Revolutionizing Adefovir Intermediate Production: A Deep Dive into One-Pot DiPMEA Synthesis
The pharmaceutical industry's relentless pursuit of cost-effective antiviral therapies has placed a premium on optimizing the synthesis of key nucleoside analog intermediates. Patent CN101607971B introduces a transformative methodology for producing 9-[2-(diethylphosphono methoxyl)ethyl]adenine, commonly known as DiPMEA, which serves as a critical precursor for the potent hepatitis B therapeutic, adefovir dipivoxil. This innovation addresses the longstanding inefficiencies inherent in multi-step batch processes by consolidating three distinct chemical transformations into a seamless, telescoped sequence. By integrating the in-situ generation of ethylene carbonate with the subsequent alkylation of adenine and final phosphorylation, the disclosed technology eliminates the need for isolating thermally sensitive intermediates. This strategic consolidation not only streamlines the operational workflow but also fundamentally alters the economic landscape of manufacturing this high-value pharmaceutical intermediate. For global supply chains, this represents a pivotal shift towards more resilient and sustainable production models capable of meeting the surging demand for chronic hepatitis B treatments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of DiPMEA has been plagued by a fragmented, linear approach that necessitates the discrete isolation of every intermediate species. As illustrated in the traditional pathway, the process begins with the separate preparation of ethylene carbonate, followed by its reaction with adenine to yield 9-(2-hydroxyethyl)adenine (HEAD), which must then be isolated before undergoing phosphorylation. This conventional wisdom dictates that each step requires its own dedicated workup, involving solvent exchanges, crystallization, and drying procedures that are both time-consuming and material-intensive. Specifically, the isolation of the HEAD intermediate often demands the use of additional solvents like toluene to facilitate purification, thereby inflating the environmental footprint and operational expenditure. Furthermore, the physical handling of intermediates inevitably leads to mechanical losses and potential degradation, capping the overall process yield at suboptimal levels, historically reported around 23%. Such inefficiencies create bottlenecks that hinder the ability of manufacturers to respond agilely to market fluctuations.
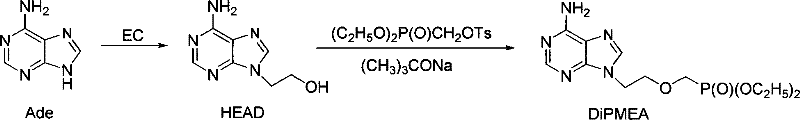
The Novel Approach
In stark contrast, the novel methodology described in the patent dismantles these barriers by adopting a telescoped, one-pot strategy that maintains the reaction momentum without interruption. Instead of halting the process to isolate ethylene carbonate or the HEAD intermediate, the new route allows the reaction mixture to evolve continuously from raw materials to the final product. The synthesis initiates with the formation of ethylene carbonate directly in the reactor, which is immediately consumed by adenine in the same medium. Without any purification break, the resulting hydroxyethyl-adenine species is directly phosphorylated using diethyl (tosyloxymethyl)phosphonate. This continuous flow of chemistry drastically reduces the number of unit operations, effectively collapsing a five-day production cycle into a mere three days. The elimination of intermediate isolation steps not only accelerates throughput but also significantly mitigates the risk of contamination and decomposition, paving the way for a more robust and economically viable manufacturing protocol.
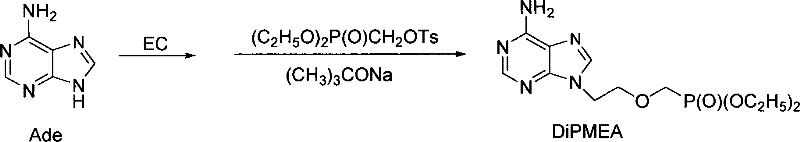
Mechanistic Insights into Tandem Alkylation and Phosphorylation
The core of this technological breakthrough lies in the precise orchestration of reaction conditions that allow disparate chemical transformations to coexist harmoniously within a single vessel. The process leverages the nucleophilic character of adenine, which attacks the electrophilic carbon of the in-situ generated ethylene carbonate under basic catalysis, typically utilizing sodium hydroxide or potassium carbonate in a polar aprotic solvent like DMF. This initial ring-opening alkylation generates the 9-substituted adenine derivative with high regioselectivity. Crucially, the reaction environment is maintained such that the newly formed hydroxyl group remains available for the subsequent nucleophilic substitution. Upon the addition of a stronger base, such as sodium tert-butoxide, the hydroxyl group is deprotonated to form an alkoxide, which then aggressively attacks the phosphonate reagent. This tandem mechanism ensures that the concentration of the reactive intermediate never drops to zero, driving the equilibrium forward and minimizing side reactions that typically occur during prolonged storage or handling of isolated intermediates.
From an impurity control perspective, this one-pot design offers a distinct advantage by preventing the accumulation of degradation products that often arise during the drying and redissolution of intermediates. In traditional methods, the exposure of the HEAD intermediate to air and moisture during isolation can lead to oxidation or hydrolysis, generating difficult-to-remove impurities that compromise the purity profile of the final API. By keeping the intermediate in solution under controlled inert atmosphere conditions, the novel process maintains a cleaner reaction profile. The use of specific bases and temperature gradients—cooling the mixture before adding the phosphonate reagent—further suppresses thermal degradation and ensures that the phosphorylation proceeds with high fidelity. This meticulous control over the reaction trajectory results in a crude product that requires less intensive downstream purification, thereby enhancing the overall mass balance and reducing the load on waste treatment facilities.
