Optimizing Octreotide Production: Advanced Solid-Liquid Phase Synthesis for Commercial Scale-up
Optimizing Octreotide Production: Advanced Solid-Liquid Phase Synthesis for Commercial Scale-up
The pharmaceutical industry continuously seeks robust manufacturing pathways for complex peptide therapeutics, particularly for high-demand molecules like Octreotide, a synthetic octapeptide analogue of somatostatin. A pivotal advancement in this domain is detailed in patent CN103102390A, which discloses a refined preparation method that strategically combines liquid-phase dipeptide synthesis with solid-phase hexapeptide assembly. This hybrid approach addresses critical bottlenecks in traditional peptide manufacturing, specifically targeting the enhancement of overall yield and the stringent control of impurity profiles. For R&D directors and procurement specialists evaluating potential partners, understanding the nuances of this technology is essential for securing a reliable API intermediate supplier capable of meeting global regulatory standards. The method described eliminates several cumbersome deprotection steps inherent in older methodologies, thereby streamlining the production workflow and reducing the cumulative loss of material at each stage of the synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Octreotide has been plagued by inefficiencies associated with full solution-phase methods or purely solid-phase strategies that struggle with scale-up. Traditional fragment condensation approaches, such as the 3+5 or 2+6 strategies mentioned in prior art, often suffer from significant material loss during the coupling of large fragments, leading to suboptimal yields that drive up the cost of goods sold. Furthermore, conventional methods frequently require harsh conditions for protecting group removal, which can induce racemization of sensitive amino acid residues, compromising the stereochemical integrity of the final product. The accumulation of deletion sequences and incomplete reaction byproducts necessitates extensive and costly purification efforts, often involving multiple chromatography runs that reduce throughput. These technical limitations create substantial supply chain vulnerabilities, making it difficult for manufacturers to guarantee consistent batch-to-batch quality and timely delivery for commercial-scale operations.
The Novel Approach
The innovative strategy outlined in the patent data introduces a sophisticated solid-liquid phase synthesis method that effectively mitigates the drawbacks of legacy techniques. By synthesizing the D-Phe-Cys dipeptide in the liquid phase with Boc protection and assembling the remaining six amino acids on a chlorotrityl chloride (CTC) resin, the process optimizes the reactivity of each segment. This segmentation allows for rigorous quality control of the dipeptide building block before it is introduced to the solid support, ensuring that only high-purity materials proceed to the elongation phase. The subsequent condensation of the liquid dipeptide with the hexapeptide resin under alkaline conditions facilitates a high-efficiency coupling reaction without the need for intermediate deprotection steps that typically degrade yield. This streamlined workflow not only enhances the total recovery of the linear octapeptide but also simplifies the operational complexity, making it an ideal candidate for cost reduction in pharmaceutical manufacturing environments.

Mechanistic Insights into Solid-Liquid Phase Peptide Assembly
To fully appreciate the technical superiority of this synthesis route, one must examine the mechanistic details of the chain assembly and cyclization processes. The selection of 2-chlorotrityl chloride resin as the solid support is critical, as it allows for mild acidic cleavage conditions that preserve the acid-labile side-chain protecting groups until the final global deprotection step. During the solid-phase elongation, the use of Fmoc chemistry with activation agents like HOBT and DIC ensures rapid coupling kinetics while minimizing epimerization risks. The liquid-phase synthesis of the N-terminal dipeptide utilizes HOSU active esters, which provide excellent stability and reactivity, allowing for precise stoichiometric control during the final fragment condensation. This dual-phase strategy leverages the scalability of solution chemistry for the initial fragment and the purification advantages of solid-phase synthesis for the chain elongation, creating a synergistic effect that maximizes efficiency.
Following the assembly of the linear octapeptide, the formation of the critical disulfide bridge between the two cysteine residues is achieved through a controlled oxidative cyclization. The patent specifies dissolving the linear precursor in a DMF-aqueous solution with a specific volume fraction, adjusting the pH to a slightly alkaline range of 6.8 to 8.0 to facilitate thiolate anion formation. The addition of an oxidizing agent, such as dilute hydrogen peroxide or iodohydrin, in a molar excess drives the reaction to completion while the aqueous component helps solubilize the polar peptide intermediates. This specific solvent system is engineered to balance the solubility of the hydrophobic peptide backbone with the polarity required for the oxidation reaction, thereby suppressing intermolecular polymerization and favoring the formation of the desired intramolecular disulfide bond. The result is a crude product with a significantly cleaner profile, ready for the final high-resolution purification steps.
