Advanced Synthetic Route for Alpha-Terpineol: High Selectivity and Scalable Manufacturing
Advanced Synthetic Route for Alpha-Terpineol: High Selectivity and Scalable Manufacturing
The global demand for alpha-terpineol, a monocyclic terpene alcohol with a distinctive lilac odor, continues to surge across the fragrance, pharmaceutical, and industrial solvent sectors. Traditionally sourced from natural essential oils or synthesized via the hydration of alpha-pinene, the supply chain for this critical chemical has long been plagued by volatility in raw material pricing and inconsistent purity profiles. However, a groundbreaking technological shift is emerging, anchored by the innovations detailed in patent CN114315512A. This patent discloses a sophisticated synthetic methodology that bypasses the limitations of natural extraction and harsh acid hydration, offering a robust, high-yield alternative for the production of high-purity alpha-terpineol. By leveraging a novel Diels-Alder cycloaddition strategy catalyzed by a unique dual-acid system, this process achieves exceptional conversion rates and selectivity, positioning it as a superior choice for discerning reliable flavors & fragrances supplier networks seeking stability and quality.
The core innovation lies in the strategic coupling of 2-methyl-3-buten-2-ol esters with isoprene. Unlike conventional methods that struggle with side reactions and equipment corrosion, this new route operates under remarkably mild conditions, typically between 30°C and 120°C, with optimal performance observed at 50-60°C. The utilization of a dual catalyst system—comprising a Lewis acid and a specialized organic phosphoric acid—facilitates a highly controlled reaction environment. This precision not only maximizes the yield of the desired alpha-terpineol ester intermediate but also drastically minimizes the formation of unwanted byproducts, thereby streamlining downstream purification processes and ensuring a final product that meets the stringent specifications required for fine chemical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
For decades, the industrial production of alpha-terpineol has relied heavily on the hydration of alpha-pinene or crude turpentine oil. While these methods utilize renewable resources, they are fraught with significant technical and economic drawbacks that hinder modern manufacturing efficiency. The traditional one-step or two-step hydration processes typically employ strong Brønsted acids, such as sulfuric acid or phosphoric acid, as catalysts. These corrosive reagents necessitate the use of expensive, corrosion-resistant reactor materials, driving up capital expenditure and maintenance costs. Furthermore, the reaction kinetics in these acidic environments are difficult to control precisely, often leading to rapid, exothermic reactions that compromise selectivity. The result is a crude product mixture containing substantial amounts of beta- and gamma-terpineol isomers, as well as polymeric byproducts, which complicates purification and lowers the overall yield of the target alpha-isomer.
Beyond the technical challenges, the supply chain for traditional raw materials introduces unacceptable volatility for procurement managers. Turpentine and alpha-pinene are natural products whose availability and pricing are susceptible to fluctuations in labor costs, seasonal weather patterns, and geopolitical factors affecting forestry industries. As noted in the background of the patent data, the rising cost of labor in key producing regions has directly inflated the price of downstream terpineol derivatives. Additionally, the environmental footprint of the traditional acid-catalyzed route is substantial, generating significant volumes of acidic wastewater that require costly treatment before disposal. These cumulative factors create a fragile supply chain that struggles to meet the consistent quality and volume demands of high-end fine chemical intermediates markets.
The Novel Approach
In stark contrast to the legacy hydration technologies, the synthetic method disclosed in patent CN114315512A represents a paradigm shift towards precision organic synthesis. This novel approach constructs the terpineol skeleton de novo via a Diels-Alder reaction between a specifically activated dienophile, 2-methyl-3-buten-2-ol ester, and the diene, isoprene. This retrosynthetic disconnection allows for the use of abundant, petrochemical-derived bulk feedstocks that are not subject to the same agricultural volatilities as turpentine. The reaction is orchestrated by a synergistic dual-acid catalyst system, which activates the ester substrate sufficiently to react with the electron-rich isoprene without requiring the extreme conditions that lead to degradation and polymerization.
