ensuring that supply can scale rapidly to meet surges in demand for cardiovascular medications. The stability of the process also reduces the likelihood of batch failures due to moisture sensitivity or solvent quality issues, guaranteeing a consistent flow of goods to downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental risks, but this green methodology inherently mitigates them. The absence of large volumes of flammable solvents reduces the fire load in the production facility, lowering insurance premiums and safety compliance costs. From an environmental standpoint, the process aligns perfectly with Green Chemistry principles by preventing waste at the source rather than treating it after formation. The reduction in VOC emissions helps facilities meet stringent air quality standards, avoiding potential fines and shutdowns. The simplified effluent profile, devoid of high ammonia nitrogen loads, makes wastewater treatment more straightforward and less energy-intensive. This environmental stewardship not only protects the corporate reputation of the supplier but also future-proofs the supply chain against tightening global regulations on chemical manufacturing, ensuring long-term business continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational benefits for potential partners. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing production lines or for qualifying new suppliers who claim to utilize this advanced methodology.
Q: How does this solvent-free method improve environmental compliance compared to traditional routes?
A: Traditional methods utilize organic solvents like dichloromethane and acid-binding agents like triethylamine, generating significant wastewater with high COD and ammonia nitrogen. This patented green synthesis eliminates both solvents and acid binders, drastically reducing hazardous waste generation and simplifying effluent treatment protocols.
Q: What are the yield and purity benchmarks achieved in the examples?
A: The patent examples demonstrate robust performance with yields ranging from 85.8% to 93% and product purity consistently exceeding 98.9%. Specifically, Example 1 achieved a 93% yield with 99.1% purity, validating the efficiency of the one-pot thermal control strategy.
Q: Why is the one-pot technique critical for supply chain stability?
A: By combining acyl chlorination and thioesterification in a single vessel without intermediate isolation, the process minimizes material transfer steps. This reduces the risk of exposure to malodorous intermediates, lowers labor costs, and shortens the overall production cycle time, ensuring more reliable delivery schedules.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl 3-[(2,2-dimethyl-1-oxobutyl)thio]propionate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is not just an environmental imperative but a commercial necessity. Our technical team has extensively analyzed the potential of the one-pot solvent-free method described in CN110963951A and possesses the engineering expertise to translate this laboratory-scale innovation into robust industrial reality. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the thermal dynamics and mixing efficiencies required for solvent-free reactions are perfectly managed at scale. Our facilities are equipped with rigorous QC labs capable of detecting trace impurities down to ppm levels, guaranteeing that every batch of simvastatin side chain meets stringent purity specifications required by global regulatory bodies. We are committed to delivering a product that not only performs exceptionally in downstream coupling reactions but also supports your sustainability goals.
We invite you to collaborate with us to optimize your supply chain for statin production. By leveraging our technical capabilities, you can access a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this green route for your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data from our pilot runs and to discuss route feasibility assessments tailored to your current manufacturing constraints. Let us help you secure a supply of high-quality intermediates that drives both profitability and environmental responsibility.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