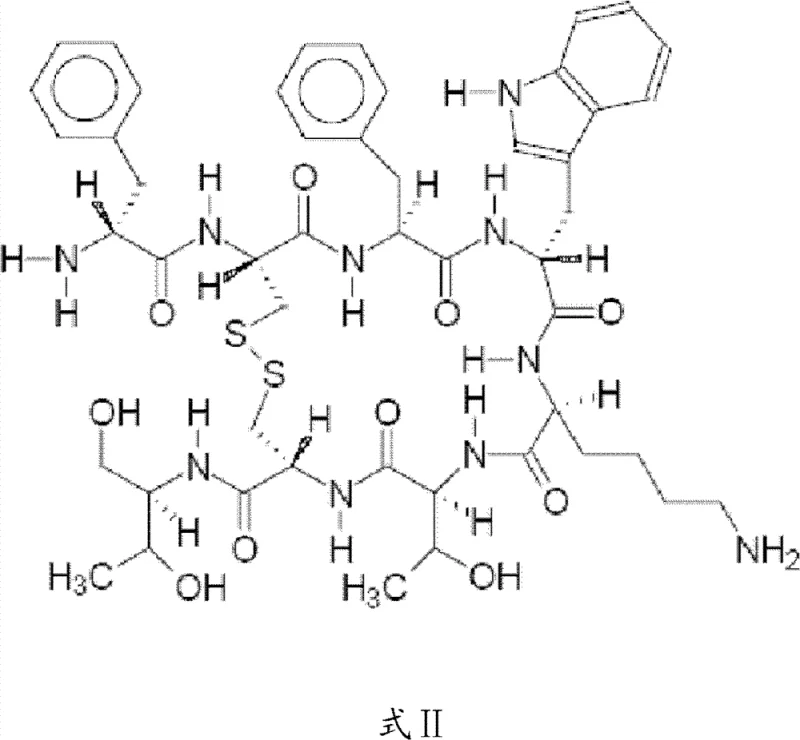
How to Synthesize Octreotide Efficiently
Implementing this synthesis protocol requires strict adherence to the defined reaction parameters to ensure reproducibility and high-quality output. The process begins with the preparation of the protected dipeptide and the stepwise loading of amino acids onto the resin, followed by the crucial condensation and cleavage steps. Detailed operational guidelines regarding solvent ratios, temperature controls, and reaction monitoring via HPLC are essential for successful execution. The following section outlines the standardized procedural framework derived from the patent specifications, serving as a foundational guide for process chemists aiming to replicate this high-yield pathway.
- Synthesize Boc-protected D-Phe-Cys dipeptide in liquid phase and assemble the hexapeptide resin sequence using Fmoc chemistry on CTC resin.
- Condense the liquid dipeptide with the solid hexapeptide resin under alkaline conditions, followed by cleavage to obtain the linear octapeptide.
- Perform oxidative cyclization in a DMF-water system using hydrogen peroxide or iodohydrin, followed by IP-RP-HPLC purification and lyophilization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this advanced synthesis methodology offers profound benefits for procurement managers and supply chain heads focused on operational excellence. The elimination of multiple protection and deprotection cycles inherently reduces the consumption of expensive reagents and solvents, directly contributing to substantial cost savings in the manufacturing process. By improving the overall yield of the linear octapeptide and the subsequent cyclization step, the process maximizes the output from raw materials, effectively lowering the unit cost of production without compromising on quality standards. This efficiency gain is particularly valuable in the volatile market of peptide APIs, where margin optimization is critical for maintaining competitiveness against generic alternatives.
- Cost Reduction in Manufacturing: The streamlined workflow significantly reduces the number of unit operations required to produce the final API, which translates to lower labor costs and reduced facility occupancy time. By avoiding the use of transition metal catalysts that require complex removal steps, the process simplifies the downstream purification train, further decreasing the operational expenditure associated with waste treatment and solvent recovery. The qualitative improvement in crude product quality means less burden on the preparative chromatography systems, extending column life and reducing the frequency of resin replacement, which are significant cost drivers in peptide manufacturing.
- Enhanced Supply Chain Reliability: The robustness of the solid-liquid phase approach ensures consistent batch-to-batch performance, which is a key factor in reducing lead time for high-purity pharmaceutical intermediates. The use of commercially available starting materials and standard resin technologies mitigates the risk of raw material shortages, ensuring a stable supply chain even during periods of market fluctuation. Furthermore, the scalability of the method allows manufacturers to rapidly ramp up production volumes to meet sudden spikes in demand, providing a strategic advantage for partners who require flexible and responsive supply solutions.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by optimizing solvent usage and minimizing the generation of hazardous waste streams. The ability to perform the cyclization in a DMF-water mixture reduces the reliance on purely organic solvents, aligning with increasingly stringent environmental regulations and corporate sustainability goals. This environmental compliance not only reduces regulatory risk but also enhances the brand reputation of the supply chain partners, making it a preferred choice for multinational corporations with strict vendor codes of conduct regarding ecological impact.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Octreotide using this specific patent methodology. These insights are derived directly from the experimental data and beneficial effects reported in the intellectual property documentation, providing clarity on the feasibility and advantages of the technology. Understanding these details is crucial for stakeholders evaluating the technical readiness and commercial viability of this synthesis route for their specific application needs.
Q: How does this method improve Octreotide purity compared to traditional solution synthesis?
A: By utilizing a solid-liquid phase hybrid approach, this method minimizes racemization during chain elongation and employs ion-pair reversed-phase chromatography to achieve purity levels exceeding 99%, significantly reducing difficult-to-remove impurities common in full solution synthesis.
Q: What are the advantages of the specific cyclization conditions described in the patent?
A: The use of a weakly polar DMF-aqueous solvent system with controlled pH (6.8-8.0) and specific oxidant ratios facilitates efficient disulfide bond formation while suppressing side reactions, leading to higher conversion rates and simplified downstream processing.
Q: Is this synthesis route scalable for industrial manufacturing?
A: Yes, the protocol avoids complex protection group manipulations and utilizes standard resin technologies and chromatography techniques that are readily transferable from laboratory to multi-kilogram production scales, ensuring consistent supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Octreotide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthesis technologies to deliver superior pharmaceutical intermediates to our global clientele. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of advanced patents like CN103102390A are fully realized in practical manufacturing settings. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of Octreotide meets the highest international standards for identity, potency, and impurity control.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our process efficiencies can translate into tangible value for your organization. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to quality and transparency as your trusted partner in fine chemical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
