Advanced Liquid Phase Synthesis of LKP Tripeptide for Commercial Scale-Up and High Purity
The global demand for effective antihypertensive agents has driven significant innovation in the field of bioactive peptides, specifically those capable of inhibiting Angiotensin-Converting Enzyme (ACE). Patent CN113461773A introduces a sophisticated liquid-phase synthesis method for LKP tripeptide (Leu-Lys-Pro), a potent ACE inhibitor derived from natural protein sources. This technology represents a critical advancement for manufacturers seeking to produce high-purity peptide intermediates for the functional food and pharmaceutical sectors. Unlike traditional extraction methods which suffer from low yields and complex purification, this synthetic route offers a controlled, reproducible pathway to generate LKP with exceptional efficiency. The process leverages a unique copper-complexation strategy to achieve orthogonal protection of lysine residues, a bottleneck in many peptide syntheses. For R&D directors and procurement specialists, understanding this methodology is essential for securing a reliable supply chain of high-value bioactive ingredients.
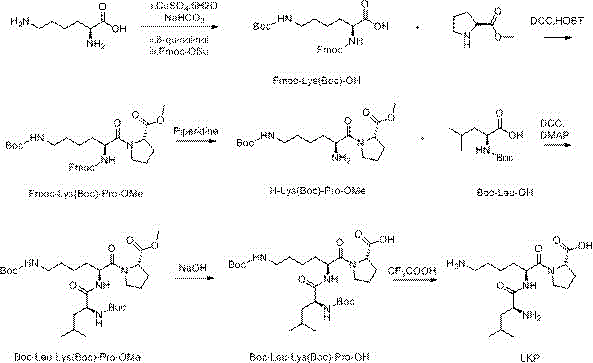
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tripeptides containing lysine has been plagued by the challenge of distinguishing between the alpha-amino and epsilon-amino groups. Conventional solid-phase peptide synthesis (SPPS), while versatile, often incurs high costs due to expensive resins and excessive solvent consumption, making it less ideal for the cost-sensitive functional food market. Furthermore, traditional liquid-phase methods frequently struggle with regioselectivity, leading to branched by-products where the peptide chain grows from the wrong nitrogen atom on the lysine side chain. These impurities are notoriously difficult to separate, resulting in lower overall yields and compromised purity profiles that fail to meet stringent regulatory standards for human consumption. The reliance on multiple protection and deprotection cycles without a robust differentiation strategy often extends lead times and increases the environmental footprint of the manufacturing process.
The Novel Approach
The methodology disclosed in CN113461773A circumvents these issues through an ingenious use of transition metal coordination chemistry. By initially complexing L-lysine with copper sulfate, the alpha-amino and carboxyl groups are effectively masked, leaving the epsilon-amino group exposed and available for selective reaction. This allows for the precise installation of the Boc protecting group exclusively at the epsilon position. Subsequent decomplexation using 8-hydroxyquinoline regenerates the alpha-amino functionality without disturbing the newly installed epsilon-protection. This sequence ensures that the subsequent coupling with proline and leucine proceeds with high fidelity. The result is a streamlined linear synthesis that minimizes side reactions, drastically simplifies purification protocols, and delivers the final LKP product with superior structural integrity and yield.
Mechanistic Insights into Copper-Mediated Selective Protection
The cornerstone of this synthesis is the formation of the bis-lysine copper(II) complex. In an aqueous environment, copper ions coordinate with the alpha-amino nitrogen and the carboxylate oxygen of the lysine molecule. This chelation effect significantly reduces the nucleophilicity of the alpha-amino group, rendering it inert to acylation by di-tert-butyl dicarbonate (Boc2O). Consequently, when Boc2O is introduced into the reaction mixture, typically in an aprotic solvent like acetone, it reacts preferentially with the uncoordinated and highly nucleophilic epsilon-amino group located on the lysine side chain. This chemoselectivity is paramount; without it, a mixture of alpha-Boc, epsilon-Boc, and di-Boc species would form, complicating downstream processing. The patent specifies maintaining a molar ratio of lysine to copper sulfate of approximately 1.0:0.5 to ensure complete complexation while avoiding excess metal waste.
Following the epsilon-protection, the copper must be removed to allow peptide bond formation at the alpha-position. The patent utilizes 8-hydroxyquinoline as a scavenging agent, which forms an insoluble green copper complex that can be easily filtered off. This step is crucial for metal clearance, ensuring the final peptide meets heavy metal specifications. Once the copper is removed, the free alpha-amino group is immediately capped with Fmoc-OSu (9-fluorenylmethoxycarbonyl succinimidyl ester). The choice of Fmoc for the alpha-position and Boc for the epsilon-position creates an orthogonal protection scheme. This means the Fmoc group can be removed later under mild basic conditions (using piperidine) without affecting the acid-labile Boc group on the side chain, allowing for the sequential assembly of the Leu-Lys-Pro sequence with absolute control over the peptide architecture.