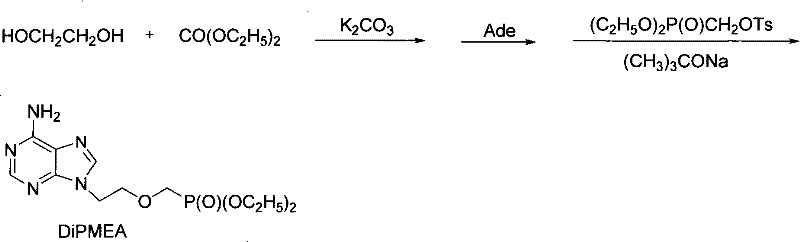
How to Synthesize 9-[2-(diethylphosphono methoxyl)ethyl]adenine Efficiently
Implementing this advanced synthesis requires a disciplined approach to reagent addition and thermal management to maximize the benefits of the telescoped design. The process begins with the generation of the cyclic carbonate precursor, followed immediately by the introduction of the nucleobase and finally the phosphonate coupling agent. Operators must adhere to strict temperature protocols, particularly during the exothermic phosphorylation step, to prevent runaway reactions and ensure safety. The following guide outlines the critical operational phases derived from the patent examples, serving as a foundational reference for process engineers aiming to replicate this high-efficiency route.
- In-situ generation of ethylene carbonate from ethylene glycol and diethyl carbonate under basic catalysis without isolation.
- Direct addition of adenine to the reaction mixture to form 9-(2-hydroxyethyl)adenine (HEAD) intermediate.
- Sequential addition of diethyl (tosyloxymethyl)phosphonate and strong base to complete the phosphorylation to DiPMEA.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this streamlined synthesis protocol translates into tangible strategic advantages that extend far beyond simple yield metrics. The primary value proposition lies in the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures and enhanced asset utilization. By eliminating the need for multiple isolation and purification stages, facilities can significantly lower their consumption of auxiliary solvents and reduce the energy burden associated with repeated distillation and drying cycles. This reduction in resource intensity not only lowers the variable cost per kilogram but also aligns production practices with increasingly stringent environmental regulations, mitigating the risk of compliance-related disruptions. Furthermore, the shortened production cycle time enhances the agility of the supply chain, allowing manufacturers to respond more rapidly to demand spikes without the need for excessive inventory buffering.
- Cost Reduction in Manufacturing: The economic impact of merging three reaction steps into one cannot be overstated, as it fundamentally removes the labor and utility costs associated with intermediate handling. By avoiding the isolation of the HEAD intermediate, the process eliminates the requirement for additional filtration, washing, and drying equipment capacity, effectively increasing the throughput of existing infrastructure without capital investment. The reduction in solvent usage, particularly the avoidance of toluene washes required in prior art methods, leads to substantial savings in raw material procurement and waste disposal fees. Additionally, the higher overall recovery rate means that less starting material is required to produce the same amount of final product, directly improving the margin structure for this pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: In the context of global pharmaceutical sourcing, consistency and speed are paramount, and this optimized route delivers on both fronts. The simplified process reduces the number of potential failure points; with fewer unit operations, there is less opportunity for human error or equipment malfunction to derail a batch. The ability to complete the synthesis in approximately three days, compared to the five or more days required by conventional methods, significantly compresses the lead time for order fulfillment. This acceleration allows supply chain planners to operate with leaner inventory levels while maintaining high service levels, thereby freeing up working capital and reducing the risk of obsolescence. Moreover, the robustness of the one-pot method ensures a more consistent quality profile, reducing the likelihood of batch rejections that can disrupt supply continuity.
- Scalability and Environmental Compliance: As the demand for antiviral therapies continues to grow, the ability to scale production efficiently is a critical competitive differentiator. This novel synthesis is inherently designed for scalability, as the telescoped nature of the reaction reduces the physical footprint required for manufacturing. The elimination of intermediate isolation steps simplifies the engineering controls needed for containment and transfer, making it easier to transition from pilot plant to commercial scale. From an environmental standpoint, the process generates significantly less waste solvent and solid byproducts, facilitating easier compliance with green chemistry initiatives. The reduced emission profile and lower waste generation not only minimize environmental impact but also streamline the permitting process for facility expansions, ensuring long-term operational viability in regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on how this technology can be integrated into existing manufacturing frameworks. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this method for large-scale production of antiviral intermediates.
Q: How does the one-pot method improve yield compared to traditional routes?
A: By eliminating the isolation of the unstable intermediate HEAD and the separate synthesis of ethylene carbonate, the process minimizes material loss during workup, increasing total recovery from approximately 23% to over 35%.
Q: What are the key cost drivers reduced in this new synthesis?
A: The method significantly reduces solvent consumption by removing the need for toluene washing steps associated with intermediate isolation and shortens the overall production cycle from over five days to approximately three days.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the simplified operation, reduced number of unit operations, and elimination of complex purification steps for intermediates make it highly scalable and environmentally friendly for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable DiPMEA Supplier
The technical advancements detailed in patent CN101607971B underscore the immense potential for optimizing the production of critical antiviral intermediates like DiPMEA. At NINGBO INNO PHARMCHEM, we recognize that translating such innovative laboratory methodologies into robust commercial processes requires deep expertise and state-of-the-art infrastructure. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of one-pot synthesis are fully realized in practice. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that validate every batch against the highest international standards. We understand that for our partners, consistency is key, and our advanced manufacturing capabilities are designed to deliver high-purity intermediates with unmatched reliability.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage these process efficiencies for their supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our mastery of advanced synthetic techniques can drive down your costs and secure your supply of essential pharmaceutical intermediates for the future.