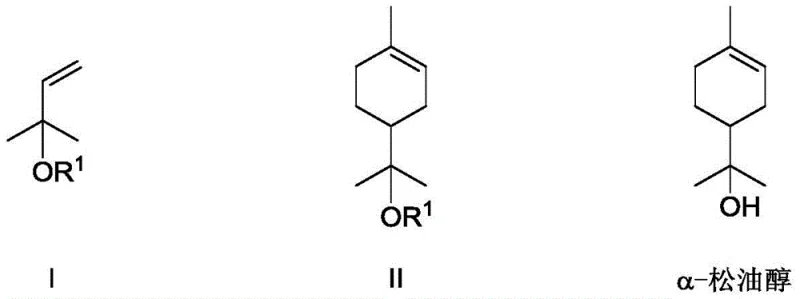
The operational advantages of this new route are profound. The reaction proceeds with high regioselectivity and stereoselectivity, yielding the alpha-terpineol ester intermediate with conversion rates reaching up to 97% and selectivity up to 98% under optimized conditions. Following the cycloaddition, the ester group is easily cleaved via a mild alkaline hydrolysis step, typically using sodium hydroxide or potassium hydroxide at temperatures as low as 25-35°C. This two-step sequence eliminates the need for corrosive mineral acids entirely, thereby reducing equipment corrosion and environmental waste. For a cost reduction in flavors & fragrances manufacturing, this translates to lower operational expenditures, simplified waste management, and a more predictable production schedule that is decoupled from the vagaries of natural resource harvesting.
Mechanistic Insights into Dual-Acid Catalyzed Diels-Alder Cycloaddition
The success of this synthetic route hinges on the intricate interplay between the Lewis acid and the organic phosphoric acid catalyst. In the first step, the Lewis acid (such as indium fluoride, zinc chloride, or scandium trifluoromethanesulfonate) coordinates with the oxygen atom of the ester moiety in the 2-methyl-3-buten-2-ol derivative. This coordination withdraws electron density from the alkene double bond, effectively lowering the energy of the Lowest Unoccupied Molecular Orbital (LUMO) of the dienophile. Simultaneously, the organic phosphoric acid component of the catalyst system likely engages in hydrogen bonding or further Lewis acid-base interactions that stabilize the transition state. This dual activation renders the substrate sufficiently electrophilic to undergo cycloaddition with isoprene, which acts as the electron-rich diene, even at moderate temperatures.
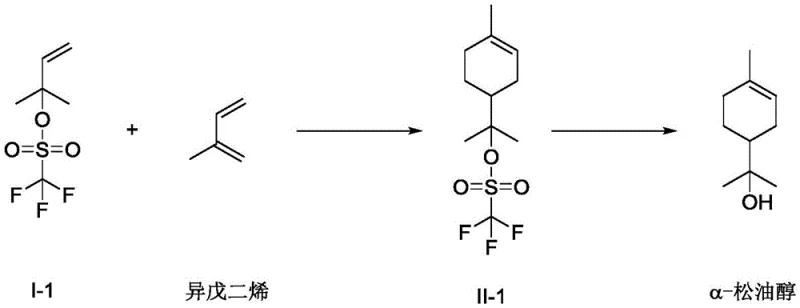
This mechanistic pathway offers superior control over impurity profiles compared to carbocation-mediated hydration. In traditional acid hydration, the formation of a carbocation intermediate often leads to Wagner-Meerwein rearrangements and polymerization, generating a complex soup of terpenes. In the Diels-Alder route, the concerted nature of the cycloaddition (or a tightly controlled stepwise ionic mechanism facilitated by the catalyst) ensures that the carbon skeleton is assembled with high fidelity. The subsequent hydrolysis step is equally clean; the use of mild bases like triethylamine or aqueous sodium hydroxide prevents the acid-catalyzed dehydration or isomerization of the newly formed terpineol. This rigorous control over the reaction mechanism ensures that the final high-purity alpha-terpineol contains minimal isomeric impurities, reducing the burden on distillation columns and ensuring a product that is ready for sensitive applications in perfumery and pharmaceuticals.
How to Synthesize Alpha-Terpineol Efficiently
The synthesis of alpha-terpineol via this patented route is designed for operational simplicity and scalability. The process begins with the preparation of the activated ester precursor, which can be synthesized from commercially available 2-methyl-3-buten-2-ol using standard acylation or sulfonylation techniques. Once the precursor is secured, the core Diels-Alder transformation is executed in a pressure-resistant reactor under an inert atmosphere. The precise stoichiometry of the catalyst system is critical; typical loadings involve 1-10 mol% of the Lewis acid and 2-20 mol% of the organic phosphoric acid relative to the isoprene. The reaction mixture is heated to the optimal range of 50-60°C for 1 to 2 hours, after which the crude ester is isolated via simple aqueous workup. The detailed standardized synthesis steps, including specific reagent quantities and workup procedures, are outlined below.