How to Synthesize LKP Tripeptide Efficiently
The synthesis of LKP tripeptide via this liquid-phase route involves a logical sequence of seven distinct chemical transformations, starting from readily available amino acid derivatives. The process begins with the copper-mediated selective protection of lysine, followed by Fmoc installation to create the key building block, Fmoc-Lys(Boc)-OH. This intermediate is then coupled with proline methyl ester, deprotected, and elongated with Boc-leucine. The final steps involve ester hydrolysis and global deprotection. This structured approach ensures high purity at every stage, facilitating easier isolation of intermediates. For detailed operational parameters, stoichiometry, and specific workup procedures required to replicate this high-yield process, please refer to the standardized synthesis guide below.
- Complex L-lysine with copper sulfate to protect the alpha-amino and carboxyl groups, then selectively protect the epsilon-amino group with Boc anhydride.
- Decomplex the copper to reveal the alpha-amino group, then protect it with Fmoc-OSu to obtain Fmoc-Lys(Boc)-OH.
- Condense Fmoc-Lys(Boc)-OH with L-proline methyl ester, remove the Fmoc group, and couple with Boc-Leu-OH to form the protected tripeptide backbone.
- Hydrolyze the methyl ester to the carboxylic acid and finally remove the Boc protecting groups using trifluoroacetic acid to yield pure LKP.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this specific liquid-phase methodology offers tangible strategic benefits beyond mere technical feasibility. The reliance on commodity chemicals such as copper sulfate, sodium bicarbonate, and standard amino acids (L-lysine, L-proline, L-leucine) ensures that raw material sourcing is stable and resilient against market volatility. Unlike proprietary catalysts or exotic reagents that may face supply bottlenecks, the inputs for this process are globally available in bulk quantities. This accessibility translates directly into cost reduction in peptide intermediate manufacturing, as the bill of materials remains predictable and competitive. Furthermore, the high yields reported in the patent examples suggest a significant reduction in waste generation per kilogram of product, aligning with modern sustainability goals and reducing disposal costs.
- Cost Reduction in Manufacturing: The elimination of expensive solid-phase resins and the reduction in solvent volumes associated with washing cycles significantly lowers the operational expenditure. The high efficiency of the copper complexation step minimizes the formation of regioisomers, thereby reducing the need for costly preparative chromatography purification. By optimizing the stoichiometry of coupling reagents like DCC and HOBT, the process avoids excessive reagent waste, further driving down the cost of goods sold (COGS) for the final active ingredient.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which largely proceed at room temperature and utilize common solvents like dichloromethane and acetone, makes the process highly transferable across different manufacturing sites. This flexibility mitigates the risk of production delays caused by equipment incompatibility. Additionally, the stability of the protected intermediates, such as Fmoc-Lys(Boc)-OH and Boc-Leu-Lys(Boc)-Pro-OMe, allows for batch stocking, enabling manufacturers to buffer against sudden spikes in demand for the final LKP product without disrupting the production flow.
- Scalability and Environmental Compliance: Liquid phase synthesis is inherently easier to scale from pilot plant to multi-ton production compared to solid-phase methods. The ability to perform reactions in standard stirred-tank reactors simplifies the engineering requirements for commercial scale-up of complex peptide intermediates. Moreover, the process generates manageable waste streams; the copper by-product is precipitated and removed early, and the organic solvents used are standard and recyclable. This facilitates compliance with increasingly strict environmental regulations regarding heavy metal discharge and volatile organic compound (VOC) emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of LKP tripeptide. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is vital for partners looking to integrate this bioactive peptide into their product portfolios.
Q: What is the key advantage of using copper complexation in LKP synthesis?
A: Copper complexation allows for the selective protection of the epsilon-amino group of lysine while masking the alpha-amino and carboxyl groups. This orthogonal strategy simplifies the synthesis route, reduces side reactions, and significantly improves overall yield compared to traditional protection methods.
Q: Is this liquid phase method suitable for large-scale manufacturing?
A: Yes, the patent describes a robust liquid phase synthesis using common reagents like DCC, HOBT, and standard solvents (DCM, acetone). The high yields reported in the examples (often exceeding 90% for intermediate steps) indicate excellent scalability and cost-efficiency for industrial production.
Q: What are the primary applications of LKP tripeptide?
A: LKP tripeptide is a potent Angiotensin-Converting Enzyme (ACE) inhibitor. It is primarily used in the development of functional foods and nutraceuticals aimed at managing hypertension, offering a natural alternative to synthetic antihypertensive drugs with fewer side effects.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable LKP Tripeptide Supplier
As the market for functional food ingredients and nutraceuticals continues to expand, the ability to deliver high-purity bioactive peptides at scale is a decisive competitive advantage. NINGBO INNO PHARMCHEM stands ready to support your development goals with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-versed in the intricacies of liquid-phase peptide synthesis, including the specialized copper-complexation techniques described in recent patents. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of LKP tripeptide meets the highest standards for safety and efficacy, ready for formulation into antihypertensive supplements.
We invite you to collaborate with us to optimize your supply chain for ACE inhibitory peptides. Our experts can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our manufacturing efficiencies can improve your margins. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you bring safe, effective, and affordable blood pressure management solutions to the global market.