- Perform a Diels-Alder reaction between 2-methyl-3-buten-2-ol ester and isoprene using a dual catalyst system of Lewis acid and organic phosphoric acid at 30-120°C.
- Isolate the crude alpha-terpineol ester intermediate through aqueous washing and phase separation.
- Hydrolyze the ester intermediate using an alkali base such as sodium hydroxide or potassium hydroxide to yield the final alpha-terpineol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling strategic advantages that extend beyond mere chemical yield. The primary benefit is the decoupling of production from the volatile turpentine market. By shifting to petrochemical-derived feedstocks like isoprene and simple alcohols, manufacturers can secure long-term supply contracts with stable pricing, insulating their operations from the seasonal and geopolitical risks associated with forestry-based raw materials. Furthermore, the elimination of strong mineral acids from the process significantly reduces the total cost of ownership for production facilities. There is no longer a need for specialized Hastelloy reactors or extensive neutralization tanks for acidic effluent, allowing for the use of standard stainless steel equipment and simplifying regulatory compliance regarding wastewater discharge.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by both raw material stability and process efficiency. By utilizing bulk chemicals that are produced on a massive global scale, the input costs are inherently lower and more predictable than niche natural extracts. Additionally, the high selectivity of the reaction means that less raw material is wasted on byproducts, maximizing the atom economy of the process. The mild reaction conditions also translate to lower energy consumption, as there is no need for the high temperatures or pressures often required to drive equilibrium in hydration reactions. This cumulative effect results in substantial cost savings across the entire production lifecycle, enhancing the margin potential for downstream specialty chemical applications.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for any multinational corporation. The traditional reliance on turpentine creates a single point of failure in the supply chain, vulnerable to forest fires, labor strikes, or export restrictions. In contrast, the feedstocks for this Diels-Alder route—isoprene and various esters—are commodities produced by multiple suppliers worldwide. This diversification of the supply base ensures that production can continue uninterrupted even if one source faces disruption. Moreover, the simplicity of the two-step synthesis allows for faster turnaround times from order to delivery, enabling manufacturers to respond more agilely to spikes in market demand without the long lead times associated with harvesting and processing natural oleoresins.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen engineering challenges, particularly when dealing with exothermic reactions or corrosive media. This novel route is inherently scalable due to its mild thermal profile and the absence of hazardous corrosive agents. The reaction can be safely intensified in larger reactors without the risk of runaway exotherms that plague strong acid hydrations. From an environmental perspective, the process aligns with modern green chemistry principles by reducing waste generation and eliminating the release of sulfur-containing compounds. This facilitates easier permitting for new production lines and supports corporate sustainability goals, making it an attractive option for companies aiming to reduce their environmental footprint while expanding capacity for commercial scale-up of complex fragrance intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this alpha-terpineol synthesis technology. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, offering a transparent view of the process capabilities. Understanding these details is crucial for R&D teams evaluating the feasibility of technology transfer and for procurement specialists assessing the quality assurances of the final product.
Q: What are the advantages of the Diels-Alder route over traditional turpentine hydration?
A: The Diels-Alder route offers significantly higher selectivity (up to 98%) and avoids the use of corrosive strong mineral acids like sulfuric acid, reducing equipment maintenance and environmental waste compared to traditional hydration methods.
Q: What catalysts are used in this novel synthesis method?
A: The process utilizes a dual acid catalyst system comprising a Lewis acid (such as indium fluoride or zinc chloride) and a specialized organic phosphoric acid derivative to activate the dienophile effectively.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the reaction conditions are mild (50-60°C) and utilize bulk chemical raw materials like isoprene, making it highly scalable and less dependent on the fluctuating supply of natural turpentine.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Terpineol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO specializing in fine chemical intermediates, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art pressure reactors and distillation columns capable of handling the specific thermal and vacuum requirements of the Diels-Alder process described in patent CN114315512A. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of alpha-terpineol meets the exacting standards required for high-end fragrance and pharmaceutical applications, guaranteeing consistency and reliability for your global supply chain.
We invite you to collaborate with us to leverage this innovative technology for your product portfolio. Our technical team is prepared to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this synthetic route can optimize your bottom line. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample quantities. By partnering with NINGBO INNO PHARMCHEM, you secure not just a supplier, but a strategic ally committed to delivering high-quality reducing lead time for high-purity alpha-terpineol solutions that drive your business forward in a competitive market.
